| EIKEN CHEMICAL (4549) |
|
||||||||
Company |
EIKEN CHEMICAL CO., LTD. |
||
Code No. |
4549 |
||
Exchange |
Tokyo Stock Exchange, First Section |
||
Industry |
Pharmaceuticals (manufacturing and sales) |
||
President |
Morifumi Wada |
||
HQ Address |
7 Yamaguchi building, 4-19-9 Taito, Taito-ku, Tokyo 110-8408, Japan |
||
Year-end |
End of March |
||
URL |
|||
*Share price is as of the end of May 20. The number of shares issued is from the latest financial settlement report (excluding treasury shares from the number of shares issued). ROE and BPS are based on actual results at the end of the previous term.
|
||||||||||||||||||||||||
|
|
|
| Key Points |
 |
| Company Overview |
|
It offers many products that occupy high market share including fecal immunochemical test reagents that occupy about 57% of the domestic share. Its unique gene amplification technology, "LAMP", is highly recognized in the world. With the fecal immunochemical test reagents and LAMP, EIKEN is aiming to become a global corporation.
"Management Vision": EIKEN group is dedicated to leveraging expertise as a medical testing pioneer in order to increase corporate value by protecting the health of the public with products and services that customers can trust. "Motto": We EIKEN provide trustworthy quality, and develop with technology. EIKEN group formulates "EIKEN WAY" as its attitude toward each stakeholder, centering these philosophy, vision and motto. 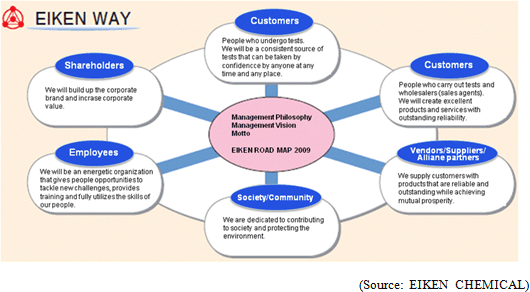 <Domestic Market>
The market scale of clinical reagents (excluding diagnostic devices) is about 338 billion yen as of 2013 (survey by the Japan Association of Clinical Reagents Industries, or JACRI). In order to control rising medical costs, the Japanese government is focusing on preventive medicine such as special health check-ups (metabolic check-ups) and cancer screenings. It is expected that this, along with the aging population, will lead to an increase in the number of samples (number of specimens). Some negative factors include the impact of population decline as a result of decreasing birth rates and revision of medical treatment fees (reduction). However, the trends of laboratory test fees which had been subject to revision of insurance (medical laboratory test fees) show that, even though they were cut by some 40% from 1997 to 2006, the fees have been stable or only slightly reduced after 2007. This is a result of advocacy on the importance of prevention and testing by the entire industry including Eiken. Thus, in the mid to long term, the domestic market is expected to slightly grow by 3% each year. According to the "Prospect of Clinical Laboratory Test Market, 2009" conducted by Yano Research Institute, Eiken Chemical ranked the 5th largest, occupying 5.8% market share, after Sysmex (6869, first section of Tokyo Stock Exchange), Roche Diagnostics (Japanese corporation of the Roche Group in Germany), Fujirebio (now known as Miraca Holdings. 4544, first section of Tokyo Stock Exchange), and Abbott Japan (Japanese corporation of the Abbott Group in the US). Out of the 115 member companies (as of April 2015) of JACRI mentioned above, about 80 are manufacturers, and there are about 15 companies with over 10 billion yen in sales. Most of them are small to medium sized companies. Because the test items of diagnostic tests range widely, each company has its own field of strength, and business segregation is already established in the industry. As a result, collaboration, such as supplying raw materials and products from other companies and manufacturing and selling them, is often observed. Against such a backdrop, the market is modestly growing. Therefore, there is currently no apparent trend of weeding out uncompetitive corporations. <Overseas Market>
According to the "Prospect of Global Clinical Laboratory Test Market, 2009" conducted by Yano Research Institute, the global clinical laboratory test reagent/device market is US$ 44.5 billion or 4.45 trillion yen (1 USD=100 yen), and, by region, the market is occupied by the USA at 41.2%, followed by Europe at 36.9% and Asia/Pacific at 12.0%. The overseas market is over ten times larger than the domestic market. In developed countries, the number of tests is increasing as aging of population progresses. Furthermore, in emerging countries, the needs for medical services are expanding because of economic and income growth. As a result, the annual growth rate of overseas market is expected to be 7 to 8%, which is much higher than that of the domestic market. Therefore, the Japanese companies in the industry are vigorously undertaking globalization of their businesses. In the global market, the global large companies such as Roche, Abbott, SIEMENS, and Beckman, whose sales are 200,000 to 900,000 million yen, are leading the market, and in order for Japanese companies to survive the competition, they must strengthen their competitiveness by, for example, developing unique products or systems. 1. What are Clinical Tests?
One type of clinical tests is the "biological test" that directly examines the body using medical equipment such as X-ray, CT, MRI, electrocardiogram, and ultrasound. Another type of clinical tests is the "medical laboratory test" that examines biological samples (specimens) obtained from people such as blood, urine/feces, and cells.The clinical test reagents made by Eiken Chemical are the ones used for medical laboratory tests. For example, they are used to test infectious diseases or to measure small amounts of blood contained in stool. They are made to support diagnosis. Most of these reagents are called in vitro diagnostics (IVD) and are regulated by the Pharmaceutical and Medical Device Act so reagent manufacturers file applications with PMDA (Pharmaceuticals and Medical Devices Agency) and obtain its approval. Users include hospitals, clinics, medical offices, medical test centers that carry out tests commissioned by medical institutions, health screening centers, public health centers, and institutions for health research, etc. 2. Major Products
Eiken Chemical mainly manufactures and sells the following types of reagents and medical devices.As they deal with a wide range of reagents, they not only sell their in-house products but also purchase and sell products from other companies. Major in-house products include fecal immunochemical test reagents, microbiological reagents, immunological and serological reagents, urinalysis test strips, genetic testing reagents, etc. The sales ratio of in-house products to other companies' products is approximately 60:40. The gross profit margin is approximately 55% for in-house products and approximately 35% for other companies' products. 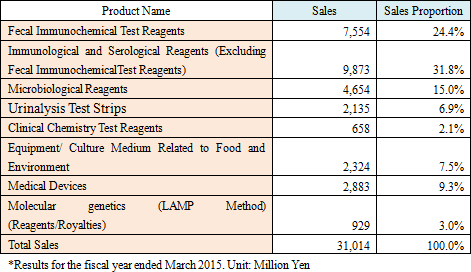 The major products for Eiken Chemical are reagents and sampling bottles for fecal immunochemical tests to specifically detect and measure human hemoglobin in feces as a colon cancer screening and diagnosis and are sold globally. Immunological and Serological Reagents (Excluding Fecal Immunochemical Test Reagents)
Starting with reagents for "LZ Test 'EIKEN'," an automated analyzer, Eiken Chemical develops, manufactures, and sells various reagents to diagnose and measure infectious disease, rheumatism, inflammation, atrophic gastritis, and prostate specific antigens.They also install and sell reagents for fully automated enzyme immunoassay devices and reagents for automatic glycohemoglobin analyzers from Tosoh Corporation. Microbiological Reagents
Since its establishment, Eiken Chemical has been developing biological specimens as well as reagents for microbiological tests for food and environment in order to prevent infectious diseases and food poisoning. Currently, it develops, manufactures and sells various reagents that are effective for diagnosis and treatment of microorganism infection, such as mediums, powder mediums, drug sensitivity test reagents, and rapid test reagents.Urinalysis Test Strips
Eiken Chemical develops, manufactures, and sells "UROPAPER III 'EIKEN'," a urine analysis test strip for testing various items such as occult blood, protein and glucose, as well as the "UROPAPER - III 'EIKEN'," a specialized test strip for fully automated urine analyzers.
Clinical Chemistry Test Reagents
Eiken Chemical develops, manufactures and sells reagents for clinical chemistry tests including "EXDIA XL 'EIKEN'" series that assist to measure and analyze biological components in blood serum and urine, with a focus on the test items that are related to lifestyle related diseases.
Equipment/ Culture Mediums Related to Food and Environment
Eiken Chemical sells reagents for microbiological tests on food to detect food poisoning bacteria as well as reagents for environmental microbiological tests and equipment and devices to measure contamination of work environments.
Medical Devices
Eiken Chemical sells various types of automated analyzers. They contract manufacturing specialized equipment that uses their in-house reagent. Since beginning sales of "OC Sensor" in 1989, they have worked continuously on technological innovation and quality improvement of this fecal immunochemical test analyzer. Also, they offer the "US," an automated urine analysis device that uses Eiken's proprietary color CCD sensor, the "BLEIA-1200," a fully automated biochemistry photogenetic immunoassay device that was the world's first of its kind in the clinical testing field, and "LoopampEXIA," a LAMP-based real time turbidity measuring device.
Molecular genetics (LAMP)
In 1998, Eiken Chemical developed and patented an innovative gene amplification technology called "LAMP." The LAMP is "simple, rapid, and accurate" and is a critical tool for Eiken's future global expansion of its business. (Details are described below)
3. Sales structure
EIKEN CHEMICAL has 11 sales offices and 2 sales divisions in Japan. Its academic department supports sales promotion. Out of 620 employees (non-consolidated) during FY 2015, about 290 belong to the sales department. As for the sales channels for medical institutions such as hospitals, the Company's direct sales partners are medical wholesale companies, and it has businesses with almost all of the wholesale companies in the medical industry. For overseas sales, EIKEN CHEMICAL has basically 1 agency per country, and the sales and maintenance are commissioned to the agencies. EIKEN's products are exported to 51 countries (FY 2015). The high proportion of overseas sales is occupied by the sales in the USA, Italy, South Korea, and Taiwan. In addition to the Europe Branch in Amsterdam (the Netherlands), the Company is strengthening its manufacturing and sales structure through its consolidated subsidiary, "EIKEN CHINA CO., LTD.," as well as aiming to expand its businesses by setting a business office in China. In the future, it will explore the possibility of making the office as a local corporation, as the size expands. The overseas sales for FY 2015 are 27.04 billion yen, out of which 22.94 billion yen, 84.8%, is from the sales of fecal immunochemical test reagents and medical devices. 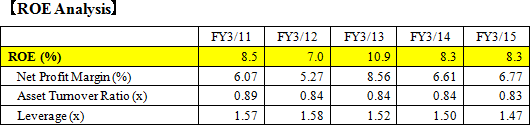 (The reason for the high ROE in FY 2013 was the increased net income margin from posting extraordinary income from land sales.) However, it is foreseen that there will be a greater call for further increase in ROE. So with the company's stated goal of developing products with high added value, it will be necessary to further increase productivity and profitability by creating new business and markets as well as lowering cost rate and SG&A expense rate. (1) Products that Occupy High Share in the Market
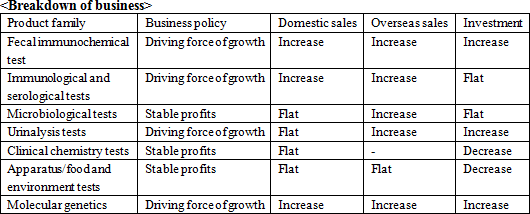
The share of Eiken's fecal immunochemical test reagents is ranked top (approximately 57%) in the domestic market. Furthermore, many of their in-house products occupy high market share in the market, for example, urinalysis test strips occupying approximately 23% (ranked second) of the market, and microbiological reagents occupying approximately 18% (ranked second) of the market.The background to how Eiken's fecal immunochemical test reagents have come to hold such a high share of the market includes that in 1987, Eiken began sales of "OC-Hemodia," a visual determination method fecal immunochemical test reagents, a product that more closely conformed to user needs when compared to competitor's products, and that in 1989 they adopted the latex photometric immunoassay method and began sales of "OC-Sensor," the world's first fully automated analyzer. Also, the Health and Medical Service Act for the Aged was revised in 1992, making it possible to have fecal immunochemical test reagents as a method in colon cancer screening and diagnosis using public funds (no cost to the patient) which led to an accelerated spread and increased competition. But in 2001, Eiken began sales of the "OC-Sensor neo," with completely remodeled functions, which increased its market share.  Meanwhile, in other countries, reagents for the chemical method (Guaiac method) based on old measuring principles are still used, which presents accuracy challenges. In 2011, the test guidelines in Europe have finally begun recommending automated analyzers that use the immunochemical method. As a result, the market is beginning to undergo a dramatic change. Furthermore, although the chemical method is also still common in the United States, which has the largest potential market, trends show a gradual shift toward the immunochemical method. This means that, in both developed and emerging countries in Europe, North America, Asia and Oceania, there is a large unexplored market. Because the fecal immunochemical tests market is a niche market, Japanese companies, the forerunners of the immunochemical method, own the most advanced technique, and hence Eiken's reagents and equipment are the global standard. (2) Focusing on research and development
EIKEN CHEMICAL is focusing on research and development of unique technologies as a research and development corporation, and the development of original products that respond to customers' needs, using the unique technologies. The number of staff assigned for research and development is about 100. The demand from the customers is higher quality of medicine. Specifically, they demand for higher differential diagnosis accuracy with high sensitivity and high quality and improved detection rate. In addition, easier usage will lead to reduction in the work of medical staff. Responding to such needs is critical. Since its establishment in 1939, EIKEN CHEMICAL has accumulated unique technologies for manufacturing reagents. Their unique technologies are applied to the measuring principles of their devices such as fecal occult blood test analyzer, automated urine analyzer, and biochemiluminescent immunoassay analyzer "BLEIA" that are designed to optimize the performance of the reagents. (3) Development of various types of products in various fields through alliance strategy
Because clinical test reagents have wide range of subjects and items, it is not possible for one company to develop, manufacture and sell all types of reagents. The other companies in the industry are focusing on the technologies and products that they are specialized in. However, as an integrated manufacturer of clinical test reagents, EIKEN CHEMICAL aims at stabilizing profit structure, expanding their own strengths through alliance strategy, and pursuing synergy effects such as complementing functions and acquiring new technologies, while dealing with a wide range of products and responding to the needs of customers and users such as medical institutions. Another reason why they cover various types of products in various fields is that they believe that covering wide range of clinical tests is their social responsibility to protect the health of the public, as is stated in their management philosophy: "protect the health of the public through health care services". (4) Competitive Advantages of the "LAMP"
Thus far the mainstream technology for amplifying genes as a process of gene tests has been what is called "PCR" Under such circumstances, in 1998, Eiken Chemical developed a unique technology called the "LAMP."Compared to the PCR, the "LAMP" offers the following superior characteristics and allows users to carry out simple, rapid and accurate gene tests.  Eiken Chemical is making focused efforts on infectious disease diagnostic test in order to establish the status of the LAMP. At the same time, it is promoting the use of the LAMP in other fields such as food production and processing, environment, agriculture/veterinary in order to spread and enhance recognition of the LAMP. In fact, the LAMP-based products have been commercialized one after another since 2002. Furthermore, for the same purposes, Eiken Chemical is actively giving licenses to external companies in order to build the LAMP camp. One of the major actions to spread the LAMP in the world is an alliance with "FIND." "FIND" stands for "Foundation for Innovative New Diagnostics" and is a non-profit organization recognized by the Swiss government, launched at a meeting of the United Nations World Health Assembly in May 2003. In its initial five years of existence, it received a grant from the Bill & Melinda Gates Foundation to start up their activities. Their goal is to develop and introduce affordable, simple and advanced diagnostic tests in order to eradicate infectious diseases in developing countries. FIND's scope of activities includes tuberculosis, malaria, and African sleeping disease. With tuberculosis, collaborative research between Eiken Chemical and FIND for a tuberculosis test using the LAMP began in July 2005. The purpose of this research is to improve the accuracy of tests by replacing the microscopy test (sputum smear test), which is the current practice in developing countries. As a result of this collaboration, improvements which are not possible with the conventional PCR such as simplified pretreatment (PURE method), improved reagents storage (store at room temperature) and simplified devices have been made to enable the developing countries to carry out the procedure. This LAMP-based product was already launched in Japan in 2011. Presently, in order to obtain endorsement from the WHO (World Health Organization), FIND has completed clinical evaluation in 14 developing countries and submitted this information to the WHO. In addition to tuberculosis and other diseases listed above, Eiken Chemical and FIND also conduct collaborative research of reagents for leishmaniasis and Chagas disease. Also, Eiken Chemical is developing a next-generation compact fully automated genetic testing device and multi-item testing chip using the LAMP. This equipment fully automates the process from specimen preprocessing (nucleic acid extraction and purification) to amplification and detection. By developing the unique protocol that exploits the LAMP's characteristics, the operation time that used to take over 2 hours with a conventional high purity nucleic acid extraction and purification device and an amplification and detection device combined, is now shortened to less than 30 minutes. First, a clinical performance test will be carried out with the goal of simultaneous detection of several respiratory infection causing germs but the usage application is extensive. It is anticipated that through these products, Eiken Chemical will accelerate the spread of the LAMP and establish its position as the global standard in a newly created market. ∗Gene amplification technology
Since the amount of genes found in a genetic test sample is extremely small, in order to detect genes, the targeted gene must be amplified first of all. Gene amplification technology, therefore, is crucially important for genetic testing.
∗African trypanosomiasis
An endemic found in tropical Africa, African trypanosomiasis is a serious tropical disease transmitted to humans by a protozoa called Trypanosoma brucei. The disease is transmitted by a tsetse fly. Trypanosoma in human blood sucked by a tsetse fly develops and propagates inside the human body in 2 to 5 weeks, before turning itself into a terminal Trypanosoma-type, which becomes a source of next round of infection. The disease causes fever, headache, and vomiting, and the patient falls into constant sleep. Since the patient cannot take meals, he or she becomes thin and complain of generalized weakness and, in many cases, leads to a complication and dies.
∗Leishmaniasis
Leishmaniasis is a disease transmitted by a protozoa called leishmania, and has various types such as visceral leishmaniasis (also known as black fever), Brazilian leishmaniasis that affects skin and mucous membranes, and tropical leishmaniasis which affects skin. All of these types are transmitted by blood-sucking insects, especially sandflies. Visceral leishmaniasis, after about three months incubation period, causes fever, sweating, diarrhea, etc. and, in about one month, causes a swollen liver and spleen, the patient develops an anemia and becomes weak if untreated, and may die in half a year to two years.
∗Chagas disease
Found in southern U.S. as well as Central and South America, Chagas disease is an infectious disease transmitted by Reduviidae, a kind of blood-sucking Triatominae. The disease does not develop symptoms immediately after infection; it usually has a latency period of about 30 years. It causes symptoms such as inflammation of sinews, liver and spleen, myalgia, myocarditis, cardiomegalia, encephalomyelitis, cardiac disturbance, etc.
|
| Fiscal Year March 2015 Earnings Results |
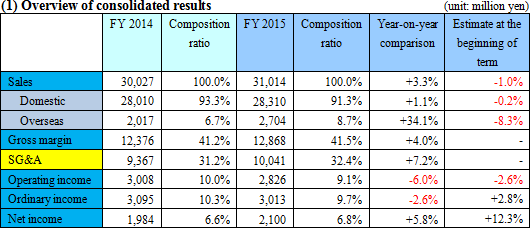 Increase in sales and operating income due to increased overseas sales
Sales increased by 3.3% from the previous term to 31,000 million yen. Domestic sales increased slightly by 1.1% from the previous term despite the negative rebound from the last-minute demand prior to the consumption tax hike and the effect of revised medical treatment fees. In terms of product category, urinalysis test strips, ABC classification reagents, and immunochemical fecal occult blood test reagents contributed. Overseas sale grew significantly by 34.1% from the previous term, mostly from fecal immunochemical test reagents and medical devices.Gross margin increased by 0.3%, but the increase in R&D expenditures, and drop in Eiken China performance resulted in a 6.0% decrease from the previous term in operating income. Due to the decrease in corporate taxes from the tax reform of 2014, net income increased by 5.8% from the previous term. The sales and operating income did not reach planned goals. 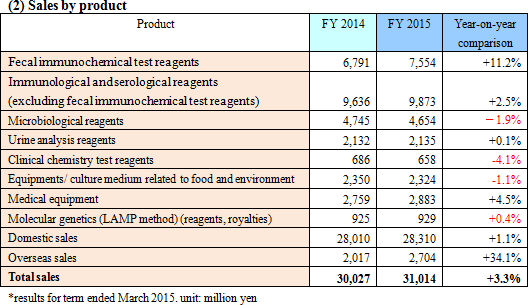 Domestically, the company focused on promotion of colorectal cancer screening and diagnosis. The company also rolled out a new medical device product, "the OC-sensor PLEDIA." Performance in North America, Europe and Asia were all good. The sales of "AIA related reagents" procured from Tosoh Corp. decreased, but the increase in testing for the evaluation of the health levels of the stomach (ABC classification) (helicobacter pylori test reagents and pepsinogen test reagents) covered this decrease. - Microbiological reagents
The sales of drug sensitivity test reagents, powder culture medium, and rapid test reagents were all below those for the previous term due to the negative rebound from the last minute demand prior to the consumption tax hike.
- Urinalysis test strips
The income from "Uropaper III Eiken," used for visual observation method, decreased. However, due to the trends toward automated testing, the sales of "UROPAPER α III EIKEN," dedicated test strip for fully automated urine analyzer, were strong both domestically and overseas. As a result, sales of urine analysis reagents remain flat. The company also released the new analyzer "US-3500."
- Clinical chemistry test reagents
The year-to-year sales decreased due to the drop in price from market share competition.
- Equipment/ Culture medium related to food and environment
The year-to-year sales decreased due to the rebound from the last minute demand prior to the consumption tax hike, and drop in price due to competition.
- Medical equipment
Domestically, the sales of immunological and serological reagents, microbiological test apparatus increased, while fecal immunochemical test analyzer and urine analyzer increased overseas.
- Molecular genetics (LAMP)
Genetic testing reagents and running royalties increased.
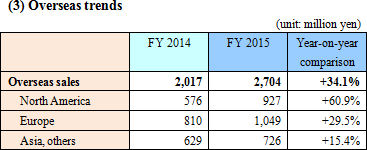 *North America
With positive impact from Obamacare, increase in the number of tests lead to significant increase in sales of fecal immunochemical test reagents and apparatus.
*Europe
Not only was the company's fecal immunochemical test used for the national colon cancer screening and diagnosis in France, but the company was also able to bid successfully for the mass testing in Madrid, Spain. The company also increased activity for the test to be adopted in England. In Italy, fully automated urine analyzer as well as its dedicated test strip grew in sales significantly.
*Asia, etc
The company also worked for the fecal immunochemical test to be newly adopted and its sales increase in Thailand, Hong Kong, Australia, New Zealand, etc.In China, the company worked on the sales promotion of LAMP products, the fecal immunochemical test reagent and apparatus. The company also acquired the permission for the LAMP tuberculosis tests, and started promoting sales. *FIND business
The advancement in the business related to the alliance with FIND was as follows:(i) Tuberculosis The company continued working to acquire WHO recommendation by conducting evaluation in developing nations, followed by its data processing and analysis. The company is hopeful to obtain this endorsement in the first half of this term. The company is currently preparing the sales structure, and is negotiating with the agent with a global sales force. (ii) Malaria The company sold screening reagents for the malaria eradication project. (iii) African trypanosomiasis The clinical studies in the Democratic Republic of Congo and Uganda were continued. Reagents for the eradication project were sold in Uganda, Malawi, Guinea, the Democratic Republic of Congo, etc. (iv) Leishmaniasis, Chagas disease The evaluation of prototype by FIND was continued. 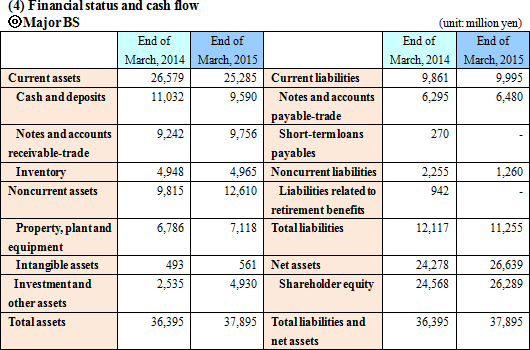 Current liabilities augmented slightly due to the increase in notes and accounts payable-trade, whereas the noncurrent liabilities decreased by 995 million yen due to the decrease in liabilities related to retirement benefits. Net assets increased by 2,361 million yen due to the increase in the retained earnings. As a result, the equity ratio increased by 3.6%, from 66.2% of the previous term end to 69.8%. 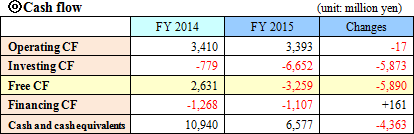 |
| Fiscal Year March 2016 Earnings Estimates |
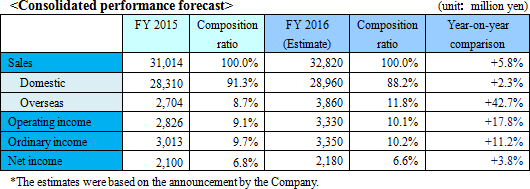 Overseas market development leads to increases in sales and profits
The sales forecast shows a 5.8% increase from the previous term to 32,800 million yen. Overseas sales, especially in Europe, will significantly increase.Travel and packaging expenses will increase due to the augmentation in the sales promotion activities, and SG&A expenses including IT investments and others required for enhancing efficiency will also increase. However, all these will be offset by an increase in the sales. The operating income is expected to grow by 17.8%, to 3,300 million yen. The growth of productivity and the number of high profit margin products will result in increased operating margin by 1%. Dividend is expected to increase to 40 yen per share, by 3 yen per share for interim and 2 yen per share for year-end, for a total increase of 5 yen per share. This will be the first increased dividend within four terms, and the dividend payout ratio is expected to be 33.5%. <Domestic>
The company will attempt to increase the sales of fecal immunochemical test reagents and apparatus, test reagents for general purpose automated analysis apparatus, "LZ test Eiken series," microbiological reagents (drug sensitivity test reagents, industrial field), Tosoh related test reagents, LAMP products, and point-of-care testing (POCT) as one of the sales promotion product group.
<Overseas>
The company will try to increase the share and advance into new market. Overseas sales ratio will increase to 11.8%, topping 10% for the first time.Europe will grow significantly since the national screenings beginning in France will be fully contributed, and England is expected to hold its first bidding in the latter half of the term. The company will establish the brand and boost the sales of the fecal immunochemical test reagents, and target national screenings like the one in France in the previous term. The company will promote the urinalysis test strips and "LZ test Eiken" series in China. The company will also develop emerging markets such as ASEAN and India. For the FIND business, the company also expects recommendation from the WHO for products related to tuberculosis, and will work on malaria related products. The company expects sales of 200 million yen this term. <Research and development>
The following points will be enhanced.
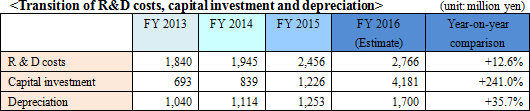 Local production and sales will be promoted for the China plant. New construction of plants outside China is an issue to be investigated in the future. <Improvement in productivity>
The improvement in production capability and the reduction of production cost will be attempted through the renewal of plants and production facilities. The decrease in the SG&A expenses will also be attempted through improvement to the sales-cost ratio and higher operation efficiency.
|
| Interview with President Morifumi Wada |
|
<The first year in retrospect, since Mr. Wada's appointment as President>
 <Development of overseas market>
<Towards a vibrant company>
<A message to our investors>
|
| Conclusions |
|
|
| Reference: New Management Framework "EIKEN WAY" and "EIKEN ROAD MAP 2009" |
|
EIKEN CHEMICAL set a goal for 2018, the company's 80th anniversary year, and established "EIKEN WAY" and "EIKEN ROAD MAP 2009" in March 2009, as a fundamental policy to promote "winning management."
Furthermore, securing safety and compliance with laws and ordinances are becoming increasingly important management challenges. As a result, the gap between corporations is expected to be larger. Under these circumstances, in order to achieve steady growth and sustainable enhancement of corporate values, EIKEN CHEMICAL recognizes that it is essential to clarify the goal of EIKEN group, optimize the efficiency of management resources, and implement the strategies to utilize environmental changes with new visions more quickly and boldly. Based on this recognition, it established the "EIKEN WAY" as a plan to practice solid management and the "EIKEN ROADMAP 2009" as basic principles to promote "winning management" with long term goals. 1. Business domain
EIKEN CHEMICAL set the "clinical diagnostics business" and "food and environment testing business" among healthcare businesses as their main business domains for which they can utilize the technologies and strengths owned by the EIKEN Group to achieve steady growth and increase in profitability. Furthermore, in these domains, they are aiming at creating new businesses for future growth.
2. EIKEN ROAD MAP 2009 Grand Vision
We will transform EIKEN into a global corporation by 2018 that can leverage expertise as a medical testing pioneer to protect the health of the public.
3. EIKEN ROAD MAP 2009 Principles of Action
 4. Fundamental policy
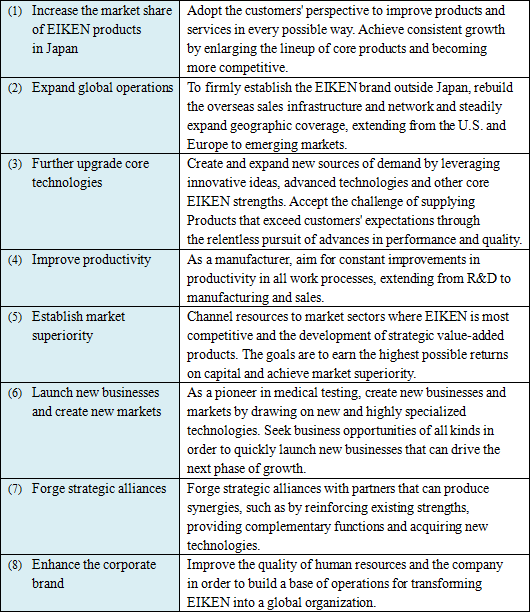 5. Management goal
To become one of the global medical testing corporations in the future, we will strive to steadily enhance profitability while creating solid business foundation. Our goal for the time being is to maintain over 10% of overseas sales ratio and over 10% of consolidated sales profit margin.
Disclaimer
This report is intended solely for information purposes, and is not intended as a solicitation to invest in the shares of this company. The information and opinions contained within this report are based on data made publicly available by the Company, and comes from sources that we judge to be reliable. However we cannot guarantee the accuracy or completeness of the data. This report is not a guarantee of the accuracy, completeness or validity of said information and or opinions, nor do we bear any responsibility for the same. All rights pertaining to this report belong to Investment Bridge Co., Ltd., which may change the contents thereof at any time without prior notice. All investment decisions are the responsibility of the individual and should be made only after proper consideration.Copyright(C) 2015 Investment Bridge Co., Ltd. All Rights Reserved. |




