Bridge Report:(4598)Delta-Fly Pharma the First Half of the Fiscal Year March 2020
 President Kiyoshi Eshima | Delta-Fly Pharma, Inc.(4598) |
 |
Corporate Information
Exchange | TSE Mothers |
Industry | Pharmaceutical products (manufacturing industry) |
President | Kiyoshi Eshima |
Address | 37-5 Nishikino, Miyajima, Kawauchi-cho,Tokushima-shi, Tokushima |
Year-end | End of March |
URL |
Stock Information
Share Price | Shares Outstanding | Total Market Cap | ROE(Actual) | Trading Unit | |
¥1,783 | 4,504,600 shares | ¥8,031million | -31.0% | 100 shares | |
DPS(Estimate) | Dividend Yield (Estimate) | EPS(Estimate) | PER(Estimate) | BPS(Actual) | PBR(Actual) |
¥0.00 | - | ¥-244.85 | - | ¥801.93 | 2.2 times |
*Share price is as of closing on December 3. The number of shares outstanding, DPS, and EPS were taken from the brief financial report for the second quarter of the term ending March 2020. ROE and BPS are the value as of the end of the previous term.
Earnings Trends
Fiscal Year | Net Sales | Operating Income | Ordinary Income | Net Income | EPS | DPS |
Mar. 2016 (Actual) | 145 | -584 | -595 | -597 | -185.53 | 0.00 |
Mar. 2017 (Actual) | 902 | 328 | 323 | 305 | 88.31 | 0.00 |
Mar. 2018 (Actual) | 150 | -243 | -244 | -246 | -71.20 | 0.00 |
Mar. 2019 (Actual) | - | -592 | -671 | -673 | -170.16 | 0.00 |
Mar. 2020 (Estimate) | - | -1,066 | -1,066 | -1,069 | -244.85 | 0.00 |
*Unit: million yen, yen
*The estimated values were provided by the company. 500-for-1 share split was conducted on Jun. 25, 2018. EPS was adjusted retroactively.
This report introduces earnings trends, progress of the development etc. of Delta-Fly Pharma, Inc.
Table of Contents
Key Points
1. Company Overview
2. Earnings Trends
3. Growth Strategy
4. Conclusions
<Reference: Regarding Corporate Governance>
Key Points
- The company develops anticancer drugs under the original concept of “module drug development,” which means that they develop new anticancer drugs that have an improved balance between clinical efficacy and safety and whose side effects are weak, by using the existing active substances with anticancer property as “modules (components)” and designing dosage and administration, combination methods, etc. with ingenuity.
- “Module drug development” has benefits for patients, including the improvement of treatment effects, the elimination of side effects, and cost reduction, and also benefits for development, including the high exclusiveness in patenting, the swiftness of development, and low development risk. The company currently has 6 drug pipelines, and 3 candidate drugs are under clinical tests, and for the other 3 candidate drugs, clinical tests are being prepared.
- In addition to module drug development, the company is characterized by the specialization in development of anticancer drugs, the development by experienced members, and efficient business operation utilizing external resources.
- The business revenue for the second quarter of the term ending March 2020 was not posted. Along with the progress of development of drug pipelines, the company made considerations and preparations for new clinical tests. R&D expenses were 598 million yen, up 307.5% year-on-year, and business expenses were 725 million yen, up 205.9% year-on-year. Operating loss was 725 million yen, up 488 million yen year-on-year.
- The earnings forecast for the term ending March 2020 is unchanged. As for business revenue, considering the uncertainties over the progress of clinical tests and license negotiations, they plan to clarify the outlook when revenue is secured. As for business expenses, since the clinical phase III test of DFP-10917 will be carried out in the U.S. in parallel with the development of drug pipelines, expenses for clinical tests will be more than those in the previous term, thus R&D expenses are predicted to be 822 million yen, up 445 million yen year-on-year. As a result, operating loss will be 1,066 million yen, up 474 million yen year-on-year.
- DFP-10917 development is making the most progress and its clinical phase III test started in the U.S. The clinical Phase III tests were updated in the U.S. Public organizations’ clinical tests registration website and patients’ registration started in November 2019. We will continue to pay attention to their progress. Further, we would like to expect the news release regarding licensing-out to pharmaceutical companies.
1. Company Overview
Delta-Fly Pharma upholds the corporate ethos: “To provide treatment methods recommendable to cancer patients among relatives with peace of mind, by diagnosing all states of cancer patients rather than seeing only cancer,” and develops anticancer drugs under the original concept of “module drug development,” which means that they develop new anticancer drugs that have an improved balance between clinical efficacy and safety and whose side effects are weak, by using the existing active substances with anticancer property as “modules (components)” and designing dosage and administration, combination methods, etc. with ingenuity.
1-1 Corporate history
The President Eshima, who was born in Tokushima Prefecture, graduated from Nagoya Institute of Technology, completed the master’s course of Tokyo Institute of Technology, and joined the Otsuka Group, a pharmaceutical company in Tokushima Prefecture, which is his hometown. Then, he was assigned to TAIHO Pharmaceutical Co., Ltd., which is a business company of the Otsuka Group.
Immediately after joining the company, he was dispatched to Faculty of Science and Engineering, Waseda University, and engaged in the development of pharmaceuticals, especially new medicines composed of functional polymers, as a researcher for about 12 years. When he was in the section that seeks seeds of pharmaceutical products in TAIHO Pharmaceutical, he saw how the business administration of U.S. bio ventures was carried out. That stirred his willingness to become independent, manage a pharmaceutical company by himself, and create medicines with a new approach, rather than engaging in development in the R&D section of a leading pharmaceutical company. He also aimed to develop a business while not only creating medicines, but also considering what he can do for patients in front of him. In 2010, when he was 61 years old, he resigned from TAIHO Pharmaceutical, and established Delta-Fly Pharma. The company is committed to the development of anticancer drugs whose side effects are weak and friendly to patients through module drug development. As of September 2019, the company has 6 drug pipelines.
It was listed in Mothers of Tokyo Stock Exchange in October 2018.
1-2 Corporate ethos and management philosophy
The corporate name “Delta-Fly” is derived from a “dragonfly.” Since dragonflies only go forward, and do not go backward, they represent the unflagging spirit, and they are also called “winning insects.” Namely, the corporate name implies the firm resolve to develop pharmaceutical products.
Corporate ethos | To provide treatment methods recommendable to cancer patients among relatives with peace of mind, by diagnosing all states of cancer patients rather than seeing only cancer |
As mentioned later, the company considers that its social mission is not to develop anticancer drugs only for eradicating “cancer,” but to provide anticancer treatment with reasonable price while curbing side effects, which are serious issues with anticancer drugs, so that patients and their family members can use it without worry.
1-3 Environment surrounding the company
According to “the demographic statistics in Japan in 2018” published by the Ministry of Health, Labour and Welfare, the mortality rate (number of deaths per 100,000 people) of malignant neoplasm (cancer) was the highest: 298.3 in 2016. It has been the highest for over 30 years since it replaced cerebrovascular disease, whose mortality rate was 134.3 while that of malignant neoplasm was 142.0, in 1981. It is increasing year by year.
It is said that the incidence of cancer is growing due to the aging of the population, the change in lifestyles, including dietary habits, etc.
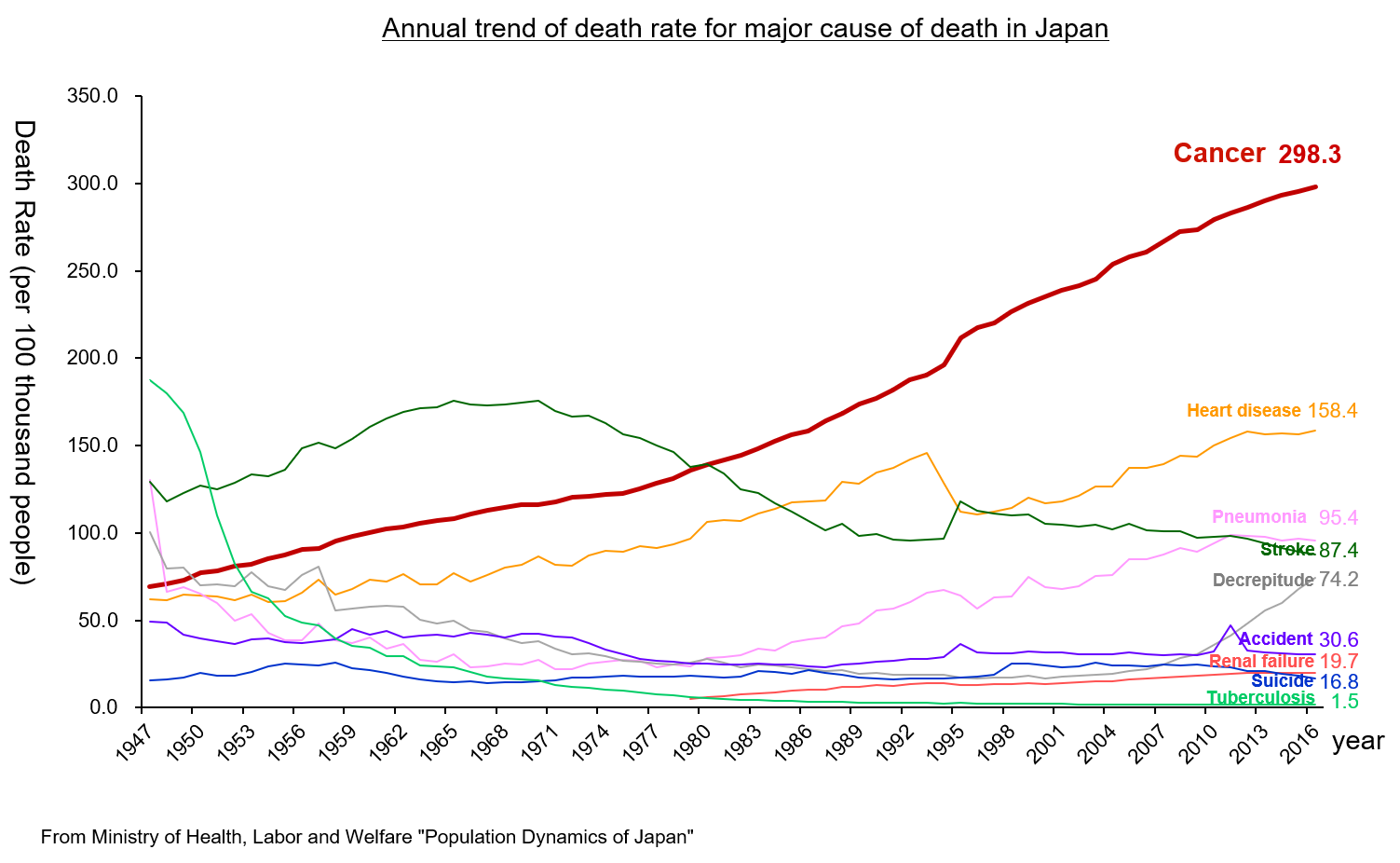
(Taken from the reference material of the company)
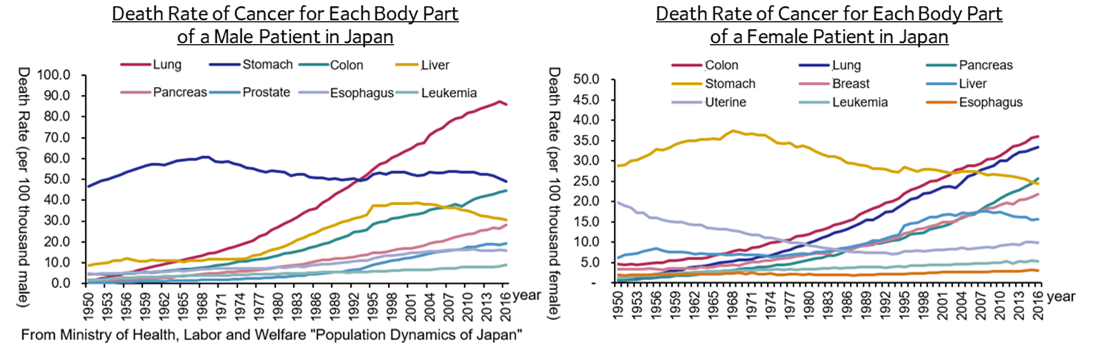
(Taken from the reference material of the company)
In these circumstances, various anticancer drugs are used, and new medicines are being developed. But, as publicly known, the side effects of anticancer treatment are significant, so there are considerable needs for the reduction of side effects from the viewpoint of improving the quality of life (QOL) of patients.
(Mechanism of side effects)
Since cancer cells rapidly divide and proliferate, anticancer drugs are designed to kill rapidly growing cancer cells. However, anticancer drugs affect not only cancer cells, but also the normal cells that rapidly divide, such as blood cells produced in bone marrow, the cells of digestive organs, the cells of genitals, and hair root cells, causing side effects, such as nausea, vomiting, hair loss, and fatigue.
1-4 Business contents
1-4-1 Delta-Fly Pharma’s method for creating medicines: Module drug development
What distinguishes the company most among a lot of bio ventures is its concept for developing medicines: “module drug development.”
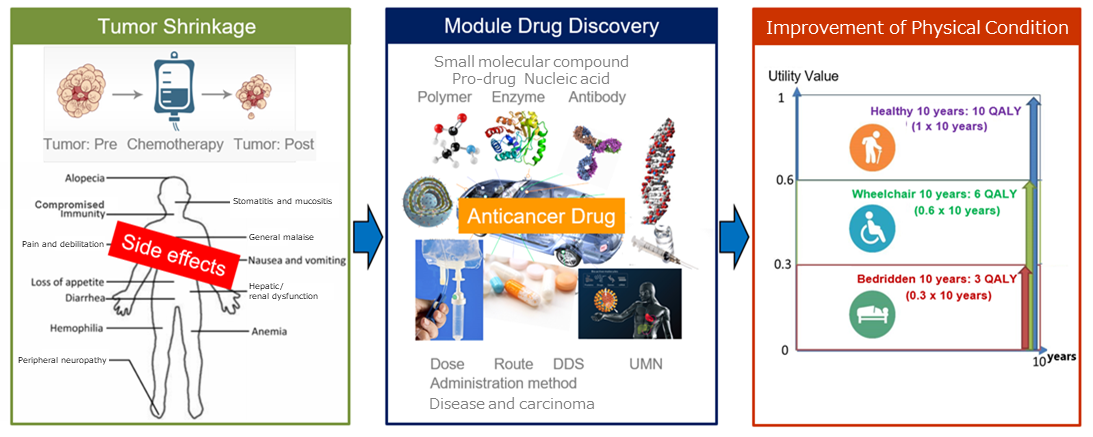
(Taken from the reference material of the company)
“Module drug development” means the development of new anticancer drugs that have an improved balance between clinical efficacy and safety by using the existing active substances with anticancer property as “modules (components)” and designing dosage and administration, combination methods, etc. with ingenuity.
Through “module drug development,” the company focuses on not only “cancer circumstances” but also the whole conditions of “cancer patients,” improves the anticancer drugs whose effects are limited and which have various side effects in a multifaceted manner, and produces medicines whose side effects are so weak that you can recommend them to your relatives suffering from cancer.
(Advantage of module drug development)
Merits for patients | ・Since medicines are created based on data on patients, treatment effects are expected to improve.・Since medicines are created based on data on patients, conventional side effects are expected to disappear.・The number of fundamental and clinical tests is small and their periods are short; accordingly, their costs are not considerable. |
Merits for development | ・Since medicines can be patented due to novelty and inventive steps, they will have high exclusivity.・Since medicines are developed based on data on patients, development speed is high.・Since medicines are developed based on data on patients, development risk is low. |
In general creation of anticancer drugs, chemicals that act on the cancer-specific parts are extracted at the stage of fundamental search and research, and possible chemicals become candidates for anticancer drugs. However, it is necessary to check their functions at the clinical stage and demonstrate efficacy and safety through clinical tests. Accordingly, the R&D period from the basic stage is long.
Meanwhile, “module drug development” does not require fundamental search or research so much, because the active substances of already used anticancer drugs are combined, and it is possible to predict efficacy and safety at the clinical stage. Accordingly, it is possible to start clinical tests in one to two years after the start of medicine development. Like this, compared with general development of anticancer drugs, the R&D is more efficient, the development period is shorter, and the risk of development, including the failure in clinical tests, is lower.
In addition, when focusing on the issues with the treatment of cancer patients, the combination of off-patent pharmaceutical products by utilizing the knowledge and know-how of anticancer drugs enables them to be patented as new anticancer drugs.
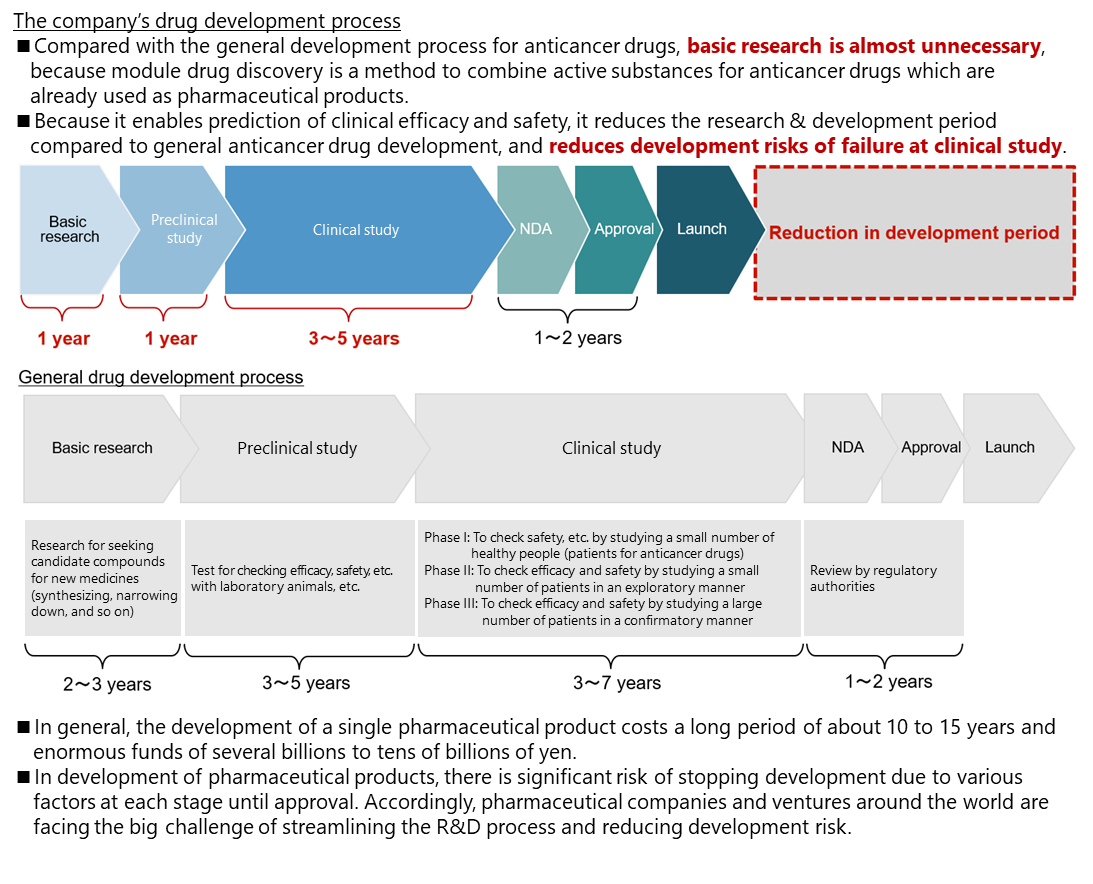
(Taken from the reference material of the company)
Nowadays, an increasing number of pharmaceutical companies engage in drug-repositioning activities to discover new effects of generic and existing medicines, for the purpose of reducing the cost for new drug development.
These are the same as “module drug development” in that existing medicines are used, but it is difficult to patent these drugs based on generic medicines and drug repositioning because of the lack of novelty and inventiveness. On the other hand, “module drug development” will make all developed drugs patented. This is a defining difference.
As long as they try to solve the problems with anticancer drugs, they can create totally new anticancer drugs. Therefore, the company is certain that “module drug development” will bring significant innovation to methods for creating medicines.
1-4-2 Business and revenue models
(Business model: to develop an efficient R&D system)
Before a new pharmaceutical product is released, it is common that “fundamental research” is first conducted, “preclinical tests (tests for checking the pharmacological actions, in-vivo kinetics, harmful effects, etc. by using animals)” and “clinical tests (scientific tests for studying the effects of pharmaceutical products, treatment technologies, etc. on human bodies)” are carried out, applications are submitted to authorities to obtain approvals, products are manufactured, and then surveys are conducted after manufacturing, marketing, and sale.
In these processes, Delta-Fly Pharma concentrates on the management of R&D, while outsourcing meticulous tasks to excellent external R&D companies and manufacturers inside and outside Japan. The company has actualized an efficient R&D system in cooperation with external cooperative institutions according to development phases. It also engages in the R&D for new anticancer drugs by using a drug delivery system in collaboration with Sanyo Chemical Industries, Ltd. (1st section of TSE; 4471).
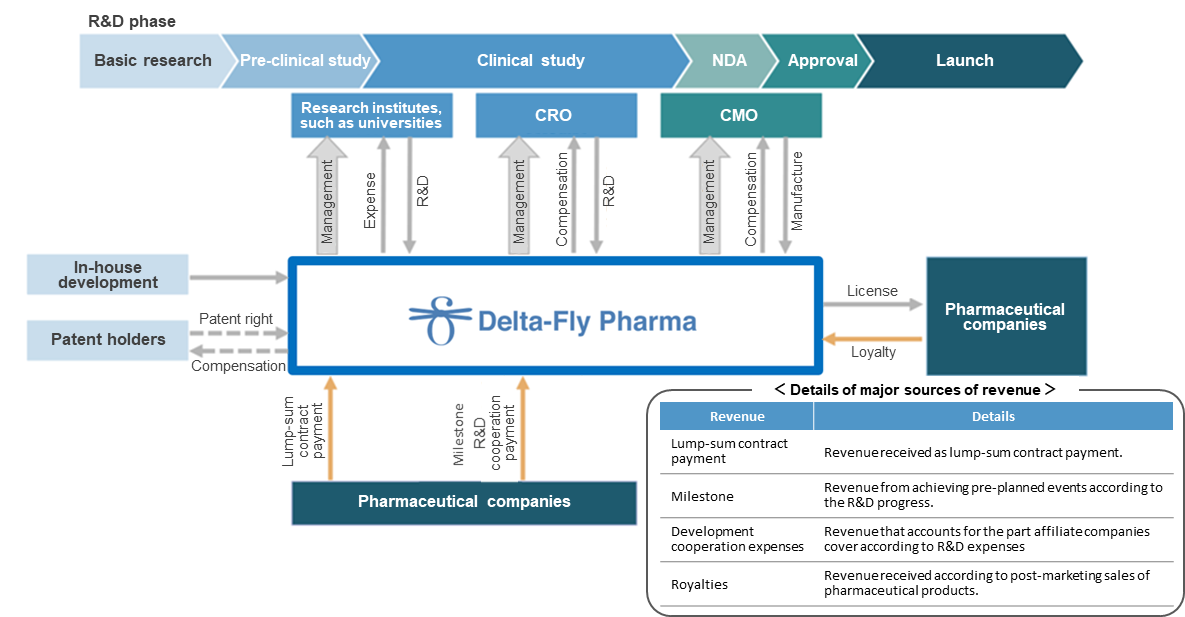
(Taken from the reference material of the company)
(Revenue model)
At the R&D stage, the main revenue sources are “lump-sum contract payment” for contracts with affiliated pharmaceutical companies, “milestone,” and “cooperation funds for development.” If collaborative products are released, the company will receive royalties according to sales.
Currently, Delta-Fly Pharma collaborates with the following two pharmaceutical companies.
Kyowa Chemical Industry Co., Ltd. (unlisted) | Signed a contract for an exclusive license for the candidate compound for anticancer drugs DFP-14323 in Japan. |
Nippon Shinyaku Co., Ltd. (1st section of TSE, 4516) | Signed a contract for an exclusive license for the candidate compound for anticancer drugs DFP-10917 in Japan. |
1-4-3 Drug pipelines
As of now, Delta-Fly Pharma has the following 6 drug pipelines in accordance with the above mentioned management policy.
The progress, current situation, and future plan for the development and commercialization of each pipeline are as shown below. Three candidate drugs are undergoing clinical tests. For the remaining three candidate drugs, clinical tests are being prepared.
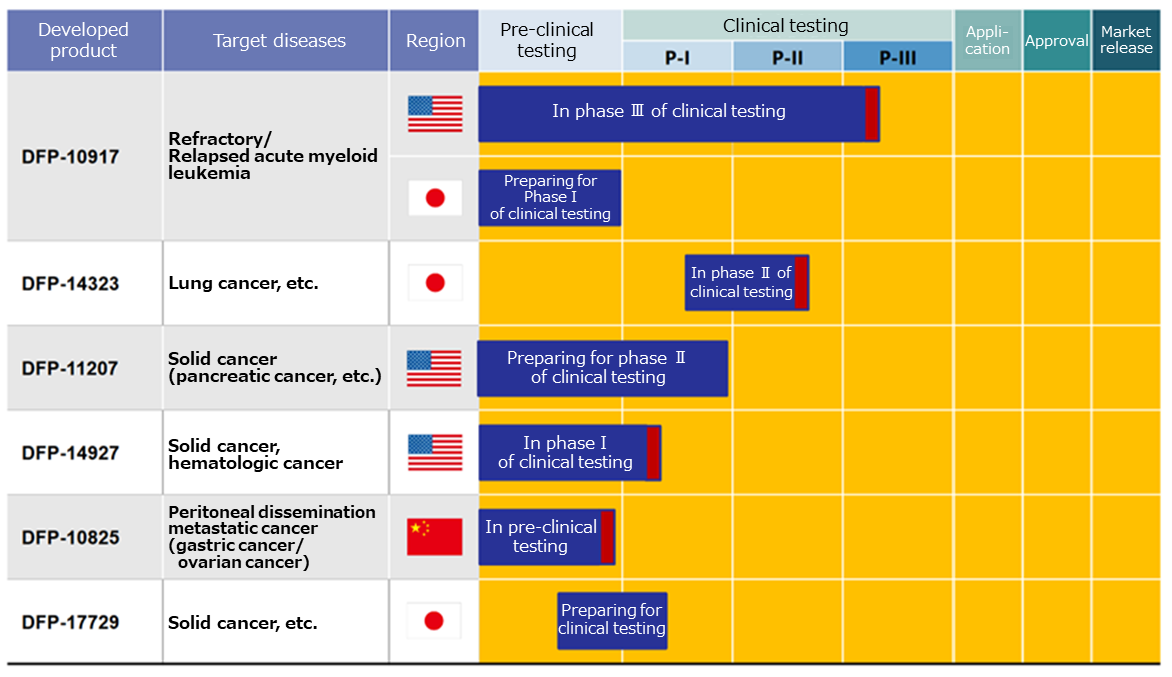
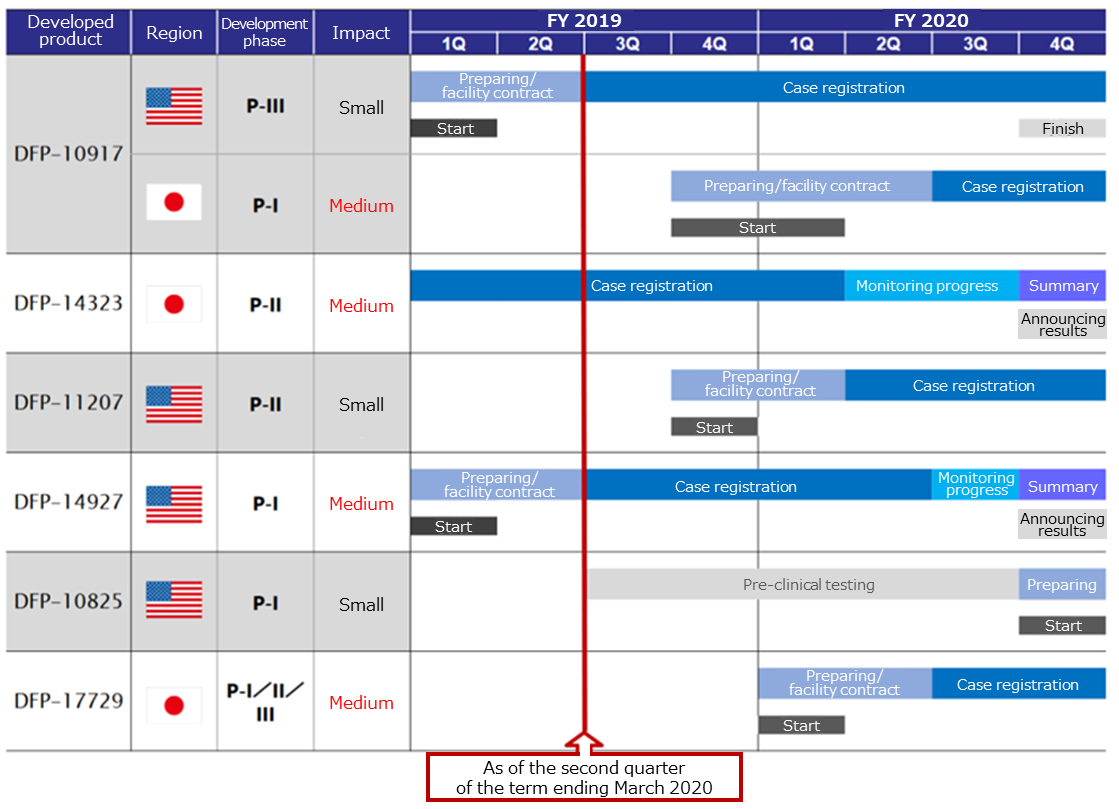
The events regarding to each pipeline, which are supposed to be held in this term or the next, are as follows.
(Taken from the reference material of the company)
1) 「DFP-10917」
Item | Outline |
Main target disease | Refractory and recurrent acute myeloid leukemia (Standard treatment methods have been established. About 70% of patients go into remission temporarily, as the cancer cells disappear from blood, but recurrence rate is high, and only 30% of patients can recover fully.) |
Characteristics of existing medicines, etc. | The existing medicine CNDAC is targeted at solid tumors. Dosage is high, and administration is conducted intravenously or orally in a short period of time. The efficacy against solid tumors is limited, and serious side effects were observed in some cases. |
Improved points and effects of modules | The dosage was reduced, and administration was conducted intravenously and continuously for a long period of time. As a result, there emerged different effects from those of conventionally used nucleic-acid derivatives (such as cytarabine and gemcitabine). It can be expected that the drug will be effective for the patients of refractory and recurrent acute myeloid leukemia, which cannot be treated with existing chemotherapy. |
Countries where patents were acquired (as of the end of Oct. 2019) | Japan, the U.S., EU, China, Australia, South Korea, and Russia |
(State of development, and future commercialization)
In the clinical phase I/II tests carried out in the U.S., the drug was effective for 48% (14/29) of patients in the phase II, indicating high effectiveness. Seeing this result, the company had a meeting with the U.S. Food and Drug Administration (FDA) after the clinical phase II test, and submitted a plan for the clinical phase III test. Consent was obtained, however, as the treatment system of refractory and recurrent acute myeloid leukemia was changed, part of the protocol for Phase III clinical testing was changed before re-submitting it to the US FDA. Phase III of clinical testing started. A startup meeting was held, and the screening of research subjects started.
The company aims to complete the phase III test by fiscal 2021, and obtain approval and sell it in the U.S. by fiscal 2022. As for Japan, the licensee, Nippon Shinyaku Co., is preparing for phase I of clinical testing. Also, the company received advice from Pharmaceuticals and Medical Devices Agency face-to-face.
2) 「DFP-14323」
Item | Outline |
Main target disease | Terminal stage lung cancer, etc. |
Characteristics of existing medicines, etc. | The existing medicine “bestatin (Ubenimex)” is targeted at blood cancer. The dosage is high, and administration is conducted intravenously or orally with a single agent. It is indicated that the drug is for blood cancer only, but it showed a survival advantage against lung cancer. |
Improved points and effects of modules | For the purpose of enhancing the antitumor effect, the dosage was reduced, and the drug was used together with a molecular target drug. As a result, the efficacy against lung cancer was confirmed. The drug is expected to improve the immune function of cancer patients, and treat terminal or elderly patients of solid tumors. |
Countries where patents were acquired (as of the end of Oct. 2019) | Japan, the U.S., Australia, Russia, Korea, China |
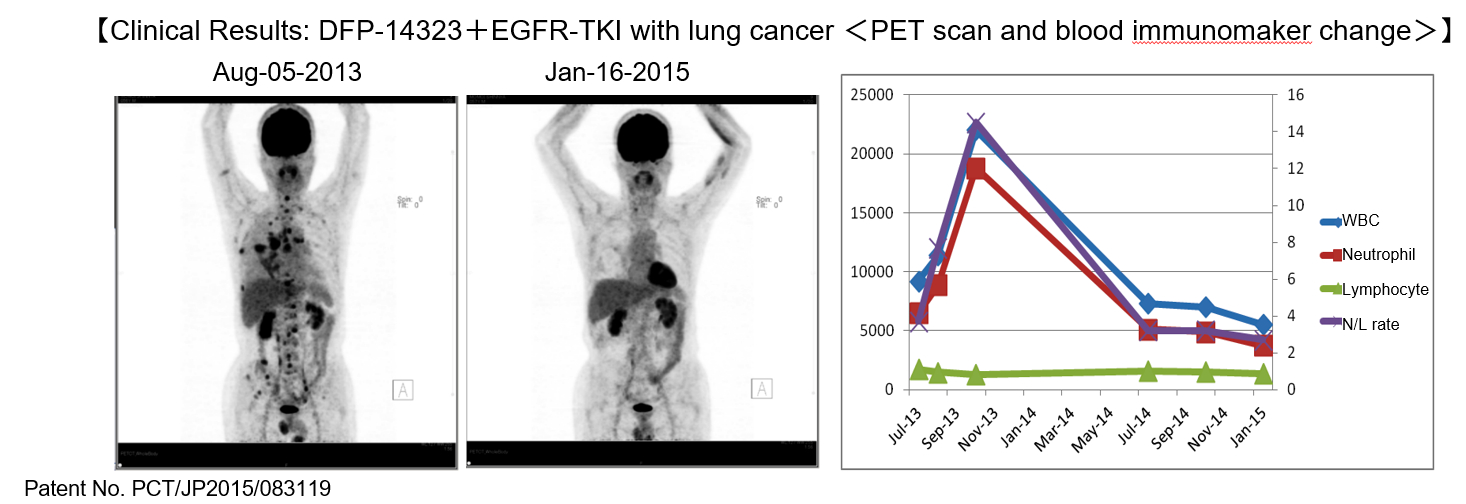
(Taken from the reference material of the company)
(State of development, and future commercialization)
As for the existing medicine Ubenimex, Nippon Kayaku Co., Ltd. obtained the approval for its efficacy and effect of “prolonging the survival period of adults suffering from acute non-lymphatic leukemia when combined with maintenance and intensive chemotherapeutic agents after remission” in Japan.
Delta-Fly started the clinical phase II test for the combined treatment of low-dose EGFR-TKI targeted at patients of EGFR gene mutation-positive non-small cell lung cancer in January 2018 in Japan、and since the facilities that conduct clinical trials have increased in Japan, the company proceeded with registering new cases.
The company aims to obtain approval for the addition of supplemental indication and sell it in Japan by fiscal 2023.
The company has concluded a contract for an exclusive license in Japan with Kyowa Chemical Industry Co., Ltd. (unlisted)
3) 「DFP-11207」
Item | Outline |
Main target disease | Solid tumors (such as pancreatic cancer) |
Characteristics of existing medicines, etc. | The existing medicine TS-1 has hematotoxicity, including the reduction of blood platelets, and it is difficult to continue treatment sufficiently. |
Improved points and effects of modules | DFP-11207 is a compound developed by combining three modularized active substances (modules I, II, and III) for sustained release, inhibition, and deactivation, in order to control the pharmacokinetics of 5-fluorouracil (5-FU), which has anticancer effects. It avoids hematotoxicity, including the decrease of blood platelets, which is caused by conventional 5-FU anticancer drugs, improves the balance between efficacy and safety, and enables long-time continuous treatment. This is a representative case of module drug development, in which the combination of compounds was improved. |
Countries where patents were acquired (as of the end of Oct. 2019) | Japan, the U.S., EU, China, Australia, Korea, Russia, Republic of China, Hong Kong |
(State of development, and future commercialization)
In the U.S., the company proceeded with the clinical phase I test for solid tumors (digestive system cancer), and checked the recommended dose at the next test and confirmed that the decrease of blood platelets does not occur as a side effect, which has been caused by conventional 5-FU anticancer drugs.
Testing the effects of food has finished, and the company summarized the process, held a discussion with clinical investigators, and formulated the plan for phase II of clinical testing with the combined use of anticancer drugs.
The company announced the results of phase I of clinical testing and the study of food effects at the conferences of the Chinese Society of Clinical Oncology (CSCO) and Japan Society of Clinical Oncology (JSCO).
It aims to obtain approval and sell it in the U.S. or in China by fiscal 2024.
4) 「DFP-14927」
Item | Outline |
Main target disease | Pancreatic cancer, gastric cancer, and myelodysplastic syndromes |
Characteristics of existing medicines, etc. | The existing medicine DFP-10917 needs to be administered for 14 days in a row, by using a pouch for continuous intravenous injection, and it was necessary to improve its convenience. The target disease has been only blood cancer. |
Improved points and effects of modules | DFP-14927, a polyethylene glycol-conjugated candidate anticancer substance, is a polymeric delivery of DFP-10917. It selectively clusters around cancer tissue, and discharges DFP-10917 effectively inside cancer cells. The frequency of administration was reduced to once per week, and intravenous drip infusion was adopted. As a result, the medicine now can be used against solid tumors and myelodysplastic syndrome as well as blood cancer. |
Countries where patents were acquired (as of the end of Oct. 2019) | Japan, the U.S., China, Australia, Russia |
(State of development, and future commercialization)
The preclinical test has been finished in the U.S. The data of the preclinical test indicate that the level of the medicine in blood is stable for a long period of time when it is administered once a week, and that there is the antitumor effect against solid tumors.
In March 2018, the company concluded a contract for collaborative development with Sanyo Chemical Industries, Ltd. and prepared for the application for the start of the clinical phase I test, and on January 18, 2019, the U.S. FDA completed the examination of the safety of Investigational New Drug (IND), and approved the clinical phase I test in the U.S. And the company started clinical phase I testing aimed at patients with digestive system cancer including pancreatic cancer and gastric cancer.
In addition to the clinical test, the company plans to discuss the possibility of administration to patients of blood cancer, including myelodysplastic syndrome.
They aim to obtain approval and start sales in the U.S. by FY 2025.
5) 「DFP-10825」
Item | Outline |
Main target disease | Gastric cancer, ovarian cancer, and peritoneal metastasis from pancreatic cancer |
Characteristics of existing medicines, etc. | Although the basic drug siRNA has a definite inhibitory effect as its basic effect, its clinical effect in systemic administration has been poor. |
Improved points and effects of modules | Nucleic acid drugs using RNA interference are expected to be the next cancer treatment drugs next to molecular-targeted cancer drugs and cancer immunotherapeutic drugs. The nucleic acid drug DFP-10825 is designed to be effective by intraperitoneal rather than systemic administration, as it specifically inhibits the factors that significantly affect cancer growth by RNA interference. In patients with ovarian cancer or stomach cancer, fluid retention such as pleural fluid and ascites (peritoneal metastasis) is observed at the terminal stage, but ascites is controlled by injecting the drug directly into the abdominal cavity to exert an effect. It is expected to relieve the pain and lead to the patients’ prolonging life. |
Countries where patents were acquired (as of the end of Oct. 2019) | Japan, the U.S., EU, China, Korea, Russia, Republic of China, Hong Kong |
(State of development, and future commercialization)
The company has already completed efficacy and pharmacokinetics tests against peritoneal metastasis that causes ascites associated with ovarian, stomach or pancreatic cancer. Preliminary investigations based on the current Good Manufacturing Practice (cGMP) standards have also been completed for the manufacture of study drugs, such as drug substances, DDS and preparations. From now on, after adding preclinical tests according to the Good Laboratory Practice (GLP) standards for conducting non-clinical tests concerning safety of drugs using a part of the funds obtained from the stock listing, the company is planning to apply for IND to the US FDA and will begin the clinical phase I test for patients with peritoneal metastasis of ovarian, stomach or pancreatic cancer in the U.S. It has also received each country’s certificates of patent that is pending.
While preparing active pharmaceutical ingredients and test drugs, they will conduct pre-clinical testing and aim to start clinical testing in Japan or the U.S. by FY 2020.
6) 「DFP-17729」
Item | Outline |
Main target disease | Terminal stage pancreatic cancer, malignant gastric lymphoma, gastric cancer, and lung cancer. |
Characteristics of existing medicines, etc. | Urinary alkalinizing agents, which are existing drugs, are targeted for hyperuricemia and others, but it has been confirmed that they provide a life-prolonging effect in pancreatic cancer and have an antitumor effect on each cancer tumor. |
Improved points and effects of modules | Normal cells are more alkaline outside the cells than inside the cells, but cancer cells are more acidic outside the cells. This is because the growth of cancer cells promotes glycolysis, producing lactic acid and hydrogen ions, and they are actively released into the extracellular space. DFP-17729 suppresses the growth of cancer by alkalizing the outside of cancer cells. It has been confirmed in animal experiments that the combined use of an anticancer drug and an immune checkpoint inhibitor enhances the effect as compared with the monotherapy with an immune checkpoint inhibitor. |
Countries where patents were acquired (as of the end of Oct. 2019) | Japan, Korea |
(State of development, and future commercialization)
The company is preparing for the additional indication of urine alkalizing agents, which are approved and sold as pharmaceutical products, as anti-cancer drugs in Japan.
Because urine alkalizing agents are already being used in clinical practice for the efficacy and effect of “acidosis improvement” to treat “hyperuricemia” and “tumor lysis syndrome,” preclinical testing is not necessary.
The company aims to expand the range of anti-tumor effects of existing drugs through combined use of anti-cancer agents and immune checkpoint inhibitors and provide new cancer treatments.
They aim to start clinical testing in Japan by FY 2020.
1-5 Four characteristics as a bio-venture
The company as a bio-venture has the following four main characteristics.
1) Module drug development
As described above, the company is patenting existing drugs, etc. by re-inventing them with ingenuity based on “modules” (components) and creating new drugs with improved balance between clinical efficacy and safety.
2) Specialized in the development of anti-cancer drugs
By working specifically on “anti-cancer drugs,” which still have limited effectiveness and cause various side effects, the company is accelerating the development of new drugs through module drug development and contributing to the improvement of the social life of cancer patients.
3) Development by experienced members
The development members consisting of people who have been engaged in research and development of anti-cancer drugs for many years at pharmaceutical companies and clinicians who are familiar with cancer patients advance the development of drugs with certainty and meet unmet medical needs. This sharply differentiates the company from others, giving competitive advantage.
4) Effective utilization of external resources
The company operates efficiently by focusing on management and operation of research and development without having factories or research institutes and proactively cooperating with external contractors and other organizations for outsourcing tasks.
2. Earnings Trends
2-1 First Half of the Fiscal Year ending March 2020 Earnings Results
1) Earnings trends
| 1H of FY 3/19 | 1H of FY 3/20 | YoY |
Operating Revenue | - | - | 0 |
Operating Cost | 237 | 725 | +488 |
R&D Expense (included in the Operating Cost) | 146 | 598 | +451 |
Operating Income | -237 | -725 | -488 |
Ordinary Income | -277 | -729 | -452 |
Net Income | -278 | -731 | -452 |
Unit: Million yen
(Operating Revenue)
Like in the same period of the previous year, there were no milestones or the like.
(Operating Cost)
Along with the development of drug pipelines, the company made considerations and preparations for new clinical tests. R&D expenses were 598 million yen, up 307.5% year-on-year, and operating cost was 725 million yen, up 205.9% year-on-year.
(Operating income)
Operating loss was 725 million yen, up 488 million yen year-on-year.
2) Financial Conditions and Cash Flows
◎Main BS
| End of Mar. 2019 | End of Sep. 2019 |
| End of Mar. 2019 | End of Sep. 2019 |
Current Assets | 3,532 | 2,990 | Total Liabilities | 63 | 157 |
Cash | 3,508 | 2,862 | Total Net Assets | 3,504 | 2,880 |
Noncurrent Assets | 35 | 47 | Retained Earnings | -2,066 | -2,797 |
Property, Plant and Equipment | 31 | 43 | Total Liabilities, Net Assets | 3,567 | 3,037 |
Total Assets | 3,567 | 3,037 | Balance of Short and Long-Term Debts | 13 | 9 |
Unit: Million yen
Equity ratio was 94.5%, 3.4 points down year-on-year.
◎Cash Flows
| 1H of FY 3/19 | 1H of FY 3/20 | Increase/Decrease |
Operating cash flow | -247 | -735 | -487 |
Investing cash flow | - | -13 | -13 |
Free cash flow | - | -748 | -748 |
Financing cash flow | -5 | 103 | +108 |
Cash and Equivalents at the end of term | 528 | 2,862 | +2,333 |
Unit: Million yen
2-2 Fiscal Year ending March 2020 Earnings Forecasts
| FY 3/19 | FY 3/20 (Estimate) | YoY |
Operating Revenue | - | - | 0 |
SG&A | 592 | 1,066 | +474 |
R&D Expenses | 377 | 822 | +445 |
Other SG&A (R&D Expenses are excluded) | 215 | 244 | +29 |
Operating income | -529 | -1,066 | -537 |
Ordinary income | -672 | -1,066 | -394 |
Net loss | -673 | -1,069 | -396 |
Unit: Million yen
The earnings forecast is unchanged.
As for business revenue, considering the uncertainties of the progress of clinical tests and license negotiations, they thought that posting milestone compensation and lump-sum contract payment would not be appropriate at this stage. They plan to clarify the outlook when revenue is secured.
As for business expenses, while proceeding with the clinical phase III test for DFP-10917 in the U.S., the company plans to start clinical phase II test for DFP-11207 and the clinical phase I test for DFP-14927 in the U.S. Moreover, they will proceed further with case registration of the clinical phase II test of DFP-14323 in Japan, and plan to start clinical tests of DFP-17729 with their domestic alliance partner.
Along with such pipeline development, expenses for clinical tests will be more than those in the previous term, thus R&D expenses are projected to be 822 million yen, up 445 million yen year-on-year.
3. Growth Strategy
The company will steadily continue the development of the 3 products that are undergoing clinical tests and the 3 products for which the company is preparing for clinical tests and they plan to release one product per term from FY 2022. Furthermore, they will focus on securing alliance partners in Japan, China, Europe, and the U.S to maximize its revenue.
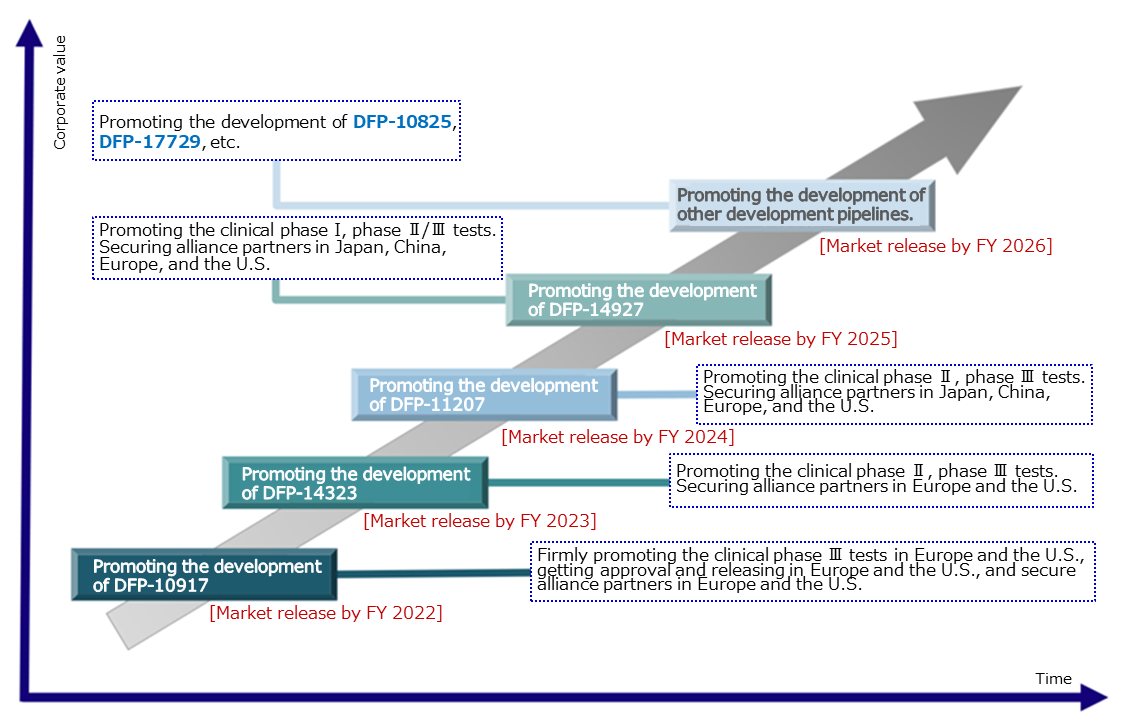
(Taken from the reference material of the company)
4. Conclusions
DFP-10917 development is making the most progress and its clinical phase III test started in the U.S. The clinical Phase III tests were updated in the U.S. Public organizations’ clinical tests registration website and patients’ registration started in November 2019. We will continue to pay attention to their progress. Further, we would like to expect the news release regarding licensing-out to pharmaceutical companies.
<Reference: Regarding Corporate Governance>
◎Organization type, and the composition of directors and auditors
Organization type | Company with audit and supervisory board |
Directors | 7 directors, including 4 outside ones |
Auditors | 3 auditors, including 2 outside ones |
◎Corporate Governance Report
The latest update: June 28, 2019.
<Basic policy>
Our company thinks that our mission is to operate our business while putting importance on the benefits of all stakeholders, including shareholders, clients, business partners, employees, and local communities, under the mission of “To provide treatment methods recommendable to cancer patients among relatives with peace of mind through module drug development.” To accomplish this, it is indispensable to develop our business stably and perpetually. Our basic policy for corporate governance is to improve systems for securing the soundness, transparency, and efficiency of business administration, which will become the base for the above-mentioned development.
<Reasons for Non-compliance with the Principles of the Corporate Governance Code>
It is written that “We follow all of the basic principles.”
This report is intended solely for information purposes and is not intended as a solicitation for investment. The information and opinions contained within this report are made by our company based on data made publicly available, and the information within this report comes from sources that we judge to be reliable. However, we cannot wholly guarantee the accuracy or completeness of the data. This report is not a guarantee of the accuracy, completeness or validity of said information and opinions, nor do we bear any responsibility for the same. All rights pertaining to this report belong to Investment Bridge Co., Ltd., which may change the contents thereof at any time without prior notice. All investment decisions are the responsibility of the individual and should be made only after proper consideration. Copyright(C) 2019 Investment Bridge Co., Ltd. All Rights Reserved. |



