Bridge Report:(6722)A&T the second term of the fiscal year ending December 2020
 Shigetaka Misaka President | A&T Corporation (6722) |
 |
Company Information
Market | JASDAQ |
Industry | Electrical equipment (manufacturing industry) |
President | Shigetaka Misaka |
HQ Address | Yokohama Plaza Bldg. 2-6 Kinko-cho, Kanagawa-ku, Yokohama-shi |
Year-end | December |
Homepage |
Stock Information
Share Price | Shares Outstanding | Total market cap | ROE Act. | Trading Unit | |
¥1,428 | 6,257,900 shares | ¥8,936 million | 9.3% | 100 shares | |
DPS Est. | Dividend yield Est. | EPS Est. | PER Est. | BPS Act. | PBR Act. |
¥24.00 | 1.7% | ¥94.30 | 15.1 x | ¥1,263.99 | 1.1 x |
*The share price is the closing price on August 4, 2020. ROE is based on the previous year.
Each figure is from the financial report for the second term of fiscal year ending December 2020.
Earnings Trend
Fiscal Year | Sales | Operating Income | Ordinary Income | Net Income | EPS | DPS |
December 2016 Act. | 10,234 | 1,015 | 1,004 | 651 | 104.14 | 20.00 |
December 2017 Act. | 10,371 | 773 | 757 | 678 | 108.41 | 20.00 |
December 2018 Act. | 10,430 | 774 | 768 | 518 | 82.80 | 24.00 |
December 2019 Act. | 11,049 | 958 | 943 | 695 | 111.21 | 24.00 |
December 2020 Est. | 10,500 | 815 | 800 | 590 | 94.30 | 24.00 |
* Estimates are those of the Company. Unit: million yen, yen.
This report outlines A&T Corporation’s earnings results for the second term of the fiscal year ending December 2020.
Table of Contents
Key Points
1. Company Overview
2. The Second Term of Fiscal Year ending December 2020 Earnings Results
3. Fiscal Year ending December 2020 Earnings Forecasts
4. Medium-Term Management Plan (FY Dec. 2018 to FY Dec. 2020) Progress
5. Current situation of the laboratory automation system (LAS) business and future activities
6. Conclusions
<Reference:Regarding Corporate Governance>
Key Points
- For the second quarter of the term ending Dec. 2020, sales decreased 5.7% year on year to 4,956 million yen. Due to the spread of the novel coronavirus, revenue declined except for analyzers and the laboratory information system. Gross profit margin improved by 2.4% due to sales of original products the company is focusing on. SG&A expenses dropped due to business trip cancellations and other factors, and operating income was flat at 423 million yen.
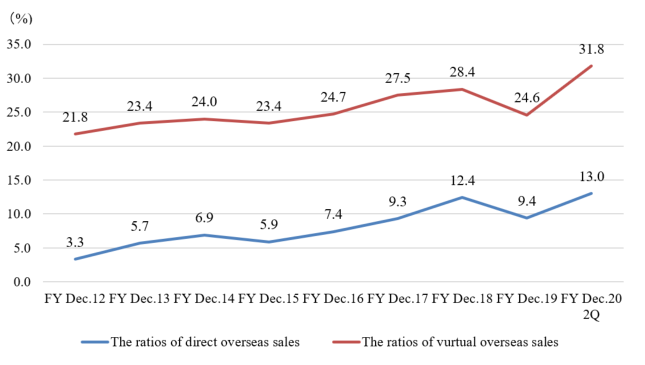
- The ratios of direct and virtual overseas sales, on which the company puts importance for growth, were 13.0% and 31.8%, respectively. Sales to China, where the company has positioned its market development as an important challenge, expanded significantly from 200-million-yen year on year to 394 million yen. The sales ratio also rose from 4.5% in the previous term to 8.8%.
- The full-year earnings forecast for the term ending Dec. 2020 has been revised downward. Sales are expected to decline 5.0% year on year to 10,500 million yen, and operating income to fall 15.0% year on year to 815 million yen. The effects of the novel coronavirus are expected to continue until around September. There is no revision to the dividend forecast, which is estimated at 24 yen/share as in the previous term. The estimated payout ratio is 25.5%.
- Due to the impact of the novel coronavirus, the company could not achieve the plan in the first half, and the full-year forecast was revised downward. Thus, it is difficult to achieve the medium-term management plan. However, one of the basic policies of the medium-term management plan, “raising the ratio of the original product sales,” is making steady progress. In addition, the overseas sales ratio was high at 13.0% for the half-year, maintaining the initial target of the medium-term management plan.
- Moreover, as the novel coronavirus impacts will continue until the beginning of autumn, the environment will remain uncertain in the short term, including how much the number of tests will recover. The company believes that the importance of promptly delivering test data to medical sites is recognized more than ever and that the need for test systems will further increase in the medium term.
- We would like to pay attention to the next medium-term management plan as to what kind of strategy the company will adopt during and after the novel coronavirus pandemic.
1. Company Overview
The core businesses of A&T Corporation are the “blood testing business,” in which the company develops, manufactures, and sells IVD devices, reagents, etc. mainly for electrolyte and glucose tests, and the “IT and automation support business,” which facilitates the streamlining of clinical tests.
The company excels at proposing an optimal one-stop solution for preparing necessary products in a laboratory, installing and operating equipment while proposing a layout, and possesses advanced technologies that are highly evaluated by leading overseas OEM clients.
1-1 Corporate History
In the 1980s, the general chemical manufacturer Tokuyama Corporation (4043, 1st section of Tokyo Stock Exchange) was expanding its business scope from materials to fine chemicals. While taking inventory of various technologies and items, Tokuyama Corporation decided to develop latex (rubber material; one of the chemical products) reagents for testing antigen-antibody reactions.
In the development process, Tokuyama Corporation formed a business tie-up with Analytical Instruments Inc., which develops, manufactures, and sells clinical test equipment and had been leading the industry by releasing such products as fully automatic blood sugar analyzer in 1978, and in Apr. 1988, they founded a joint venture for distributing their products, A&T Corporation. (“A” of Analytical Instruments and “T” of Tokuyama were combined.)
In November 1990, the company established Esashi Factory, which is now the primary production site, in Iwate Prefecture.
In 1994, A&T Corporation underwent an absorption-type merger, integrating the diagnosis system division of Tokuyama Corporation. The period from the 1980s to the 1990s was the growth period of the clinical testing industry, in which many core technologies were developed, and the company expanded its business steadily while taking advantage of that trend.
In Jul. 2003, the company issued over-the-counter shares. It is now listed in the JASDAQ market of Tokyo Stock Exchange.
1-2 Management Philosophy, etc.
A&T Corporation upholds its corporate ethos: “Support medical care and contribute to people’s health around the world,” and aims to improve the quality of medical care and reduce cost, under following three management policies.
1. C.A.C.L. | Commit to research and development of unique products and technologies in all areas’ of “C.A.C.L.” in clinical laboratory testing. |
2.Consistent Framework | Increase market value and reduce the cost of products through an integrated system of development, manufacture, distribution and customer support. |
3.Alliance | Promote market expansion and quality improvement of products with business partners domestic and overseas. |
* C.A.C.L.: Acronym of “Chemicals (diagnostic reagents),” “Analyzers (Analyzers),” “Computers (laboratory information system),” and “Lab-Logistics (laboratory automation system)” in the field of products required for operating a clinical test room
1-3 Market Environment
◎ Market scale
(Domestic and global markets)
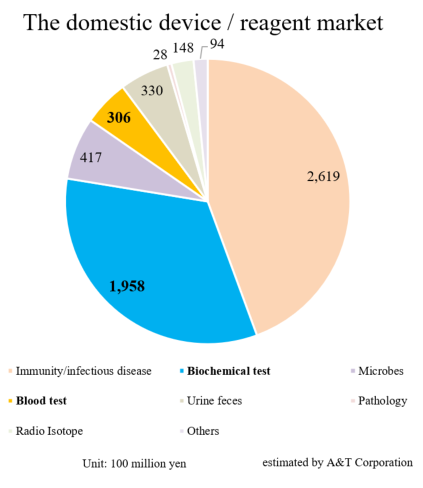
Based on the information in the website of the Japan Association of Clinical Reagents Industries, A&T Corporation estimated that the scale of the Japanese market of related devices and reagents is about 590 billion yen. The market scales of biochemical tests and hematology tests are 195.8 billion yen and 30.6 billion yen, respectively.
(Trend of IVD devices)
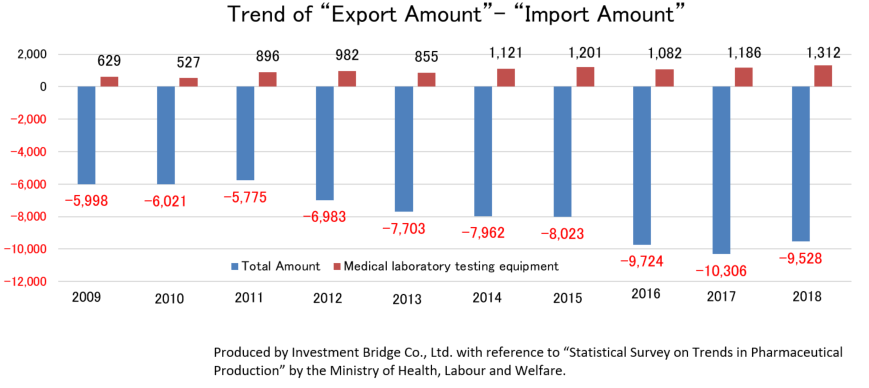
According to “Statistical Survey on Trends in Pharmaceutical Production” by the Ministry of Health, Labour and Welfare, the scale of the Japanese medical products market (domestic production amount) in 2018 was about 1.9 trillion yen. Products for medical treatment is dominant, and medical Analyzers, which is handled by A&T Corporation, has a market scale of about 180 billion yen.
While there is a significant excess of imports of the overall medical product, there is an excess of exports of IVD devices. This indicates how competitive Japanese companies are. Hitachi and Canon Medical Systems (former Toshiba) supply testing equipment to Roche in Switzerland and Abbott in the U.S., respectively. Likewise, A&T Corporation supplies OEM products to Siemens. Namely, testing equipment made in Japan is now indispensable in the global clinical testing field.
| 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 | 2017 | 2018 |
Total amount |
|
|
|
|
|
|
|
|
|
|
Production | 15,762 | 17,134 | 18,085 | 18,952 | 19,055 | 19,895 | 19,456 | 19,146 | 19,904 | 19,490 |
Import | 10,750 | 10,554 | 10,584 | 11,884 | 13,008 | 13,685 | 14,249 | 15,564 | 16,496 | 16,204 |
Export | 4,752 | 4,534 | 4,809 | 4,901 | 5,305 | 5,723 | 6,226 | 5,840 | 6,190 | 6,676 |
Export - Import | -5,998 | -6,021 | -5,775 | -6,983 | -7,703 | -7,962 | -8,023 | -9,724 | -10,306 | -9,528 |
IVD devices |
|
|
|
|
|
|
|
|
|
|
Production | 1,102 | 1,035 | 1,452 | 1,587 | 1,471 | 1,695 | 1,807 | 2,010 | 1,729 | 1,832 |
Import | 125 | 100 | 90 | 122 | 214 | 203 | 226 | 252 | 170 | 209 |
Export | 754 | 628 | 985 | 1,104 | 1,069 | 1,324 | 1,427 | 1,334 | 1,356 | 1,521 |
Export - Import | +629 | +527 | +896 | +982 | +855 | +1,121 | +1,201 | +1,082 | +1,186 | +1,312 |
*Unit: 100 million yen.
◎Companies in the same field
Code | Corporate name | Sales | Sales growth rate | Operating income | Profit growth rate | Operating income margin | ROE | Market cap | PER | PBR |
4549 | Eiken Chemical | - | - | - | - | - | 10.3 | 78,635 | - | 1.8 |
6678 | Techno Medica | 9,200 | -6.2 | 1,000 | -20.5 | 10.9% | 7.2 | 15,636 | 21.6 | 1.1 |
6722 | A&T | 10,500 | -5.0 | 815 | -15.0 | 7.8% | 9.3 | 8,936 | 15.1 | 1.1 |
6869 | Sysmex | - | - | - | - | - | 12.9 | 1,727,494 | - | 6.2 |
6951 | JEOL | - | - | - | - | - | 12.4 | 154,634 | - | 3.4 |
*The results for this term were forecasted by the company. The units are million yen, %, and times. Share price-related indices are based on the closing prices on August 4, 2020.
* Eiken Chemical, Sysmex and JEOL have not published their estimates.
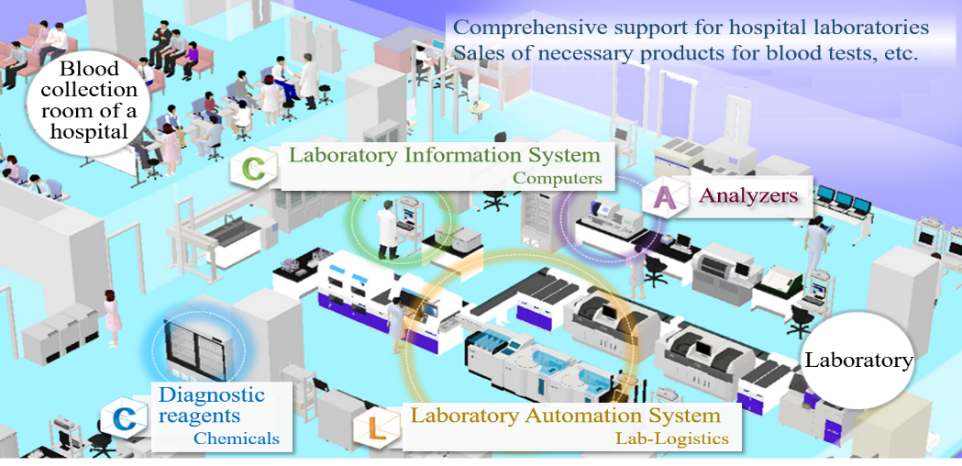
1-4 Business Description
In addition to the development, manufacturing, and sale of products, including testing devices and reagents used in the clinical testing rooms of hospitals, A&T Corporation offers customer support. The company also offers comprehensive consulting services, including the proposal for the layout of a laboratory, installation and operation.
(Source: the company)
What is clinical testing?
Clinical tests can be classified into “biopsies” for directly examining the body with medical equipment, such as X-ray equipment, CT, MRI, electrocardiographic and ultrasonic equipment, and “laboratory tests” for examining biological samples (specimens), such as blood, urine, stool, and cells, collected from patients.
A&T Corporation handles products used for laboratory testing, especially blood tests.
There are a variety of blood tests conducted at hospitals and in comprehensive medical checkups, including the tests of the hepatic system, the renal system, uric acid, the lipid system, glucose metabolism, blood cells, and infectious diseases. A&T Corporation mainly conducts business related to “electrolyte tests” and “glucose tests.”
“Electrolyte tests”
The water content constitutes about 60% of the human body, as body fluids, including intracellular fluid and blood plasma. Body fluids are classified into electrolytes, which are mineral ions that dissolve in water and conduct electricity (such as sodium, potassium, calcium, and chlorine), and non-electrolytes, which dissolve in water, but do not conduct electricity (such as glucose and urea).
Each electrolyte takes important roles for keeping human beings alive while maintaining a healthy balance - “sodium” adjusts the water content of the body, “potassium” controls muscles and nerves, “calcium” forms bones and teeth, conveys nervous stimuli, and coagulates blood, and “chlorine” supplies oxygen to the inside of the body. If the concentration of electrolytes in blood is abnormal, there is a possibility that the kidneys or hormones are malfunctioning.
The purpose of electrolyte tests is to measure the concentration of each electrolyte ion in body fluid, detect the disruption of a balance, and then diagnose a disorder in the body. Sampled blood and urine are examined with testing device.
*Major diseases
Sodium | Diabetic coma, dehydration, acute nephritis, chronic renal failure, nephrotic syndrome, heart failure, hypothyroidism, Addison disease, diabetic acidosis, etc. |
Potassium | Acute renal failure, chronic renal failure, respiratory insufficiency syndrome, etc. |
Calcium | Malignant tumor, multiple myeloma, hyperparathyroidism, renal failure, hypoparathyroidism, vitamin D deficiency, etc. |
Chlorine | Dehydration, renal failure, chronic nephritis, emphysema, etc. |
“Glucose tests”
The sugar in blood plasma (blood sugar) is composed mostly of glucose, which is the only energy source for the central nervous system, including the cerebrum. When the stomach is empty (over 5 hours after eating), the liver emits about 8 grams of glucose per hour, and the brain consumes about half of them, and muscles and red blood cells consume one fourth of them, respectively.
Blood sugar level in its normal condition is strictly controlled while keeping a balance between the increase through the absorption from the intestine and the generation in the liver and the decrease through the consumption in the muscles. If this control does not work properly, hyperglycemia or hypoglycemia will occur.
A glucose test is conducted for measuring the concentration of glucose in blood or urine.
*Major diseases
Hyperglycemia | Diabetes (insulin, which is a hormone secreted from the pancreas, does not work, and so cells cannot use glucose in blood), pancreatitis, thyroid disease, postgastrectomy dumping syndrome, etc. |
Hypoglycemia | Liver damage, hypopituitarism, adrenal hypofunction, etc. |
1.Business Field
The business of A&T Corporation is composed of the “blood testing business,” in which the company develops, manufactures, and sells clinical testing devices, reagents, supplies, etc. for blood tests, and the “IT and automation support business,” which facilitates the streamlining of manual work in hospital laboratories with IT and automated systems. The company comprehensively supports hospital laboratories.
(Since this company conducts this business only, neither its brief financial reports nor securities reports contain segment information. It should be noted that the company discloses the sales of each product series in reference materials for briefing results, etc., but not the sales of each type of business.)
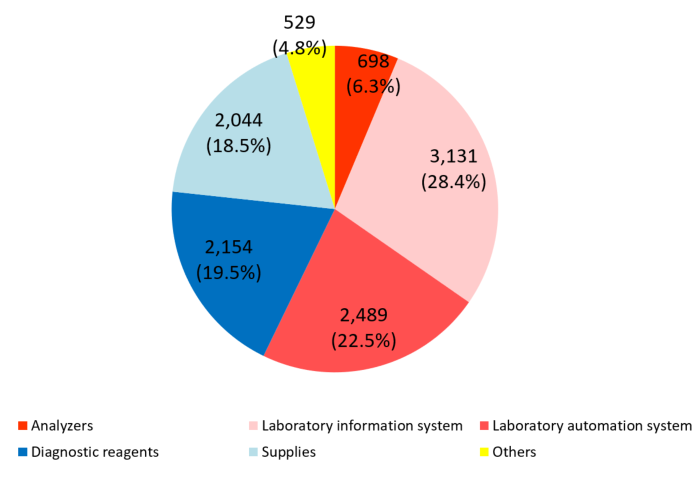
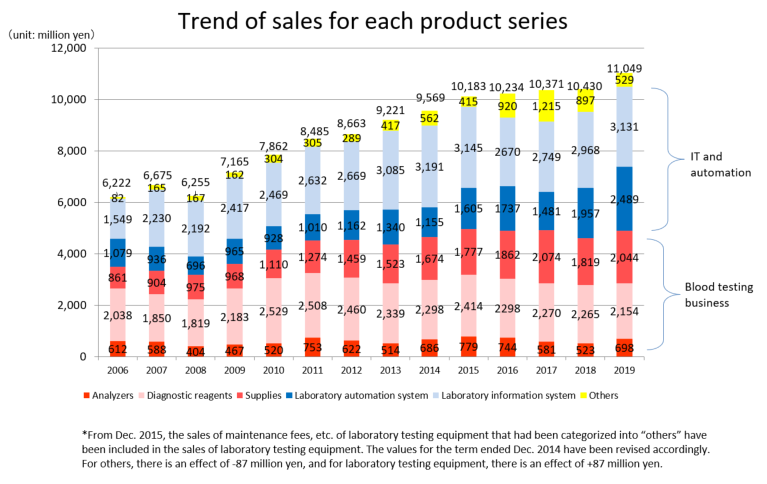
Product series | Results for FY Dec.19 | Ratio to total sales |
Clinical testing devices and systems | 6,320 | 57.2% |
Analyzers | 698 | 6.3% |
Laboratory information system | 3,131 | 28.4% |
Laboratory automation system | 2,489 | 22.5% |
Diagnostic reagents | 2,154 | 19.5% |
Supplies | 2,044 | 18.5% |
Others | 529 | 4.8% |
Total | 11,049 | 100.0% |
The red letters denote the “Blood testing business” (analyzers, diagnostic reagents, and supplies), while the blue letters denote the “IT and automation support business” (Laboratory Information System, Laboratory Automation System). The sales distribution ratio for the term ended December 2019 was 44.3% for the blood testing business, and 55.7% for the IT and automation support business.
1) Blood testing business
Outline
The company works on developing, manufacturing, and selling Analyzers for clinical tests such as “electrolyte tests” and “glucose tests,” reagents for clinical tests (for measuring the concentrations of electrolytes, blood sugar, etc.), and supplies (such as sensors installed in analyzer), and offers customer support.
Electrolyte analyzer
 | Glucose analyzer
 |
(Source: A&T’s website)
Commercial distribution
*Inside Japan
The company directly sells analyzers, reagents, and supplies to small and medium-sized hospitals via 8 branches nationwide. As of now, about 4,300 units of equipment are in operation.
*Outside Japan
The company sells analyzers as an OEM. It supplies electrolyte units, which are the specialty products of the company, to other Japanese and overseas manufacturers, including JEOL (6951, 1st section of TSE). The OEM clients combine the unit with large-size clinical chemistry analyzers and sell them. As an OEM, JEOL supplies products to Siemens, which is one of global enterprises handling large-size clinical chemistry analyzers.
Business model
Once Analyzers is newly installed, clinical reagents and supplies will be continuously delivered, and the maintenance service for the equipment will be offered.
Once adopted, it is rare for client hospitals to change manufacturers considering the continuity of test data and usability, and so it is difficult for new manufacturers to enter the market. 7 to 10 years later, upgraded models will replace them. This characterizes this business field.
Major enterprises in this field
Sysmex (6869, 1st section of TSE), Hitachi High-Tech (unlisted), JEOL (6951, 1st section of TSE), Fuji Film Wako Pure Chemical (unlisted), ARKRAY (unlisted)
2) IT and automation support business
Outline
In the case of blood tests, it is necessary to convey patient’s blood (specimen) sampled in a blood collection room to a clinical laboratory and manually set the specimen at testing equipment.
As several kinds of tests need to be conducted for many specimens at the same time, this work is extremely labor-intensive and inefficient, and the human error of taking a wrong specimen is difficult to avoid.
In these circumstances, A&T Corporation supports the streamlining of the testing process with the following 2 systems.

(Source: A&T’s website)
Laboratory Information System (LIS) | Software for a clinical laboratory, which receives requests for tests from medical doctors, sends a command for testing to Analyzers, and inputs test results in electronic medical charts, etc. accurately and swiftly. This also manages cost, etc. and serves as a core system of a laboratory. |
Laboratory Automation System (LAS) | The specimens delivered to a laboratory are automatically conveyed to Analyzers via a computer-controlled conveyor line, and then undergo measurement. Blood tests, which had been manually conducted, are fully automated, to streamline and speed up the testing process. |
It is expected that the installation of LAS will decrease the necessary number of workers from 7-8 to about 2, and the necessary time of testing from 90 min. to 30-40 min.
Through the introduction of LIS, it became possible to put together the data of test results, which had been printed out for each test item, and give feedback to medical doctors swiftly and accurately. In addition, the data mining function is helpful for reducing the number of times of abnormal value retesting and the duration of testing.
Commercial distribution
*Inside Japan
Targeting the laboratories of medium and large-sized hospitals, the sales division of A&T Corporation sells LIS in cooperation with hospital information system manufacturers, including Hitachi, IBM, and Fujitsu, and LAS in cooperation with large-size clinical chemistry and immunoassay analyzer manufacturers, including Hitachi, Canon Medical Systems, and JEOL, as comprehensive solutions*。
*For the details of comprehensive solutions, see the section “1-5 Characteristics and Strengths.”
*Outside Japan
Previously, the company has been selling LAS directly in Korea, China, etc., but in China it has started OEM supply. In the United States, OEM sales of blood aliquoting modules, which are the main components in LAS, are made to partner companies.
Business model
In addition to the maintenance service of LIS and LAS after their installation, the company can connect additional systems, customize the system, and so on for LIS, and can offer maintenance services, sell supplies, and so on for LAS. For both of the systems, stable sales can be expected.
Like Analyzers, clients are rarely motivated to shift to other manufacturer’s equipment, considering usability, data continuity, etc. The price range per transaction is 10 to 90 million yen for LIS, and 10 to 200 million yen for LAS.
Major enterprises in this field
LIS: Sysmex CNA (subsidiary of Sysmex), local vendors, etc.
LAS: IDS (unlisted), Hitachi, Ltd., Siemens, etc.
2.Development systems
The company established development groups by product types in order to apply elementary technologies cultivated over many years to a wide range of product development. In addition, it places a chief technology officer for each field including electricity, machinery, and chemistry and promotes product development through management of a matrix organization structure.
90 staff members or more are employed at the headquarters and Shonan Office.
The research and development cost for the term ended December 2019 was 987 million yen. It will continue to actively promote research and development with a target sales of around 10%.
3.Production systems
There are two production sites: Shonan Factory, in Kanagawa Prefecture, for manufacturing some clinical reagents and supplies and Esashi Factory, in Iwate Prefecture, for producing equipment, Laboratory Automation System (LAS) and some clinical reagents.
The company manufactures high-quality, safe products with advanced equipment under rigorous management. In cooperation with the development section, the company is striving to improve quality and streamline operation.
In order to develop the foundation for expanding sales further, the company constructed a new building with a total floor area of 7,300 m2 at Esashi Factory by investing 1.7 billion yen in August 2017.The Company strengthens capabilities considerably through this construction.
4.Sales routes and methods
As mentioned above, A&T Corporation sells its products to client hospitals via 8 branches in Japan, by utilizing its capability of proposing comprehensive solutions.
Outside Japan, the company supplies products to overseas clients and dealers including Siemens through domestic OEM partners such as JEOL.
To expand its business scale by supplying products as an OEM like this is the basic strategy, and the company concentrates on the diversification of OEM clients.
The direct overseas sales ratio that the company sells directly to overseas clients and dealers for the term ended December 2019 were 9.4.%.
But, as the ratio of virtual overseas sales, including the (estimated) overseas sales via domestic OEM clients, for the term ended Dec. 2015, Dec. 2016, Dec. 2017, and Dec. 2018 were 23.4%, 24.7%, 27.5%, 28.4% respectively, the ratio of overseas sales is in upward momentum.
Especially in the term ended Dec. 2018, sales to China soared to 780 million yen, more than doubled from 351 million yen in the previous fiscal year. In the term ended Dec. 2019, the performance was sluggish at 532 million yen (down 31.8% year on year) due to the changes in the Chinese financial environment, but the company will keep cultivating the Chinese market, with the aim of achieving a ratio of virtual overseas sales of over 50% in the future.
(The ratio of overseas sales, unit: %)
| FY Dec.12 | FY Dec.13 | FY Dec.14 | FY Dec.15 | FY Dec.16 | FY Dec.17 | FY Dec.18 | FY Dec.19 |
Direct | 3.3 | 5.7 | 6.9 | 5.9 | 7.4 | 9.3 | 12.4 | 9.4 |
Virtual | 21.8 | 23.4 | 24.0 | 23.4 | 24.7 | 27.5 | 28.4 | 24.6 |
1-5 Characteristics and strengths
◎Capability of proposing comprehensive solutions
A&T Corporation handles products mainly for electrolyte and glucose tests and does not handle large products for analyzing other tests. However, client hospitals need to install a variety of testing instruments in their clinical laboratories.
To meet their needs, the Laboratory Automation System (LAS) has an automatic conveyor line that is compatible with not only its own products, but also other manufacturers’ instruments.
There are few manufacturers that possess technologies for producing systems for connecting their own products and other manufacturers’ products freely and conveying them. Accordingly, the company occupies about 30% of the Japanese market.
The sales staff of the company not only delivers equipment, but also proposes a layout for the most efficient testing with 3D CAD or the like, while considering the area and shape of a laboratory.
All above, the company can offer optimal one-stop solutions for preparing necessary products in a laboratory, installing and operating equipment while proposing a layout. This is highly evaluated by client hospitals.
◎Advanced technologies in specific fields
A&T Corporation handles products mainly for “electrolyte tests” and “glucose tests.” Especially, its advanced technology for electrolyte analyzers can be verified by the fact that its products are supplied to JEOL, which is a leading manufacturer of measurement devices, including medical instruments, and Siemens, which is a large global company.
As mentioned in the section of the market environment, Japanese medical Analyzers is highly competent in the world, and A&T Corporation contributes to the competitiveness of Japanese products.
1-6 ROE analysis
| FY Dec.12 | FY Dec.13 | FY Dec.14 | FY Dec.15 | FY Dec.16 | FY Dec.17 | FY Dec.18 | FY Dec.19 |
ROE (%) | 12.2 | 10.7 | 9.5 | 15.7 | 10.9 | 10.4 | 7.4 | 9.3 |
Ratio of net income to sales [%] | 5.60 | 5.11 | 4.76 | 8.28 |
6.37 |
6.54 |
4.97 |
6.30 |
Total asset turnover ratio [times] | 1.02 | 0.98 | 1.00 | 1.04 |
1.03 |
0.92 |
0.84 |
0.90 |
Leverage [times] | 2.14 | 2.13 | 1.99 | 1.83 | 1.67 | 1.73 | 1.79 | 1.64 |
Since there was no longer an extraordinary loss, which was posted in the term ended Dec. 2018, the ROE in the term ended Dec. 2019 exceeded 8%, which is said to be a common goal for Japanese enterprises suggested by Ito Review, etc. For the term ending Dec. 2020, the ratio of net income to sales is estimated to be 5.6%.
2. The Second Term of Fiscal Year ending December 2020 Earnings Results
(1) Business Results
| FY 12/ 19 2Q | Ratio to sales | FY 12/ 20 2Q | Ratio to sales | YoY change | Change against estimates |
Sales | 5,255 | 100.0% | 4,956 | 100.0% | -5.7% | -8.5% |
Gross profit | 2,262 | 43.0% | 2,252 | 45.4% | -0.4% | - |
SG&A | 1,840 | 35.0% | 1,828 | 36.9% | -0.6% | - |
Operating Income | 421 | 8.0% | 423 | 8.5% | +0.5% | -11.7% |
Ordinary Income | 411 | 7.8% | 417 | 8.4% | +1.4% | -11.2% |
Quarterly Net Income | 310 | 5.9% | 306 | 6.2% | -1.2% | -17.1% |
*Unit: million yen
Sales declined and profits increased slightly. Lower than planned.
For the second quarter of the term ending Dec. 2020, sales decreased 5.7% year on year to 4,956 million yen. Due to the spread of the novel coronavirus, revenue declined except for analyzers and the laboratory information system. Gross profit margin improved by 2.4% due to sales of original products the company is focusing on. SG&A expenses dropped due to business trip cancellations and other factors, and operating income was flat at 423 million yen. The ratios of direct and virtual overseas sales, on which the company puts importance for growth, were 13.0% and 31.8%, respectively. Sales to China, where the company has positioned its market development as an important challenge, expanded significantly from 200-million-yen year on year to 394 million yen. The sales ratio also rose from 4.5% in the previous term to 8.8%.
(2) Sales of each product series
Product series | FY 12/19 2Q | Composition Ratio | FY 12/ 20 2Q | Composition Ratio | YoY |
Clinical Testing devices and Systems | 2,751 | 52.4% | 2,706 | 54.7% | -1.6% |
Analyzers | 330 | 6.3% | 405 | 8.2% | +22.5% |
Laboratory Information System (LIS) | 1,461 | 27.9% | 1,515 | 30.6% | +3.7% |
Laboratory Automation System (LAS) | 959 | 18.2% | 785 | 15.9% | -18.1% |
Diagnostic reagents | 1,074 | 20.4% | 1,018 | 20.5% | -5.2% |
Supplies | 1,060 | 20.2% | 1,035 | 20.9% | -2.4% |
Others | 369 | 7.0% | 195 | 3.9% | -47.0% |
Total | 5,255 | 100.0% | 4,956 | 100.0% | -5.7% |
*Unit: million yen.
◎Clinical Testing devices and Systems
Sales decreased.
Direct sales of glucose analyzers decreased while sales of electrolyte analyzers to OEM customers increased, leading to a rise in the sales of analyzers.
Sales of the laboratory information systems were also augmented in the first half due to strong new and renewal demand in the first quarter (January-March).
Sales of laboratory automation systems improved due to increased sales to Chinese OEM customers and new projects in Japan and South Korea in the first quarter (January-March). Nonetheless, as responding to the novel coronavirus was the customers’ top priority, the operation period of some projects was postponed, and the sales of large-scale projects during the same period of the previous year (April-June 2019) were more concentrated than usual. Hence, sales decreased.
◎Diagnostic reagents
Sales decreased.
In the second quarter (April-June), sales of reagents for glucose analyzers and electrolyte analyzers declined because of the decrease in the number of laboratory tests due to restricting outpatient visits to hospitals and cancelling or postponing medical examinations.
◎Supplies
Sales decreased.
While sales of sensors for electrolyte analyzers to existing OEMs increased slightly, sales of supplies for laboratory automation systems decreased.
◎Others
Sales decreased.
Following the basic policy of increasing the ratio of original product sales to improve profitability, sales of other manufacturers’ products associated with the laboratory information system and laboratory automation system projects decreased.
(3) Financial standing and cash flows
Main Balance Sheet
| End of December 2019 | End of June 2020 |
| End of December 2019 | End of June 2020 |
Current Assets | 7,663 | 7,156 | Current liabilities | 3,643 | 3,093 |
Cash | 1,304 | 2,455 | Payables | 805 | 563 |
Receivables | 4,286 | 2,770 | ST Interest Bearing Liabilities | 1,700 | 1,700 |
Inventories | 1,994 | 1,853 | Noncurrent liabilities | 473 | 321 |
Noncurrent Assets | 4,218 | 4,166 | LT Interest Bearing Liabilities | 450 | 300 |
Tangible Assets | 3,642 | 3,601 | Total Liabilities | 4,117 | 3,414 |
Intangible Assets | 36 | 36 | Net Assets | 7,764 | 7,908 |
Investment, Others | 539 | 528 | Total liabilities and net assets | 11,881 | 11,322 |
Total assets | 11,881 | 11,322 | Total Interest-bearing Liabilities | 2,150 | 2,000 |
|
|
| Capital Adequacy Ratio | 65.3% | 69.8% |
*Unit: million yen
Current assets decreased 506 million yen from the end of the previous term due to a drop in trade receivables. Noncurrent assets declined 52 million yen from the end of the previous term. Total assets fell 558 million yen from the end of the previous term to 11,322 million yen.
Total liabilities decreased 702 million yen from the end of the previous term to 3,414 million yen due to a decline in payables and repaying interest-bearing liabilities.
Net assets increased 143 million yen from the end of the previous term to 7,908 million yen due to an increase in retained earnings etc.
As a result, the equity ratio increased 4.5 points from 65.3% at the end of the previous term to 69.8%.
◎Cash Flow
| FY12/19 2Q | FY12/20 2Q | Change |
Operating Cash Flow | 876 | 1,511 | +635 |
Investing Cash Flow | -56 | -60 | -3 |
Free Cash Flow | 819 | 1,451 | +631 |
Financing Cash Flow | -500 | -300 | +200 |
Term End Cash and Equivalents | 1,371 | 2,455 | +1,084 |
*Unit: million yen
The surplus of operating CF and free CF expanded due to a decrease in inventories. The cash position has risen.
3. Fiscal Year ending December 2020 Earnings Forecasts
(1) Consolidated earnings forecasts
| FY Dec. 19 | Composition ratio | FY Dec. 20 Est. | Composition ratio | YoY | Correction ratio |
Sales | 11,049 | 100.0% | 10,500 | 100.0% | -5.0% | -7.9% |
Gross profit | 4,735 | 42.9% | 4,500 | 42.9% | -5.0% | -9.1% |
SG&A | 3,777 | 34.2% | 3,685 | 35.1% | -2.4% | -6.2% |
Operating Income | 958 | 8.7% | 815 | 7.8% | -15.0% | -20.1% |
Ordinary Income | 943 | 8.5% | 800 | 7.6% | -15.2% | -20.0% |
Net Income | 695 | 6.3% | 590 | 5.6% | -15.2% | -24.4% |
*The forecasted values were provided by the company. Unit: million yen
Earnings forecast revised. Sales and profit decline.
The full-year earnings forecast for the term ending Dec. 2020 has been revised downward. Sales are expected to decline 5.0% year on year to 10,500 million yen, and operating income to fall 15.0% year on year to 815 million yen. The effects of the novel coronavirus are expected to continue until around September. There is no revision to the dividend forecast, which is estimated at 24 yen/share as in the previous term. The estimated payout ratio is 25.5%.
(2) Sales for each product series
Product series | FY Dec. 19 | Composition ratio | FY Dec. 20 Est. | Composition ratio | YoY | Correction ratio |
Clinical Testing devices and Systems | 6,320 | 57.2% | 5,955 | 56.7% | -5.8% | -9.5% |
Analyzers | 698 | 6.3% | 750 | 7.1% | +7.3% | -1.3% |
Laboratory Information System (LIS) | 3,131 | 28.4% | 3,095 | 29.5% | -1.2% | -12.6% |
Laboratory Automation System (LAS) | 2,489 | 22.5% | 2,110 | 20.1% | -15.3% | -7.5% |
Diagnostic reagents | 2,154 | 19.5% | 2,000 | 19.0% | -7.2% | -7.4% |
Supplies | 2,044 | 18.5% | 2,045 | 19.5% | +0.0% | -5.3% |
Others | 529 | 4.8% | 500 | 4.8% | -5.6% | 0.0% |
Total | 11,049 | 100.0% | 10,500 | 100.0% | -5.0% | -7.9% |
*Unit: million yen
*Laboratory information system and Laboratory automation system
The operations of some projects have been postponed to the next term or later, and the operation period of some projects are in the process of readjustment. Thus, the sales of the laboratory information system are expected to decrease instead of increasing, as was predicted in the previous forecast. The laboratory automation system is forecasted to witness a further decrease in sales. The company will continue to focus on customer follow-up by adjusting the operation period and so on.
*Diagnostic reagents
Sales are expected to decline instead of increasing, which was predicted in the previous forecast because of the decrease in the number of laboratory tests due to limiting outpatient visits and postponement and discontinuation of medical examinations. Demand will continue to decrease until September 2020, but recovery is expected from October 2020.
*Others
The company will continue to reduce the sales of other manufacturers’ products and focus on the sales of its original products according to the medium-term management plan.
(3) Business measures in response to the novel coronavirus crisis
The company will continue to carry out production activities and will maintain product sales and customer support while establishing strict coronavirus prevention measures.
Regarding sales activities, the company has been limiting the activities since April, by reducing advertising etc. However, it will resume sales activities while giving top priority to the safety of customers, business partners, and employees.
Also, although the Crisis Response Headquarters was dissolved, to prevent the spread of the novel coronavirus infections and improve work efficiency, the company will actively employ telework, flexible working hours, online meetings, etc.
4. Medium-Term Management Plan (FY Dec. 2018 to FY Dec. 2020) Progress
(1) Recognition of the business environment
<Business environment analysis>
| Blood testing business | IT and automation support business |
Business environment | *Due to the change in the sales environment, the sales to some OEM clients may decline. *The domestic testing markets for electrolytes, glucose, etc. will reach a plateau. *The growth of the overseas market, especially China, is remarkable. | *The scale and competition of the domestic market have not changed significantly and are on a plateau. Overseas demand is high. *The amount per order is large, but the period until the next update is as long as 5 to 10 years. *Once a product is installed, it tends to be adopted again at the time of the next update (relatively easy to defend). On the other hand, it is relatively difficult to reel in customers from competitors (difficult to attack). *Recently, the overseas demand for LAS has been strong. |
In addition to the basic recognition of the business environment above, responding to the novel coronavirus (customers/in-house) has become an important management issue.
*There is a shift toward an uncertain environment in the clinical testing market in Japan and overseas.
*In Japan, visits to hospitals are decreasing. Online medical treatment has been launched. Blood tests in hospitals are expected to decrease.
*Tests for minor illnesses and medical examinations at regional central hospitals are on a downward trend.
(2) Important objectives
1 | To deal with the fact that sales are concentrated on specific OEM clients |
2 | To compensate for the decline in sales to some OEM clients, and find new clients |
3 | To increase gross profit (increasing the sales of original products) |
4 | To reduce the cost for securing product quality |
5 | To put the business in the Chinese market, which is growing rapidly, on track as soon as possible |
6 | To reform ways of working and train personnel |
(3) Progress of Basic Policy
Basic Policy | Situation |
To increase the ratio of sales of original products, and improve profitability | Other sales (other manufacturers’ products) are expected to decrease 500 million yen in the current term. Gross profit is expected to decline 200 million yen due to the impact of the novel coronavirus. However, the company will continue to focus on selling its original products. |
To enhance business operation in China, and boost the ratio of overseas sales | Sales of laboratory automation system for China were sluggish due to the impact of the novel coronavirus, but the company will continue to strengthen business in China. The company is considering the incorporation of the office of expatriate employees in China. |
To cement the cooperation between development and manufacturing sections, and establish systems for developing and producing high-quality products stably | The company has reduced the rate of defective products and improved quality by introducing improved products of major sensors. |
To reform ways of working and train personnel thoroughly | Full-scale introduction of telework mainly at Yokohama headquarters, was accomplished. This will be continued after the novel coronavirus pandemic has subsided. The company will establish a consistent company-wide training program. |
(4) Numerical goals and progress status
| FY 12/ 18 | FY 12/ 19 | FY 12/ 20 | ||||
Plan | Act. | Plan | Act. | Plan | Initial estimates | Revised estimates | |
Sales | 10,500 | 10,430 | 11,200 | 11,049 | 12,000 | 11,400 | 10,500 |
Ordinary Income | 800 | 768 | 1,000 | 943 | 1,250 | 1,000 | 800 |
Ordinary margin to sales | 7.6% | 7.4% | 8.9% | 8.5% | 10.4% | 8.8% | 7.6% |
Direct sales rate in overseas | - | 12.2% | - | 9.43% | Over 10% | Over 10% | Over 10% |
*Unit: million yen
*FY December 2018
Although the number of clinical testing devices and systems increased, OEM sales of sensors remained sluggish due to the response to overseas regulations and holding-off buying from price revision. Ordinary income did not reach the target due to a decrease in sales of sensors and initial costs for new products exceeding the plan. However, the ratio of overseas sales exceeded 10% for the first time with strong Chinese OEMs sales for the laboratory automation system. As a result, it reached the target a year ahead of the plan.
*FY December 2019
Following the basic policy, the sales of original products increased, but could not make up for the decline in sales in China and the sales to some OEM clients. Accordingly, overseas sales were sluggish. The company failed to achieve the target ordinary income, due to the increase of recruitment mid-career engineers and the augmentation of personnel expenses.
*FY December 2020
Due to the impact of the novel coronavirus, sales and profit are expected to decrease instead of the previous forecast that predicted its increase. The company will focus on achieving the revised forecast.
As for the overseas direct sales ratio, the target of 10% or more is maintained.
5. Current situation of the laboratory automation system (LAS) business and future activities
The market scale of the laboratory automation system is 10 billion yen in Japan, and as huge as 770 billion yen outside Japan (including some sales of analytical equipment). Especially, in the Chinese market, it is estimated that the scale will grow at an annual rate of 20%.
In the Chinese market, the company took a lead with “the close strategy,” which is to produce equipment, feeding systems, and reagents supplied to laboratories by major companies in Europe and the U.S., such as Beckman Coulter, Roche, and Abbott, all in house.
As the market is rapidly growing, many demand a system with a higher degree of freedom than the conventional close strategy, and the “open strategy,” in which not only equipment produced in house, but also equipment produced by other companies are adopted, is recognized increasingly.
However, the company intends to pursue overseas markets by implementing a sales strategy that takes into account the characteristics of each country’s market while keeping the “open strategy” as its foundation.
*Domestic market
Under the open strategy, the salesperson plays a consultant role, listens to the requests for testing flow line and a connecting device for each hospital, and proposes the optimum system configuration.
The company sells them by themselves. Target customers are large and medium-sized hospitals.
The main products are CLINILOG V4, CLINILOG STraS, MPAM+, etc.
*South Korean market
Like in Japan, under the open strategy, the salesperson plays a consultant role, listens to the requests for testing flow line and connecting device for each hospital, and proposes the optimum system configuration.
Agencies conduct sales activities. Target customers are large-sized hospitals.
The main products are CLINILOG V4, CLINILOG STraS, etc.
*Chinese market
Uses “semi-open strategy,” which is selling basic package system with certain restrictions such as, connecting other manufacturers’ product is possible, but the number of connections is limited and bending is not an option. Using this strategy, the company conducts OEM sales to RUNDA, the company’s alliance partner.
Target customers are large and medium-sized hospitals.
The main product is the CLINILOG V4 OEM device, “CLINILOG RD.” The company has prepared several basic installation patterns for V4.
*Western markets
Module sales are conducted by OEM to US partners.
The target customers are clinical test rooms all over the world except Japan, and the main product is the MPAM+ OEM device “P540.”
6. Conclusions
Due to the impact of the novel coronavirus, the company could not achieve the plan in the first half, and the full-year forecast was revised downward. Thus, it is difficult to achieve the medium-term management plan. However, one of the basic policies of the medium-term management plan, “raising the ratio of the original product sales,” is making steady progress. In addition, the overseas sales ratio was high at 13.0% for the half-year, maintaining the initial target of the medium-term management plan.
Moreover, as the novel coronavirus impacts will continue until the beginning of autumn, the environment will remain uncertain in the short term, including how much the number of tests will recover. The company believes that the importance of promptly delivering test data to medical sites is recognized more than ever and that the need for test systems will further increase in the medium term.
We would like to pay attention to the next medium-term management plan as to what kind of strategy the company will adopt during and after the novel coronavirus pandemic.
<Reference: Regarding Corporate Governance >
Organization type and the composition of directors and auditors
Organization type | Company with audit and supervisory committee |
Directors | 11 directors, including 2 external ones |
Corporate Governance Report
Last update date: March 31, 2020
<Basic policy>
As a top priority, our company aims to secure the effectiveness of corporate governance and actualize fair business administration by putting importance on the transparency, fairness, and speed of decision making and business execution. In addition, we adopted the system of audit and supervisory committee in order to separate the supervision and execution of business administration, actualize highly transparent management, and streamline the decision-making process of the board of directors.
<Reasons for Non-compliance with the Principles of the Corporate Governance Code (Excerpts)>
The company fully follows the 5 items of basic principles of the corporate governance code.
This report is intended solely for information purposes and is not intended as a solicitation for investment. The information and opinions contained within this report are made by our company based on data made publicly available, and the information within this report comes from sources that we judge to be reliable. However, we cannot wholly guarantee the accuracy or completeness of the data. This report is not a guarantee of the accuracy, completeness or validity of said information and opinions, nor do we bear any responsibility for the same. All rights pertaining to this report belong to Investment Bridge Co., Ltd., which may change the contents thereof at any time without prior notice. All investment decisions are the responsibility of the individual and should be made only after proper consideration. Copyright (C) 2020 Investment Bridge Co., Ltd. All Rights Reserved. |
To view back numbers of Bridge Reports on A&T Corporation (6722) and other companies and to see IR related seminars of Bridge Salon, please go to our website at the following url: www.bridge-salon.jp/


