Bridge Report:(6090)Human Metabolome Technologiesthe Fiscal Year June 2019
 Ryuji Kanno President | Human Metabolome Technologies (6090) |
 |
Company Information
Market | Mothers of Tokyo Stock Exchange |
Industry | Service Industry |
President | Ryuji Kanno |
HQ Address | 246-2 Mizukami, Kakuganji, Tsuruoka-city, Yamagata |
Year-end | End of June |
Homepage |
Stock Information
Share Price | Shares Outstanding | Total market cap | ROE Act. | Trading Unit | |
¥820 | 5,853,800 shares | ¥4,800 million | -41.9% | 100 shares | |
DPS Est. | Dividend yield Est. | EPS Est. | PER Est. | BPS Act. | PBR( x) |
¥0.00 | - | ¥-51.59 | - | ¥193.11 | 4.2 x |
*The share price is the closing price on the end of September 13. Number of shares outstanding, ROE, DPS, EPS and BPS are based on actual results of the previous term end of June.
Earnings Trend
Fiscal Year | Sales | Operating Income | Ordinary Income | Net Income | EPS | DPS |
March 2016 Act. | 780 | -70 | -71 | -71 | -13.41 | 0.00 |
March 2017 Act. | 914 | -43 | -40 | -61 | -10.86 | 0.00 |
March 2018 Act. | 938 | -140 | -149 | -156 | -26.92 | 0.00 |
June 2019 Act. | 989 | -526 | -515 | -596 | -101.92 | 0.00 |
June 2020 Est. | 920 | -300 | -299 | -302 | -51.59 | 0.00 |
*Unit: million yen, yen. Forecasts are those of the Company. Due to the change in the fiscal year end, the fiscal year ending June 2019 is 15 months. Net income is net income attributable to owners of the parent. The same applies hereinafter.
This Bridge Report provides the overview Fiscal Year ended June 2019 earnings results, etc. of Human Metabolome Technologies Inc and an interview with President Kanno.
Table of Contents
Key Points
1.Company Overview
2.Fiscal Year ended June 2019 Earnings Results
3.Progress and activities of the two businesses
4.Fiscal Year ending June 2020 Earnings Forecasts
5.Interview with President Kanno
6.Conclusions
<Reference: Regarding Corporate Governance>
Key Points
- The sales for the FY ended June 2019 were 989 million yen. In the metabolomics business, small and medium-scale transactions were healthy, but there were no large-scale transactions, whose sales were posted in the previous year, so the performance was sluggish. Operating loss was 526 million yen. In the metabolomics business, sales dropped and the cost for investment (for increasing staff, developing new analysis plans, etc.) augmented, increasing the deficit. As for the biomarker business, the company continued investment in the commercialization of a depression biomarker.
- For the FY ending June 2020, it is estimated that sales will be 920 million yen and operating loss will be 300 million yen. The company aims to increase sales to the level of the FY ended March 2018 (940 million yen), which was a 12-month accounting period. While making the new analysis plan contribute to the company’s full-year performance and cultivating the new market of metabolomics, the company aims to develop a new portfolio including the depression biomarker, mental health, and exosome-related businesses, into which the company will make inroads for the first time.
- In order to make the biomarker business profitable as soon as possible, the company launched a new exit strategy. From the viewpoint of prevention, the company will work on the establishment of 3 new business portfolios: “the mental health (mentality evaluation) project,” “the exosome project,” and “the project for developing health condition and disease risk prediction models.”
- We interviewed President Kanno about the review on the financial results in the previous term, the current situation and future outlook of the two businesses, the meanings of the change of the representative, his message toward shareholders and investors, etc. He said, “We have not yet lived up to the expectations of shareholders and investors sufficiently in the aspect of the speed of commercialization, but under the new management structure, all staff members of our company will join hands and strive to make the business profitable as soon as possible. I would appreciate your continued support.”
- The results of the metabolomics business were sluggish, but the company’s advantage in the metabolome analysis market is unchanged, and the interest in the usefulness of metabolomics is growing in a broad range of fields. Accordingly, the metabolomics business can be expected to grow steadily in this term and the future. As for the biomarker business, whose monetization is delayed, we, investors, are curious about the outcomes of the exit strategy launched under the new president Hashizume. It may be difficult to ask for the short-term results, but we would like to pay attention to quarterly trends and progress.
1.Company Overview
Human Metabolome Technologies Inc. is a venture company originated from Keio University, Its metabolome analysis technology is highly recognized throughout the world as a basic methodology. Its business model is to steadily generate profit through the commissioned metabolome business while investing in and working on research and development of biomarkers which estimates future prospects. The company is pursuing continuous growth with a stable revenue base under this strategy.
1-1 Corporate History
In 2001, Professor Tomoyoshi Soga of Keio University, Institute for Advanced Biosciences, developed a system to measure broad species of small-molecule metabolites (metabolome), which is called the CE-MS system. The new innovative system realized to capture a wide range of metabolites at once, while none of the conventional approaches do not, which means it used to be necessary to involve multiple experimental conditions, each of which has a small coverage.
Since before that, the metabolome analysis technology had been used for fundamental biological research, medicine creation, development of disease biomarkers, etc. and the growth of social needs for it was expected. Therefore, as the CE-MS method was established, Human Metabolome Technologies Inc. was founded in July 2003 by Professor Soga and Professor Masaru Tomita of the same university, Keio University, etc. for the purpose of commercializing it. It was the first venture company originated from Keio University, with a financial support from Keio University’s entrepreneurship fund.
The Company promoted research and development of its core technologies and also began organizing and establishing more specific commercialization process and business models in order to accelerate its growth speed through enhancing its visibility and raising funds for research and development. The Company got listed on the Mothers Section of the Tokyo Stock Exchange in December 2013, ten years after its establishment.
1-2 Corporate Philosophy
The Company defines the meaning of its existence as follows.
“To contribute to people’s health and joyful lives through research and development using the up-to-date metabolome analysis technologies for children in the future.”
The Company also sets the following 5 “Common Values”
1. We walk together with our customers. In order to respond to the current and future needs of our customers, we understand the workflow of our customers’ research and development, and develop solutions that meet our customers’ satisfaction. We communicate with our customers with passion wholeheartedly. |
2. We value the latest technology development and high quality. We never forget the venture spirit, and will continuously make efforts and investments in order to always develop the world’s leading metabolomics. We will also attempt to quickly produce high quality results that go beyond our customer’s expectation. |
3. We value teamwork. We will try to have open communication, trust our colleagues and respect for various opinions in order to maximize the power of teamwork. We will also work on self-development and elevate individual capacity in order to enhance the overall capacity of the team. |
4. We act fairly and honestly. As good citizens, we will comply with laws and ordinance and continue taking honest, ethical and responsible acts, in order to acquire trust from our customers, shareholders, and local communities and families. |
5. We contribute to the future of children. We will achieve appropriate life and work balance in order to pursue children’s happiness in families. |
1-3 Points to Understand the Company
The Company’s business overview is described later in this report together with the explanation of key words, “metabolomics” and “biomarker”; however, many technical terms show up in the section, and that may prevent the readers from understanding. Here are three points briefly provided before jumping to the section to glancing at the Company’s business scope.
➀ Significance of Social Presence
Biomarkers are in vivo compounds that are used as indicators to assess the current state of specific diseases. Blood sugar” for diabetes, “γ-GPT” for liver function disorder, and “uric acid” for gout are the representative examples of well-known and widely used biomarkers.
The Company discovered a biomarker for “major depressive disorder”, one of the current major social issues, and is developing diagnostic agents to quantify the condition of the disease.
Because there is no prevailing method to objectively measure the status of depression, despite the increase in the number of patients with depression, some serious problems are emerging regarding the therapy of depression. For example, patients who would have been cured if they had properly treated are still suffering or in over-prescription. If the diagnostic agents using the Company’s biomarker become to be widely used, these issues are expected to be solved, and social loss will be subsequently reduced. This social significance cannot be overlooked to understand the Company.
② Excellent Technological Capacities
The Company is highly recognized thanks to the “metabolome analysis technology” which is used to examine the complicated behavior of metabolites in human bodies to identify biomarkers.
The biomarker for depression is just an example. In Oct. 2017, the biomarker for acute encephalopathy was patented in Japan. With the technology, the Company is expected to identify and develop various new biomarkers in the future.
③ Stable Business Model
The Company’s current core business is the “commissioned metabolome business”, supporting research and development activities of research institutions and pharmaceutical companies that occupy the greater part of its sales. Sales of FY June 2019 were 988 million and operating income was 232 million, showing steady income. On the other hand, the “biomarker business”, which is expected to achieve significant growth in the mid- to long-term, is still operated in a small scale and experiencing losses. However, the Company has already established a balanced business model, in which the profit generated from the commissioned metabolome analysis business is invested into the biomarker business for its growth. This model is notable among many bio-venture companies that are suffering from gaining profit.
1-4 About depression
In the biomarker business, the company's future growth driver, the current target disease is depression. This summarized the outline and the current situation in Japan about depression and major depressive disorder.
◎What is Depression?
Depression is a type of mood disorder and is a brain dysfunctional state for various reasons such as accumulated physical and mental stress. Because the brain is not functioning properly, people in the depressed mood feel negative and low-esteem. This causes a vicious cycle in which people with depression feel more stress for matters that they could otherwise handle.
The “major depressive disorder” refers to the state in which depression mood continues even after the sources of stress are removed. In this regard, it is distinguished from adjustment disorders or some anxiety disorders and is considered a brain dysfunction, instead of a mere response to stress.
(Note: “Major” of the major depressive disorder means “primary” and does not mean “serious” depression.)
◎Number of depression patients in World and Japan
In 2012, the World Health Organization (WHO) announced that at least 350 million people were with depression which is a mental disorder. Almost 1 million lives are lost due to suicide every year, and over half of them are assumed to be due to depression.
According to the “Patient Survey” conducted by the Ministry of Health, Labor, and Welfare (MHLW) every three years targeting health facilities across the country, the total number of patients with mood disorders including depression increased from 430,000 in 1996 to 950,000 in 2011 (2.2x). The “Patient Survey” shows statistical data of the number of patients who visit health facilities. It is known that the consultation rate of patients with depression is low. Thus, according to the MHLW, it is suspected that the actual number of depression patients might be larger.
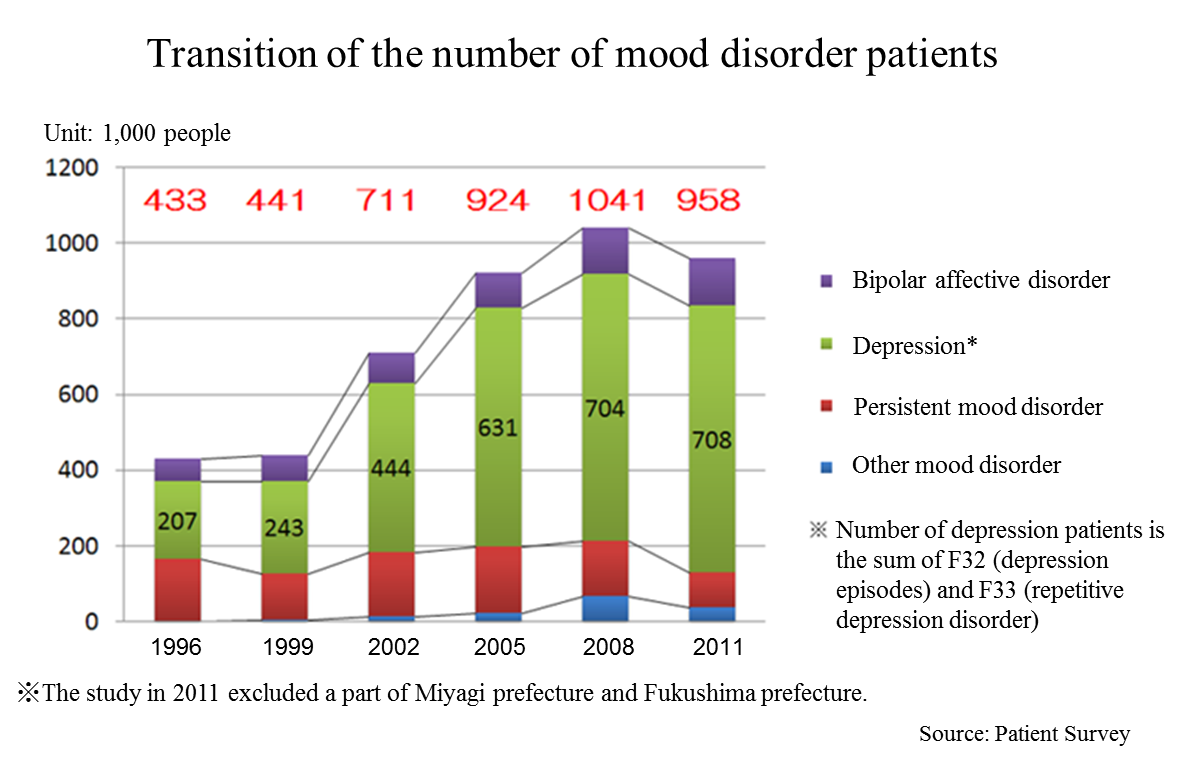
It is said that majority of depression patients are women and young people, however, in Japan, it is also frequently found among middle-aged people, and the social and economic impact of depression is significant. Needless to say, that depression distresses both patients and their families, and its social aspects cannot be passed over either; it leads reduction on the productivity of organizations and even suicide.
In Japan, economic losses due to depression and suicide are estimated to be about 3 trillion yen. If there were no such losses, GDP would increase about 2 trillion yen (an estimate by the Ministry of Health, Labour and Welfare, 2010).
Economic losses around the world are estimated to be about 62 trillion yen in 2002 (Screening for Depression in Adults: A Summary of the Evidence. Ann Intern Med. 2002.). The current economic losses are estimated to be over 100 trillion yen.
◎Treatment of Depression
If someone is diagnosed as having depression, the common treatment is prescription of “antidepressant drugs.” The antidepressant drugs can be categorized into several groups such as selective serotonin reuptake inhibitor (SSRI), serotonin-noradrenaline reuptake inhibitor (SNRI), and tricyclic antidepressant. In addition, anti-anxiety drugs or sleep-inducing drugs can be used, depending on the symptom.
The important thing under drug therapy is that the patients comply with the prescribed amount and frequency after they are informed about the effects and side effects. However, patients with depression often reduce the amount or frequency without doctors’ permission as they do not feel the symptom to be very serious, they worry side effects, etc. In these cases, the patients do not show improvement as doctors expect, and the doctors prescribe more drugs or change the types of drugs. That would often result in the delay of recovery or excessive administration of drugs, for lack of trust between doctors and patients. Therefore, it is essential to have objective assessment standards quantifying the state of depression or proving the depression has been cured. The biomarker and diagnostic agents of the depression that the Company is currently working on are extremely important for prompt and appropriate treatment of the disease.
1-5 Metabolomics and Biomarker
In order to understand the outline of the Company’s business activities, it is necessary to have a certain understanding of two key words, “metabolomics” and “biomarker”.
<What is metabolomics?>
Living organisms including human beings consist of some parts with various functions such as muscles, internal organs, and bones. “Metabolites” such as amino acids, fatty acids, and nucleic acids are common and major components of these organs. Metabolites play crucial roles for entire life activities.
Metabolites are provided by food and are consumed in the process of daily actions such as exercise. They move in a body and cells in accordance with their functions and are converted into new compounds through various chemical reactions, which are called “metabolism”. Adjusting body temperature, breathing, moving heart, digesting and absorbing food, transforming old cells into new ones are all operated by metabolism. The “compound conversion” to a new compound is based on a certain flow called metabolic pathway.
One of well-known approaches to understand the mechanisms of a human body is “genomics”, analyzing genes. Now, automated sequencing and computer analysis of genetic information (DNA base sequences) is available, and nearly all the information in the human genome has already been deciphered. However, much about the relationship between the roles of genes and diseases remains unknown. Recently, more researchers lean towards investigating metabolic profiles, in addition to genetic information coming out of genome analysis, in order to understand the relationships between a human body and diseases. Consequently, research and use of “metabolomics (metabolome analysis)” targeting all metabolites is becoming increasingly popular.
The metabolome analysis is mainly used in the following fields:
Research on pathological mechanisms at research institutions such as universities. |
Exploration and pharmacological and toxicological studies by pharmaceutical companies. |
Improvement of productivity by companies that manufactures products using fermentation. |
Component analysis as well as exploration and confirmation of functionality at food companies |
<What are biomarkers?>
A human body puts various vital functions under high-sophisticated manipulation to minimize internal and external influences, and subsequently keeps the body condition stable. That mechanism is called “homeostasis”. For example, body temperature and heartbeat may change temporarily, but return to regular ranges.
Diseases lead abnormal homeostasis and metabolic compound, which are different from those in healthy conditions. This metabolite compound is called a biomarker. By measuring a biomarker, the current status of a specific disease can be objectively assessed.
Blood sugar as a pancreas function indicator, γ-GTP as a liver function indicator, biomarker PSA for prostatic cancer and biomarker CA19-9 for pancreatic cancer are examples of well-known biomarkers.
Biomarkers have been studied for a long time in order to monitor the status of diseases. These days, with new methods to analyze multiple compounds with higher sensitivity all at once, study results of various new biomarkers have been publishing one after another. Among the biomarkers that are explored through metabolomics technologies are the followings:
Biomarkers to diagnose the presence of diseases |
Biomarkers to assess effects of treatments |
Biomarkers to predict side effects caused by drug administration |
Biomarkers to predict effects of drug administration |
1-6 Business Contents and Business Model
The main businesses pillars of the Company are the “metabolome business” and “biomarker business”. It is trying to expand the metabolome study related market by disseminating the excellence of its CE-MS system, the Company’s core technology, to research institutions and pharmaceutical companies, and securing income base by expanding the metabolome business in Japan and overseas.
Previously, the company allocated the profit from the “metabolome business,” which is the current mainstay business, to the investment in the R&D of the promising “biomarker business,” and applied the intellectual property obtained through the R&D to the development of medicines and disease diagnosis, with the goal of growing in the mid to long terms. From the term ending Mar. 2017, the company will accelerate the investment in the biomarker business by procuring funds from the outside in order to flourish in the future.
Earnings structure and customers of each business are as follows.
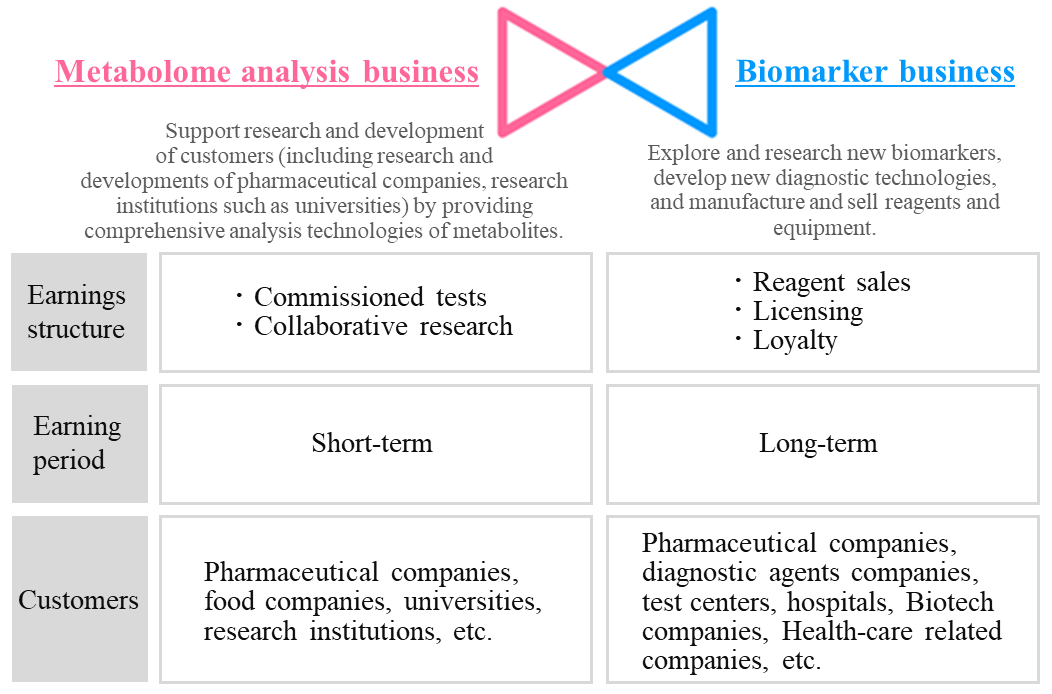
(Source: the company)
① Metabolome Business
“Sales were 988 million yen and operating income was 232 million yen for FY June 2019”
The Company receives orders from various customers, e.g., private companies such as pharmaceutical companies and food companies as well as universities and public research institutes.
The scheme of the service is as follows. A customer sends samples to the Company, and then metabolites are extracted from the samples. The extracted metabolites are measured by the CE-MS system, and the acquired data is analyzed. After all, the report is delivered to the customer. The data obtained from the service are used for various purposes; basic biological study, assessment of drug effectiveness and toxicity assessment by pharmaceutical companies, universities, and research institutions, analysis of fermentation process and functional evaluation of functional foods by food companies. The data is contributing to the progress of research and development activities of customers. In recent years, not only healthcare, food, but also interest towards health-related companies is emerging rapidly. The Company has not only accumulated a wealth of achievements with about 5,540 tests since the foundation up to the fiscal year June 2019, but also receives a high reputation from customers for their quality.
◎Deployment in the Overseas Market
In order to distribute the commissioned metabolome analysis service in Asia, the Company concluded an exclusive sales authority agreement with Young In Frontier Co., Ltd. in South Korea in June 2011. Moreover, the Company hired a sales representative in charge of Asia-Pacific area to develop Asian market including Singapore and Hong Kong, outside of South Korea. Furthermore, in order to expand its business in the North American market, it also established its sales subsidiary, Human Metabolome Technologies America, Inc., in October 2012 at Cambridge, Massachusetts, USA, home to many medical research institutions.
In order to further accelerate overseas development, in May 2017, the company established a subsidiary (sub-subsidiary) "Human Metabolome Technologies Europe B.V." in Europe (Netherlands) through HMT-A.
◎”C-SCOPE” –a service package of the commissioned metabolome analysis service for cancer study
In August 2012, the Company launched “C-SCOPE”, a service package of the commissioned metabolome analysis service well organized for cancer study. C-SCOPE was developed to respond to the needs; they would like to measure concentrations of specific metabolites changing inside cancer cells with higher sensitivity and higher accuracy.Its technologies are based on unique and efficient metabolites extraction method from cancer cells and highly sensitive analytical system.
Cancer is the number one cause of death in Japan since 1981 and occupies about 30% of the recent total causes of death. According to the MHLW, the costs for cancer research are increasing year by year; in 2012, ¥35.7 billion were spent. It is an urgent task for many pharmaceutical companies to develop effective new anticancer drugs.
The “Warburg effect” is the phenomenon that most cancer cells have glycolytic rates of several to dozens of times to that of normal cells. Although this effect was proposed as far back as over 80 years ago, the research on the effect made little progress since there was no method to comprehensively measure metabolites back then.
Thanks to the dramatic advancement in metabolomics technologies in recent years, development of anticancer drugs which act as metabolic inhibitors are underway.
The Company’s metabolome analysis technology based on the CE-MS system is considered as one of the effective analysis systems applicable at each stage of cancer research; from the basic study of cancer biology to clinical application in the process of anticancer drugs development.
② Biomarker Business
“Sales were 0.4 million JPY and operating loss was 204 million JPY for FY June 2019”
The Company considers the business related to biomarkers, which play an important role in occasions such as early diagnosis or monitoring treatment effects, as a driver for future growth, and is proceeding with biomarker discovery and development of clinical test drugs through collaborative research and development with universities, pharmaceutical companies, and diagnostic drugs companies.
The Company develops new diagnostic methods by using biomarkers, which were acquired through its own R&D, or biomarkers introduced from outside. Additionally, through the process of product development and clinical development, the Company produces and sells in vivo diagnostic drugs and diagnostic equipment.
The sales in this business are composed of cooperation money for R&D from pharmaceutical companies in cooperative research, milestone revenues, and loyalty gained from the sales of the drugs when they are commercialized.
Furthermore, in order to make the business profitable early, the company is developing 3 new business portfolios: “the mental health (mentality evaluation) project,” “the exosome project,” and “the project for developing health condition and disease risk prediction models.”
◎Intellectual Property Policy
The personnel in charge of intellectual property and contracts works on patent application and requests for examination of all projects and are in close collaboration with the patent attorneys of the Company and its collaborative research institution. They are also responsible for negotiation concerning collaboration research agreements and development of agreement documents. The Company attempts to maximize its rights when obtaining a patent of newly discovered biomarkers.
Since the scope of the rights differs depending on the marker, the Company generates patent application documents in a way that would cover a wide scope of the rights such as chemical structure of the biomarker, usage for diagnostics and drug development, detection method and measurement equipment. Furthermore, the Company makes it a principle to file international patent applications in accordance with the Patent Cooperation Treaty, in anticipation for the future license agreement and market based on the information of clinical test drugs, test equipment companies and pharmaceutical companies of various countries.
As of June 2017, the "basic patent" on the method of biomarkers of depression diagnosis etc. has been registered in Japan, the US and China. It has been filed in Europe and it is undergoing the patent evaluation there.
The patent on the method of measuring ethanolamine phosphate (PEA) has been registered at all four stations in Japan, the US, China and Europe.
◎Example of Biomarker Business: Depression Biomarker
The Company especially focuses its research and development on a) the central nervous system disorder such as depression (e.g. mood disorder and mental disorder) for which there are few objective diagnosis methods b) diseases that
have become social problems such as metabolic syndrome, including hepatitis and diabetes, and their related diseases. Its current focus is the biomarker for depression.
Diagnosis of the major depressive disorder is conducted using the diagnostic standards provided by the American Psychiatric Association or the standards of the World Health Organization (WHO). However, both of them largely reflect the subjective view of the doctor or patient, and unlike other diseases, no diagnostic method has been established on the basis of objective indicators.
The Company conducted collaborative research with the National Center of Neurology and Psychiatry, and discovered a blood biomarker of the major depressive disorder.
Blood samples were collected from approximately 30 patients and 30 healthy people, and the blood components were compared through metabolome analysis using the CE-MS system. As a result, the patients with major depressive disorder showed lower concentration of (PEA) in their serum.
Through further analysis, it was found that a) PEA is a specific biomarker for major depressive disorder, and b) PEA level will return to the healthy standard range when MDD is treated.
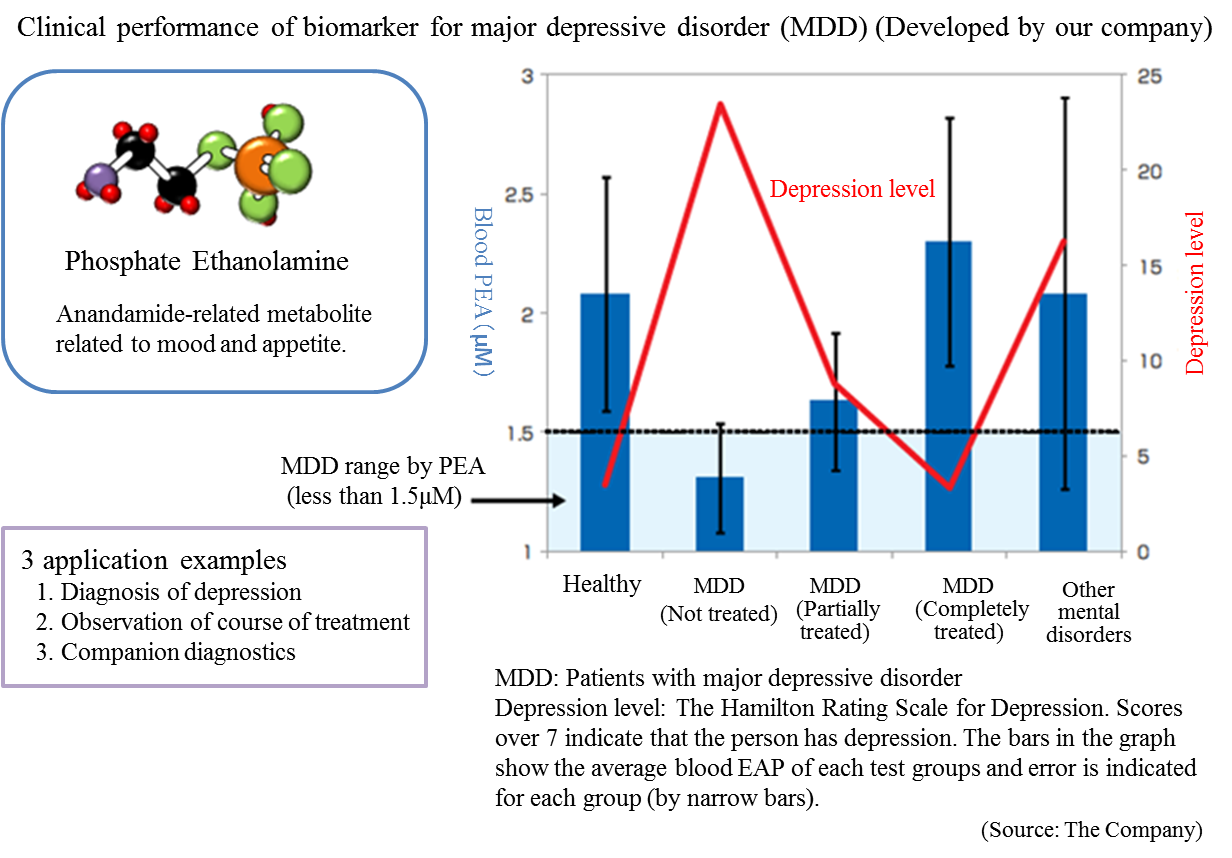
(Source: the company)
*Companion diagnostics: Refers to clinical tests to predict effects and side effects of pharmaceutical products before administration. By checking the responses of individual patients towards a drug before treatment, more effective drug administration can be provided.
◎Identification of Disease Biomarkers
The Company uses the following three connections and systems for identifying biomarkers in order to expand biomarker development pipelines.
<Connection with the Customers for Commissioned Analysis or Collaborative Development>
The Company accepts requests from universities and companies for the tests for finding biomarkers. The Company also receives proposals for collaborative development before or after the tests. Currently, a collaborative development project for diabetic nephropathy biomarkers is ongoing.
<Direct Proposal to Researchers and Physicians>
The Company’s researchers directly propose research plans for the development of disease biomarkers to researchers or physicians, and establish collaborative study agreement with the institution based on approvals from the collaborative researchers or physicians. The target diseases are chosen according to the number of patients, compatibility to the analysis technologies of the Company, degree of social contribution, and necessity of biomarkers. In addition to major depressive disorder, the Company is working on the development of biomarkers for non-alcoholic steatohepatitis and fibromyalgia.
<HMT Research Grant for Young Leaders in Metabolomics>
The Company offers a grant (HMT Research Grant for Young Leaders in Metabolomics) to graduate students to disseminate the usefulness of metabolomics in the society and nurture young researchers. From the research themes submitted from graduate students across the world, the Company chooses excellent proposals, and supports their research by awarding metabolome analysis service without a fee. Fourteen students have been awarded in the last 4 years. Some of these study results actually led to the identification of biomarkers and evolved to collaborative study with the Company, for example, infectious disease-related encephalopathy biomarker.
2.Fiscal Year ended June 2019 Earnings Results
(1) Consolidated Business Results
| FY 3/18 | Ratio to sales | FY 6/19 | Ratio to sales |
Sales | 938 | 100.0% | 989 | 100.0% |
Gross profit | 742 | 79.1% | 672 | 68.0% |
SG&A | 883 | 94.2% | 1,198 | 121.2% |
Operating Income | -140 | ― | -526 | ― |
Ordinary Income | -149 | ― | -515 | ― |
Net Income | -156 | ― | -596 | ― |
*Unit: million yen. 3/18 is the 12-month term from April 2017 to March 2018, while FY 6/19 is the 15-month term from April 2018 to June 2019.
The investment for growth increased, and a loss was continuously posted.
Sales were 989 million yen. In the metabolomics business, small and medium-scale transactions were healthy, but there were no large-scale transactions, whose sales were posted in the previous year, so the performance was sluggish.
Operating loss was 526 million yen. In the metabolomics business, sales dropped and the cost for investment (for increasing staff, developing new analysis plans, etc.) augmented, increasing the deficit. As for the biomarker business, the company continued investment in the commercialization of a depression biomarker.
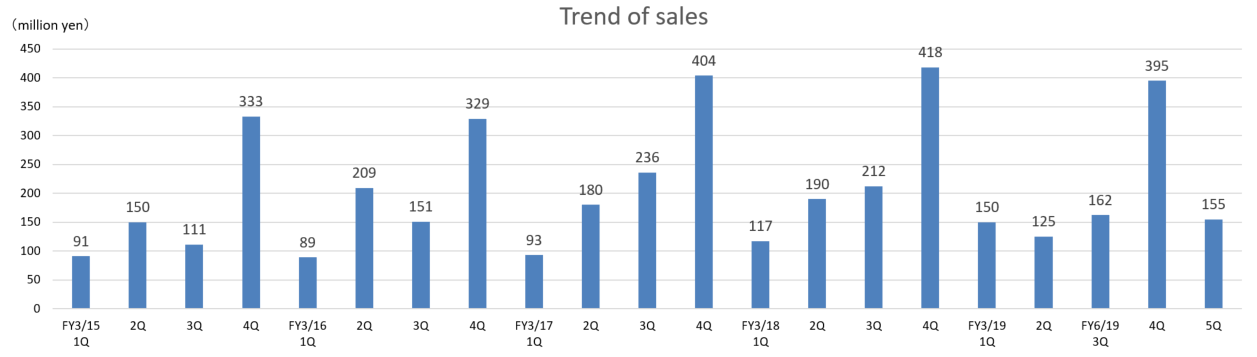
As shown in the above graph, the metabolomics business, which is now the core business of the company, tends to undertake analysis tasks mainly in January to March.
The company changed the account closing month from March to June, for the purpose of alleviating the effects of seasonal changes in sales, etc. on results and formulating management plans, etc. efficiently.
(2) Highlights by Major Segment
◎Metabolome business
| FY 3/18 | FY 6/19 |
Sales | 936 | 988 |
Operating Income | 445 | 232 |
*Unit: million yen. FY 3/18 is a 12-month settlement from April 2017 to March 2018. FY 6/19 is a 15-month period from April 2018 to June 2019.
The amount of orders received as of the end of June 2019 was 263 million yen, up 97 million yen from the end of June 2018.
◎Biomarker business
| FY 3/18 | FY 6/19 |
Sales | 2 | 0 |
Operating Income | -185 | -204 |
*Unit: million yen. FY 3/18 is a 12-month settlement from April 2017 to March 2018. FY 6/19 is a 15-month period from April 2018 to June 2019.
(3) Financial standing and cash flows
◎Main BS
| End of March 2018 | End of June 2019 |
| End of March 2018 | End of June 2019 |
Current Assets | 1,762 | 1,214 | Current liabilities | 122 | 116 |
Cash | 1,261 | 948 | Lease obligations | 9 | 10 |
Receivables | 220 | 70 | Noncurrent liabilities | 47 | 37 |
Securities | 200 | 100 | Lease obligations | 32 | 20 |
Noncurrent Assets | 158 | 153 | Total Liabilities | 168 | 152 |
Tangible Assets | 140 | 131 | Net Assets | 1,753 | 1,214 |
Intangible Assets | 9 | 10 | Shareholders’ Equity | 1,693 | 1,112 |
Investment, Others | 10 | 12 | Total liabilities and net assets | 1,921 | 1,367 |
Total assets | 1,921 | 1,367 |
|
|
|
*Unit: million yen
Current assets decreased 548 million yen from the end of the previous term, due to the decline cash and deposits, trade receivables, etc. Noncurrent assets were almost unchanged, and total assets dropped 553 million yen to 1,367 million yen. Total liabilities declined 15 million yen to 152 million yen, due to the decrease in lease obligations.
As retained earnings dropped further, net assets decreased 538 million yen to 1,214 million yen.
As a result, capital-to-asset ratio declined 6.5 points from 89.2% to 82.7%.
◎Cash Flow
| FY 3/18 | FY 6/19 |
Operating Cash Flow | -178 | -265 |
Investing Cash Flow | -87 | -54 |
Free Cash Flow | -265 | -320 |
Financing Cash Flow | 4 | 3 |
Cash and equivalents | 1,361 | 1,048 |
*Unit: million yen. FY 3/18 is a 12-month settlement from April 2017 to March 2018. FY 6/19 is a 15-month period from April 2018 to June 2019.
The deficits of operating CF and free CF expanded, due to the augmentation of current net loss before taxes and other adjustments. The cash position declined.
(4) Topics
① The blood biomarker for depression underwent patent evaluation in Europe.
Phosphoethanolamineas biomarker of depression, whose patent was jointly applied for by the company and the director Noriyuki Kawamura of Kawamura Institute of General Practice, underwent patent evaluation in Europe.
This patent has been already registered in Japan, the U.S. and China.
② The joint research course “Metabolomics Innovation Course” established in Graduate School of Medicine, Hirosaki University
Hirosaki University and the company established the joint research course “Metabolomics Innovation Course” in Graduate School of Medicine, Hirosaki University on May 1, 2019, and held a ceremony for opening the course on June 7.
In January 2017, the company joined the innovation-triggering program “COI (Center of Innovation) STREAM” (led by the specially appointed professor Shigeyuki Nakaji of the social medicine course of Graduate School of Medicine, Hirosaki University), and the newly established course is aimed at “finding biomarkers that would contribute to mental and physical health” and “biomarkers that would help predict diseases at an extremely early stage” by analyzing big data on a large number of items obtained from living specimens in Iwaki Health Promotion Project, etc. and Omics-based data, mainly metabolomics ones.
Furthermore, it aims to develop a model for predicting health conditions with multi-markers using mechanical learning and a model for predicting future disease risks and put them into practice.
The company will keep concentrating on the development of biomarkers with the CE-MS method, which is the core technology, and aims to create diagnosis techniques and solutions with unique value by utilizing discovered innovative biomarkers.
③ Replacement of the representative director scheduled
At the meeting of the board of directors held in August 14, 2019, they decided to replace the representative director, considering the results of deliberation by an arbitrary nomination/remuneration committee.
◎ Replacement of the representative director
Name | New post | Current post |
Ryuji Kanno | Director and chairperson | Representative director and president |
Katsuhito Hashizume | Representative director and president | Director |
This replacement is aimed at coping with significant changes in the business environment, rejuvenating the top management, strengthening and vitalizing the management base under a new management structure, achieving sustainable growth, and improving its corporate value.
Mr. Hashizume joined HMT in March 2018, after serving as the head of the business development department, the head of the sales department, the head of the entrusted development department, etc. at Takara Bio Inc. In December 2018, he was appointed as a director and executive officer, a president of the biomarker business company, and a representative director and president of HMT Biomedical Co., Ltd. This is scheduled to be approved at the 16th annual meeting of shareholders on September 21, 2019.
3. Progress and activities of the two businesses
(1) Metabolomics business
① External environment
Metabolomics has evolved from a technology for academia, including universities and laboratories, into a technology for the industrial field. In these circumstances, the markets of healthy foods, including functional foods, and the health-oriented markets are expanding with such keywords as sports, food products, sleep, and stress. An increasing number of enterprises are interested in the usefulness of metabolomics for grasping human health conditions and aim to develop new businesses related to healthcare. Then, a new market is emerging.
In the field of development of medicines, there are growing needs for various things, including the research into intestinal bacteria, the early detection, diagnosis, and treatment methods for neuropsychiatric disorders, such as dementia and Alzheimer's disease, the practical application of medical technologies, including medicines for refractory diseases, and the search for biomarkers for anticancer companion diagnostic agents, and the use of metabolomics is increasingly considered effective.
As for overseas situations, the scale of the U.S. market is 5 times larger than that of the Japanese market. The average annual growth rate of the Chinese market is 10-20%, and the Chinese market is estimated to become larger than the Japanese market from 2020 to 2022. Accordingly, this business can be expected to grow significantly inside and outside Japan.
Assumed market scale
The year 2017
The U.S. | 6.24 billion yen |
Germany | 1.35 billion yen |
The U.K. | 1.21 billion yen |
France | 0.85 billion yen |
Japan | 1.23 billion yen |
China | 0.91 billion yen |
(Taken from the reference material of the company)
② Primary measures
In these favorable circumstances, the company implemented the following measures in the FY ended June 2019.
Theme | Measures | Results |
(1) To increase sales by releasing new products in growing markets | 【Enhancement of sales promotion in the food product field】 To make large-scale transactions by intervening in clinical trials To offer comprehensive solutions by creating new health value To appeal to clients with its value to conclude annual contracts with them 【Enhancement of sales promotion in the medical field】 To cultivate the neuropsychiatric, emerging, and re-emerging infectious disease fields by releasing new products Sales promotion focused on the field of research into refractory diseases Enhancement of activities for making large-scale transactions aimed at finding biomarkers | Large-scale orders decreased in the food product field.
Orders from Japanese pharmaceutical companies decreased, and the sales of products for pharmaceutical companies dropped. |
(2) To put more energy into overseas business— Acceleration of overseas business by fostering global brand power | 【the U.S.】 To make large-scale transactions, expand target areas for sales expansion targeting mainly clinical research and pharmaceutical companies and fortify the organizational base with a new structure 【Europe】 To enhance activities for making clinical transactions focused on specific diseases Foray into the food product field 【Asia】 To make inroads into the Chinese market, cultivate the Asian market further, and expand the target area for sales expansion | In the U.S., sales dropped, because the filling of vacancies for sales staff after the retirement of some sales staff was delayed, governmental clients demanded discounts, and so on. To deal with major problems, countermeasures have been designed.
In Europe, the company made transactions with mega pharma companies and medicine creation ventures, showing steady growth.
In China, the company received some orders. From now on, it will put importance on alliances. |
(3) To develop new businesses | To develop mid-term businesses other than the business of undertaking metabolome analysis | The company opened a joint research course in Graduate School of Medicine, Hirosaki University |
◎ U.S. market
In the U.S., where the number of orders received and sales declined from the previous term, the company will take the following measures, to recover its performance.
*Enhancement of popularity
The popularity of the company’s brand is still low, and its marketing is insufficient. Accordingly, the company will improve its website, intensify the activities of Webinar (online seminar), and participate in academic conferences mainly in the fields in which the company specializes.
*Price
As the company has confidence in quality, it has provided products at higher prices than competitors, but there are growing requests for lower prices from academia, etc. Accordingly, the company will make prices comparable to competitors’ when it is not busy, and measure its effects, and then discuss the possibility of revising prices based on the results.
*Outbound sales
The filling of vacancies for sales staff and the education of new sales staff were delayed, and the number of business talks in outbound sales decreased, but the sales staff recruited last fiscal year are now prepared as a whole. In addition, the company aims to visualize the sales process and improve sales efficiency.
*Large-scale transactions
The number of calculable large-scale transactions in the U.S. is actually smaller than that in Japan. If they rely on small-scale transactions, there is a risk that sales will not be stabilized. Therefore, the company will enhance marketing with reference to the successful cases in the pharmaceutical and medicine-creation fields in Europe, and propose medium-sized projects to major clients with the aim of obtaining grants, to increase large-scale transactions.
◎ Chinese market
The company received several orders, and completed them with quality highly evaluated.
However, through the tightening of domestic regulations in China, sending human-derived samples to Japan was prohibited. In addition, it would take a lot of time to establish a nationwide marketing channel and a laboratory by itself.
In this situation, the company will prioritize the securing of partners while utilizing the uniqueness of the CE-MS platform and analysis, rather than trying to make inroads into the Chinese market by itself. In parallel, the company will discuss services suitable for the Chinese market.
(2) Biomarker business
As the company became technically able to offer the PEA testing to 350 million patients of depression around the world by developing the “PEA measurement reagent kit based on the enzyme method,” it is developing a foundation for commercialization. In order to make this business profitable early, the company decided to work on the establishment of 3 new business portfolios in the FY ending June 2020.
(Recognition of the current situation)
The following facts have been confirmed from the progress and results of the PEA tests (clinical feasibility tests) on multiple specimens conducted inside Japan.
In some cases, even normal people have a low PEA value like depression patients, due to the differences among individuals. |
Since the difference in PEA concentration variation between normal people and depression patients is relatively small, it is important to obtain a stable PEA measurement. To do so, it is necessary to appropriately manage specimens when sampling them, storing them, etc. and it is essential to overcome the problems with specimen management and usage assuming the use in the medical care field. |
PEA remains a promising candidate for a biomarker, but the processes the company has engaged in have many problems to solve before full-scale commercialization. Accordingly, the company decided to address the problems from the following two aspects: “diagnosis” and “prevention,” in order to make the business profitable early.
As for “prevention,” the company will work on the establishment of new business portfolios, which is the key to making the business profitable early.
① Diagnosis
The company will discuss specimen management methods, etc. again, as the problems with them have emerged, and conduct research into not only diagnosis of depression, but also the examination of treatment effects (follow-up), etc.
Concrete activities are as follows.
<Clinical and academic research>
Methods for the transportation, management, etc. of specimens are being discussed. |
The company plans to conduct clinical feasibility tests in the U.S. |
The company plans to discuss the correlation between the recovery from depression and the PEA value. |
Research for elucidating the mechanism of the decrease in plasma PEA with model animals for depression |
The clinical feasibility tests in the U.S. are expected to give a lot of feedback that would contribute to domestic commercialization.
<Situation of development of detection technologies>
Development of a measurement method with liquid chromatography The company is striving to improve the sensitivity and robustness of PEA measurement and reduce its cost. |
Development of an enzyme method reagent kit The company is developing a reagent kit for large-scale versatile biochemical analysis equipment. |
The company will further improve the development activity it has been conducting.
② Prevention
The company will discuss the development of evaluation indicators that would contribute to the prevention, early detection, and prevention of recurrence of depression. The company will proceed with the development of new business portfolios.
New portfolio 1: the mental health (mentality evaluation) project
Due to the enforcement of the amended Industrial Safety and Health Act, every office which has 50 or more workers is now obligated to conduct a stress check. As enterprises that advocate “health-oriented business administration” are increasing, the needs for mental health care have been growing rapidly in recent years.
In this trend, the company will work on the development of indicators for evaluating mental conditions as measures for preventing stress and depression rather than the diagnosis of depression.
The company plans to carry out collaborative research with the department of medicine of a prestigious university, with the aim of establishing evaluation indicators using multiple factors, including PEA.
Assumed clients are enterprises, mental healthcare consulting firms, etc.
New portfolio 2: Exosome project
Focusing on the exosome*, which is considered as a mine of biomarkers, the company acquired the domestic and overseas exclusive distribution rights for “ExoIntactTM Exosome Purification Reagent Kit” for research, which was developed by Siliconbio Inc. (Higashi-hiroshima-shi, Hiroshima Prefecture) (in April 2019, for the domestic one; in June 2019, for the overseas one), and started selling the product.
*Exosome
A particle with a diameter of 30 to 150 nm secreted from a cell. It has been confirmed that it exists in body fluids, such as blood, urine, breast milk, and saliva.
The exosome includes micro RNAs, nucleic acids, proteins, and metabolites. It is considered to transmit information among cells in a living organism.
At present, a lot of research into the exosome is being conducted around the world, because the materials inside it and the exosome itself may function as a biomarker for diagnosing diseases and may be utilized for treating various diseases.
“ExoIntactTM Exosome Purification Reagent Kit” can purify the exosome easily and swiftly compared with the conventional ultra-centrifugal method, which requires expensive apparatus.
In addition, the peptide and magnetic beads that have a strong affinity with the phospholipid included in the exosome membrane are used, so that the exosome can be eluted under mild conditions without using any denaturants. Since the exosome can be purified as it is, it can be applied to various applications. Accordingly, the company aims to enrich the product lineup and release reagents for research into the exosome and services of undertaking such research, and will proceed with product development in the exosome industry, including medicine, healthcare, and food products for not only people, but also pet animals.
Assumed clients include universities and laboratories.
New portfolio 3: the project for developing health condition and disease risk prediction models
Through collaborative research with universities, the company will search for biomarkers for early detection of diabetic nephropathy.
As mentioned above, the company established the joint research course “Metabolomics Innovation Course” in Graduate School of Medicine, Hirosaki University, with the aim of developing models for predicting health conditions and disease risks, and putting them into practical use.
4.Fiscal Year ending June 2020 Earnings Forecasts
(1) Earnings Forecasts
| FY 6/19 | FY 6/20 Est. |
Sales | 989 | 920 |
Operating Income | -526 | -300 |
Ordinary Income | -515 | -299 |
Net Income | -596 | -302 |
*Unit: million yen. The forecasted values were provided by the company. FY 6/19 is a 15-month period from April 2018 to June 2019.
The company will work on the sustainable growth of sales, the improvement in productivity of the metabolomics business, and the redevelopment of portfolios in the biomarker business.
It is estimated that sales will be 920 million yen and operating loss will be 300 million yen. The company aims to increase sales to the level of the FY ended March 2018 (940 million yen), which was a 12-month accounting period.
While diffusing the new analysis plan and cultivating the new market of metabolomics, the company aims to develop new portfolios including the depression biomarker, mental health, and exosome-related businesses, into which the company will make inroads for the first time.
The 4 major managerial policies are as follows.
① Sustainable growth of sales and attainment of budget targets
The company will develop a foundation for a mid-term leap by cultivating new domains and regions with new products and analysis plans, and developing new businesses.
② Improvement in productivity of the metabolomics business
The company will streamline sales activities by revising sales methods, etc., and improve productivity by shortening analysis time, etc.
③ Redevelopment of portfolios in the biomarker business
The company will proceed with the practical application and commercialization of depression biomarkers, and develop new pipelines and biomarker-related businesses with the aim of making the business profitably early.
④ Securing of loyal shareholders and retention of them through dialogues
The company will continue IR activities for institutional and individual investors.
(2) Trends by segment
◎Metabolome analysis business
| FY 6/19 | FY 6/20 Est. |
Sales | 988 | 908 |
Operating Income | 232 | 161 |
*Unit: million yen. The forecasted values were provided by the company. FY 6/19 is a 15-month period from April 2018 to June 2019.
Major activities are as follows.
(In Japan)
* | To cultivate new healthcare markets in the food product and chemical fields in addition to targeting research institutes, academia, and pharmaceutical companies. |
* | To expand the range of clients by enhancing sales activities and sales support, and make small and medium-scale transactions, without sticking to large-scale ones. |
* | To improve the productivity of processes from the transportation of specimens to the submission of reports, including analysis, and reduce costs after receiving orders. |
(Overseas)
* | To cultivate the markets of medicine creation and academia via distributors in Europe. |
* | To increase the rate of utilization of analysis equipment in a period from April to November, and enhance price elasticity strategically, for the purpose of boosting sales in Europe and the U.S. |
* | To establish a new tie-up model in the Chinese market, where laws and regulations have been amended. |
◎Biomarker business
| FY 6/19 | FY 6/20 Est. |
Sales | 0 | 12 |
Operating Income | -204 | -172 |
*Unit: million yen. The forecasted values were provided by the company. FY 6/19 is a 15-month period from April 2018 to June 2019.
(3) Priority investment items
Items | FY 6/19 | FY 6/20 Est. | Overview |
R & D expenses | 189 | 150 | Development associated with the mental health project and the commercialization of depression biomarkers. |
Capital investment | 152 | 56 | The focus is on investments to increase the sensitivity of metabolome analysis. The investment for the new market is one paragraph. |
*Unit: million yen. The budget is announced by the company 6/19 is a 15-month period from April 2018 to May 2019.
5.Interview with President Ryuji Kanno
We asked President Kanno about the results of the previous term, the current condition and future prospects of both businesses, the significance of the reshuffling of representatives, and for his message toward shareholders and investors.
Q: “Firstly, could you give us your review of the results of the previous term?”
A: “Our metabolomics business received no large orders in the previous term, so it was sluggish. However, the market is firm, and the numbers of customers and inquiries are on an upward trend. We were able to receive orders from large pharmaceutical companies in Europe, and in this term, we are aiming for improved performance in the United States, too.”
Unfortunately, our metabolomics analysis business was sluggish since it received no large orders in the previous term. However, the market is firm, and we are by no means losing our market share. The numbers of customers and inquiries are on an upward trend.
Furthermore, I think the fact that we were able to receive orders from large European pharmaceutical companies was an important result that would improve our company's value.
On the other hand, we saw decreases in sales and orders in the United States. However, we are starting to see a way to improve our performance, including our pricing strategy, so we are aiming for an improvement in this term.
Regarding the Chinese market, which is sure to achieve rapid growth, we had not predicted that there would be a prohibition on taking human-derived samples out of the country. However, there exist companies that pursue business opportunities in this growing market and want to work with us. We will continue to cultivate the market steadily, while forming alliances.
Q: “How is the biomarker business going?”
A: “We came out with a new exit strategy for achieving monetization as soon as possible. In the field of prevention, we will work on the creation of three new business portfolios, which are the “the mental health (mentality evaluation) project,” “the exosome project,” and “the project for developing health condition and disease risk prediction models.”
We came out with a new exit strategy for achieving monetization of our biomarker business as soon as possible.
While ethanolamine phosphate (PEA) remains a plausible biomarker for major depressive disorder, there are many challenges that need to be solved before full-fledged commercialization can be achieved. Therefore, we have decided to work on “diagnosis” and “prevention” as two exits in our exit strategy. Under “diagnosis,” we will go beyond merely diagnosing major depressive disorder, and will carry out research aimed at the verification of the effectiveness of treatment (follow-up observation). Under “prevention,” which we will develop as a new business portfolio, we will study the development of indicators that contribute to the prevention, early detection, and relapse prevention of major depressive disorder.
In the field of “prevention,” we will work on the creation of three new business portfolios, which are the “the mental health (mentality evaluation) project,” “the exosome project,” and “the project for developing health condition and disease risk prediction models.” We see these portfolios as the key for achieving monetization as soon as possible.
Q: “At the current ordinary general meeting of shareholders, you will be appointed as a director and chairperson as a result of the reshuffle of representatives. What is the goal of this change?”
A: “This is a reshuffle aimed at responding to the large changes in the business environment in addition to sustainable growth and development. Furthermore, it aims to increase our enterprise value. I think the new president Mr. Hashizume is well suited, given that he can use his extensive experience, knowledge, and expertise to create solutions by offering ideas and perspectives which are new to our company. My primary duties are to figure out how we maintain relations with stakeholders and to improve and develop them.”
This is a reshuffle aimed at responding to the large changes in the business environment in addition to sustainable growth and development. Furthermore, it aims to increase our enterprise value. Moreover, this reshuffle aims to rejuvenate the managerial class, in addition to working towards the strengthening and invigoration of our management base as the foundation of our new management structure.
The new president Mr. Hashizume has many years of experience developing biotechnology-related businesses, starting with Takara Bio, Inc.
I think he is well suited, given that he can use his extensive experience, knowledge, and expertise to create solutions by offering ideas and perspectives which are new to our company in both the metabolomics and biomarker businesses.
As chairperson, my primary duties are to figure out how we maintain relations with stakeholders and to improve and develop them.
Unfortunately, I’m currently unable to spend sufficient time on them.
While we have always emphasized investor relations, for increasing our company’s value, I think it's essential to connect with all sorts of people outside of the field of investor relations, so that as many people as possible can understand the value of our company.
Q: “Lastly, please tell us your message toward shareholders and investors.”
A: “Unfortunately, we are presently unable to sufficiently meet the expectations of our shareholders and investors with regard to the speed of commercialization. However, with the guidance of our new management structure, our whole company will work in unison to achieve monetization as soon as possible. Therefore, we ask for the continued support of our shareholders and investors.”
The use of our core services of metabolomics by private enterprises, particularly by interested food companies, has increased. This is due to our quality control and the demonstrable value of our products, which we have achieved over many years of working with the technologies of academic institutions such as universities and laboratories. Therefore, we are contributing to the progress of technologies that are used in the world of industry.
Additionally, with the increased need for development of new healthcare-related businesses aimed at healthy life expectancy and preventive medicine, we are seeing efforts by companies from a wide range of fields outside of foodstuffs and medicine. Therefore, we expect the demand for metabolome analyses to increase steadily.
While aiming to capture domestic demand on the one hand, we are also aiming to open up the European, American, and Chinese markets using our company's unique technologies.
With regard to our biomarker business, we regard monetization as a great challenge, and have created a new exit strategy.
Unfortunately, we are presently unable to sufficiently meet the expectations of our shareholders and investors with regard to the speed of commercialization. However, with the guidance of our new management structure, our whole company will work in unison to achieve monetization as soon as possible. Therefore, we ask for the continued support of our shareholders and investors.
6.Conclusions
The results of the metabolomics business were sluggish, but the company’s advantage in the metabolome analysis market is unchanged, and the interest in the usefulness of metabolomics is growing in a broad range of fields. Accordingly, the metabolomics business can be expected to grow steadily in this term and future. As for the biomarker business, whose monetization is delayed, we, investors, are curious about the outcomes of the exit strategy launched under the new president Hashizume. It may be difficult to ask for short-term results, but we would like to pay attention to quarterly trends and progress
<Reference: Regarding Corporate Governance>
◎Organization type
Organization type | Company with audit and supervisory committee |
Directors | 6 directors, including 3 outside ones |
◎Corporate Governance Report
Updated on November 28, 2018
<Reasons for Non-Compliance with the Principles of the Corporate Governance Code (Excerpts)>
It is mentioned that “Our company follows all of the basic principles of the Corporate Governance Code.”
This report is intended solely for information purposes, and is not intended as a solicitation for investment. The information and opinions contained within this report are made by our company based on data made publicly available, and the information within this report comes from sources that we judge to be reliable. However, we cannot wholly guarantee the accuracy or completeness of the data. This report is not a guarantee of the accuracy, completeness or validity of said information and opinions, nor do we bear any responsibility for the same. All rights pertaining to this report belong to Investment Bridge Co., Ltd., which may change the contents thereof at any time without prior notice. All investment decisions are the responsibility of the individual and should be made only after proper consideration. Copyright(C) 2019 Investment Bridge Co., Ltd. All Rights Reserved. |



