| CellSeed (7776) |
|
||||||||
Company |
CellSeed Inc. |
||
Code No. |
7776 |
||
Exchange |
JASDAQ |
||
Industry |
Precision Instrument (Manufacturing) |
||
President |
Setsuko Hashimoto, Ph.D. |
||
HQ Address |
Haramachi 3-61, Shinjuku-ku, Tokyo |
||
Year-end |
December |
||
URL |
|||
* Stock price as of closing on 2015/8/27. Number of shares at the end of the most recent quarter excluding treasury shares.
|
||||||||||||||||||||||||
|
|
We present this Bridge Report along with analysis of the first half of fiscal year December 2015 earnings results for CellSeed Inc.
|
| Key Points |
 |
| Company Overview |
|
<Cell-Sheet Engineering and Cell-Sheet Regenerative Medicine>
"Cell-sheet engineering" is the general-purpose platform technology for regenerative medicine. Cells are taken from patients to be cultivated into sheet form for use in various treatments. However, the most revolutionary aspect of "cell-sheet engineering" is the ability to use temperature-responsive cell cultureware equipment to collect undamaged cells, which had not been possible previously. Cells are normally cultivated in culture dishes (To increase the number of cells) and proliferated while adhering to the surface of culture dishes. When cells are collected, they are stripped from the dish using trypsin and other hydrolase proteins, which leads to damage of the protein surface of the cells (extracellular matrix) and causes cells to deform (cell damage). Therefore, these damaged cells cannot be grafted or functioned properly. On the other hand, temperature-responsive cell cultureware can be used to naturally collect cells from the surface of the culture dish by altering the temperature to change cell adhesion characteristics, allowing for the collection of cell sheets that are close to their natural condition with cell protein characteristics remaining undamaged (Possible to create tissue and organs that are close to their natural conditions). In addition to filling the space between cells, cell surface proteins (extracellular matrix) also play a structural role and provide a foothold for cells to bond and control cell proliferation and differentiation. This is a critical substance to allow cells to function as cells, and contribute to the repair (Regeneration) of the affected area.
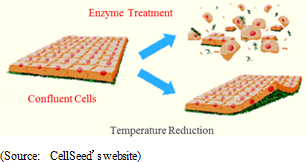 By reducing the cultivation temperature, the characteristics of temperature-responsive polymers are changed to allow cell sheet detachment without damaging the cell surface protein (Extracellular matrix). Traditionally, trypsin and other protein type hydrolytic enzyme procedures were used. However, the protein type hydrolase degradative enzyme destroys cells binding and adhesion factors, leading to significant damage of cells.
|
| Market Environment and CellSeed's Mission |
|
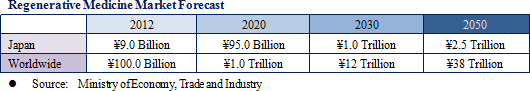
(1) Growth Market: Regenerative Medicine Market Expected to Reach ¥2.5, ¥38 Trillion in Japan, Overseas by 2050
 (2) Medical Product Transition, and "Cell-Sheet Engineering" as a Regenerative Medicine Platform Technology
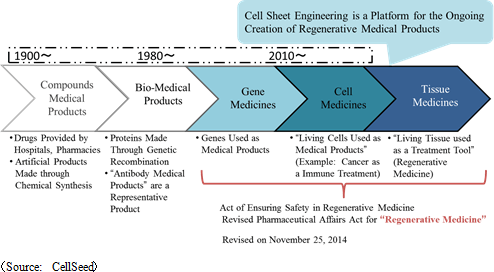 In addition to the challenging technology and cost issues, no supportive establishment of legal systems or policy reviews were available (Legal restrictions based upon the assumptions of pharmaceutical compounds acted as barriers to the diffusion of biotechnology based pharmaceuticals) at the time when "biotechnology-based pharmaceuticals" first appeared. In those days, establishing the market itself was questioned. Now, after 30 years, the sales of biotechnology-based pharmaceuticals accounts for 30% of the total pharmaceutical product sales. The technological and cost hurdles of "tissue-based pharmaceuticals" remain in place, but Prime Minister Abe's administration has identified these technologies as a strategic growth realm and has therefore promoted legal revisions designed to promote the diffusion of and facilitate the environment for "biotechnology-based pharmaceuticals", including the introduction of an "early approval system" in the Pharmaceuticals and Medical Devices Act and recognition of "outsourced cell cultivation processing" in the Act of Ensuring Safety in Regenerative Medicines, etc. (3) CellSeed's Mission
・Realize regenerative medicines quickly・Promote cell sheet technologies throughout the world ・Contribute to medical reforms by providing safe and high-quality products and services CellSeed aims to accelerate its growth by implementing these missions and predicting the movements of the future industrialization of regenerative medicine, and to shift from a company supporting clinical development of the basic research at universities to a new stage as a company to realize regenerative medicine and to generate profits. |
| Progress in the Midterm Business Strategy (FY12/15-17) |
|
<Midterm Business Plan Overview ・ Develop Cell Sheet Regenerative Medicine, Achieve Global Diffusion>
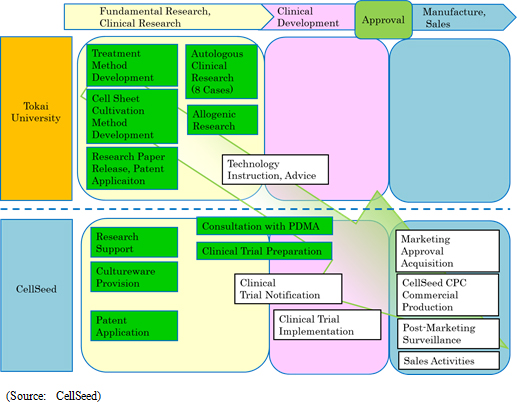
In light of the enforcement of the new law regarding regenerative medicine, CellSeed has identified Japan as the top priority hub of cell sheet regenerative medicine development. Consequently, efforts to achieve commercialization of regenerative medicine products at an early stage and to fortify development and sales of temperature-responsive cell cultureware will be implemented as a means of securing earnings. CellSeed has also identified epithelial cell sheet for esophageal regeneration and regenerated cartilage sheet as the prioritized in-house development pipelines, and the developed cell sheet regenerative medicine products will be expanded on a global basis.
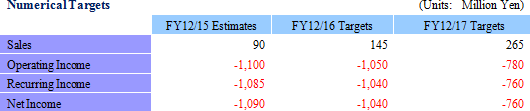 Estimated sales include only those of the intelligent cell cultureware business. The product and service line-up will be fortified along with strengthening of the information sharing and customer service functions as a part of CellSeed's strategy of expanding sales channels to a diverse range of research institutions and corporations that handle cells, let alone those entities in the regenerative medical realm. With regards to profit and loss, increases in clinical trial related expenses, labor expenses arising from fortified marketing functions, and developmental expenses due to new product development for temperature-responsive cell cultureware are anticipated. In addition, the CPC is expected to begin operations in fiscal year December 2016, and subsequent depreciation expense from the start of operation has also been incurred into operating expenses. At the same time, scale merit is expected to be derived from an increase in sales and the transfer of fabrication of temperature-responsive cell cultureware to Dai Nippon Printing Co., Ltd. (DNP) are expected in fiscal year December 2017, which leads to a significant improvement in profitability, supposedly exceeding the increase in sales. <Progress in Achievements>
(1)Cell cultivation Facility Newly Constructed
A cell processing center (CPC) will be built within the Telecom Center Building, 2-5-10 Aomi, Koto-ku, Tokyo (Total floor space of approximately 763 square meters). Capital investment for the CPC is expected to amount to ¥630 million and it is expected to be completed during the first half of 2016. CellSeed seeks to achieve a stable supply structure for regenerative cell sheets with a view to commercial production at an early stage, and to operate the new plant in line with the Ministry Ordinance Regarding Standards for Management of Quality and Production of Regenerative Medical Products. The CPC is comprised of four separate units, with the current cultivation method, able to produce regenerative cell sheets for 100 patients. Development of automated equipment is being promoted, along with efforts to improve productivity and increase output capacity. In addition, the headquarters will be moved to the same building as the CPC in order to pursue efficiency of operations (scheduled in January 2016).
(2)Early Commercialization of Regenerative Medical Products
Esophageal Regeneration Epithelial Cell Sheet
Epithelial cell sheet for esophageal regeneration is cell sheet used as regenerative treatment method in esophageal cancer (Prevention of esophageal narrowing) and was developed by the Institute of Advanced Biomedical Engineering and Science of Tokyo Women's Medical University. The cells are taken from the patient's oral mucosa and cultivated to be the cell sheet in the temperature-responsive cell cultureware. The cultured cell sheet is transplanted to the affected area of the patient after the surgery to remove the esophageal cancer.
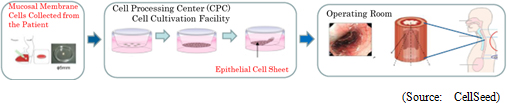  Clinical Research Results of 30 Investigator-initiated Cases both Within and Outside of Japan
Until now, in the realm of esophageal cancer treatment using epithelial cell sheet for esophageal regeneration, investigator-initiated clinical research has been performed on 20 cases in Japan (10 cases at the Tokyo Women's Medical University, and another 10 cases at other Universities in Japan) and another 10 cases in Sweden (all at the Karolinska University Hospital). The efficacy and safety of the treatment have been reported in dissertations and at academic conferences, etc.
Corporate Sponsor-initiated Clinical Trials Begins in Japan, and Sweden
A fundamental development agreement has been formed with Tokyo Women's Medical University in Japan for corporate sponsor-initiated clinical trials. During the current term, preparations are being made to facilitate a clinical trial structure including strategic consultations with the Pharmaceuticals and Medical Devices Agency (PMDA) in preparation for submission of clinical trials notification. Moreover, corporate sponsor-initiated clinical trials are being planned in Sweden for the acquisition of sales approval in Europe, and CellSeed Sweden AB was established in May 2015 for the promotion of these trials. Besides the PMDA provides instruction and advice to improve the safety of medical products and equipment.
Market Size
According to the estimate by CellSeed that is based on the information on the cancer patients published by Center for Cancer Control and Information Services, National Cancer Center, as well as the survey conducted by Nikkei Newspaper on "the 47 Hospitals that have performed over 80 cases surgery for esophageal cancer patients", there were 20,700 esophageal cancer patients in 2012, and approximately 20 % (4,140) of them underwent endoscopic esophageal resection. These over 4,000 patients can be considered as potential recipients of cell-sheet treatments. A large number of esophageal cancer patients are male, and the disease prevalence rate is on the rise. Given that the growth in the number of deaths is lower than that of the disease prevalence rate, the demand for treatments for post esophageal cancer surgery and esophageal narrowing is estimated to be on the rise.
Regenerated Cartilage Sheet
Regenerated cartilage sheet is used for indications of cartilage defect and osteoarthritis caused by sports injuries and aging (both have no permanent cures currently), and allows fundamental regeneration of the cartilage surface by transplanting cell sheets (Regenerated cartilage sheet) made from cultivated cartilage cells.
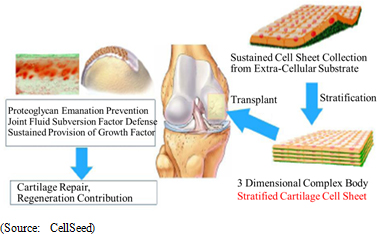  (3) Global Business Deployment
As explained earlier, CellSeed Sweden AB has been established as a 100% owned subsidiary in May 2015. CellSeed Sweden AB will lead the clinical trials for esophageal regeneration epithelial cell sheets in Europe (A contract with a local CRO services company has been already concluded). In addition, CellSeed Sweden participated in the International Society for Stem Cell Research (ISSCR) 2015 convention held in Stockholm in June and had discussions with professors of the Karolinska Institute.
CellSeed Sweden AB Overview
(4)Promotion of Cell Cultureware Development and Expansion of Earnings Generation Opportunity
Outsourcing full-scale manufacturing of "temperature-responsive cell cultureware" to DNP has begun from this spring. Moreover, efforts will be made to develop cell cultureware, cell sheet transplant devices and other new products. In addition to the development for the new products in the realm of conventionally used for research application and earnings growth will be pursued through efforts to develop new products in the clinical application.
(5) Capital Sourcing: Issuance of Options for New Shares through Third Party Placement
While funds required for clinical trials expected to be conducted until fiscal year December 2017 have already been secured, options for new shares will be issued through a third party placement to secure working capital for preparation of in-house cell processing center facilitation, operations, intelligent cell cultureware business-related purchases and promotion of businesses inside and outside of Japan.
13th Options for New Shares
|
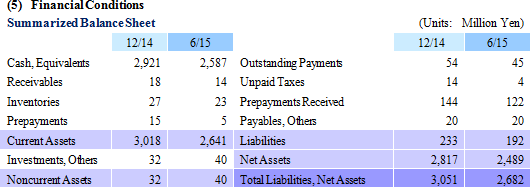
| First Half of Fiscal Year December 2015 Earnings Estimates |
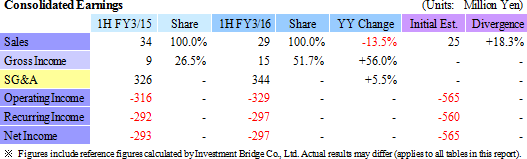 Temperature-Responsive Cell Cultureware Sales of ¥29 Million Booked
With regards to temperature-responsive cell cultureware, sales of products during the first half, which had been expected to be delivered during the second half, contributed to the booking of ¥29 million in sales, compared with estimates of ¥25 million. At the same time, a portion of the ¥134 million (¥98 million in the previous first half) in research and development expenses which had been expected to be booked in the first half were delayed to the second half in the cell sheet regenerative medicine business allowing operating loss to decline to ¥209 million from ¥227 million.
  |
| Conclusions |
|
CellSeed's step-up seems to be smooth from a "research support company" that is at the stage of seeking to "connect fundamental research seeds at university with clinical development" to become a company that is able to "create earnings by realizing commercial applications of regenerative medicine" under the new management team. Disclaimer
This report is intended solely for information purposes, and is not intended as a solicitation to invest in the shares of this company. The information and opinions contained within this report are based on data made publicly available by the Company, and comes from sources that we judge to be reliable. However we cannot guarantee the accuracy or completeness of the data. This report is not a guarantee of the accuracy, completeness or validity of said information and or opinions, nor do we bear any responsibility for the same. All rights pertaining to this report belong to Investment Bridge Co., Ltd., which may change the contents thereof at any time without prior notice. All investment decisions are the responsibility of the individual and should be made only after proper consideration.Copyright(C) 2015 Investment Bridge Co., Ltd. All Rights Reserved. |




