| CellSeed Inc. (7776) |
|
||||||||
Company |
CellSeed Inc. |
||
Code No. |
7776 |
||
Exchange |
JASDAQ |
||
Industry |
Precision Instrument (Manufacturing) |
||
President |
Setsuko Hashimoto, Ph.D. |
||
HQ Address |
Telecom Center Building, Aomi 2-5-10, Koto-ku, Tokyo |
||
Year-end |
December |
||
URL |
|||
* Stock price as of closing on February 22, 2017. Number of shares at the end of the most recent quarter excluding treasury shares. ROE and BPS are the actual values of the previous term.
|
||||||||||||||||||||||||
|
|
* Estimates are those of the company. From FY12/16, the definition of net income has been changed to net income attributable to the parent company shareholders (the same shall apply hereinafter).
This Bridge Report presents fiscal year 2017 earnings results and the future outlook of CellSeed Inc. |
|
| Key Points |
 |
| Company Overview |
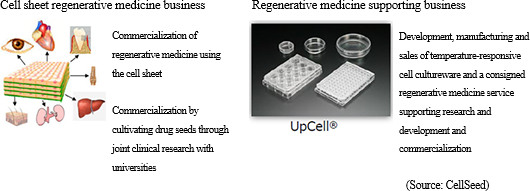  <Regenerative Medicine and CellSeed's Business Strategies>
Regenerative medicine is a form of medical treatment that can regenerate organs that have reduced, damaged, or lost functions. Stem cells are cells that can be differentiated into various cells to be used as key components within regenerative medicine technology applications. Currently, there are three types of stem cells including "ES cells" created from fertilized eggs, "iPS cells" induced pluripotent cells, and "organ stem cells" from various living organs. While "ES cells" created from fertilized eggs have the ability to be differentiated into all cell types, there are ethical issues regarding the use of fertilized eggs and they have yet to become commercialized due to this issue. Four types of genes are introduced to "iPS cells" to be cultivated into different types of cells including skin as induced pluripotent cells, and research has been promoted in the wake of the Nobel Prize awarded to Professor Shinya Yamanaka of Kyoto University. However, much time is expected to be required before commercialization can be achieved as the differentiation process needs to be explained.At the same time, CellSeed views somatic stem cells as the closest stem cells to being commercialized, and it is conducting clinical development of applications of cultivated cell sheets to be used in transplants to patients' affected regions (esophageal and knee cartilage regenerative medical products). Specifically, the company promotes clinical trials with the aim of submitting an application for approval of sale of "epithelial cell sheets for esophageal regeneration" in 2018 along with preparations for the clinical trials of "regenerated cartilage sheet" in 2017. Regenerative medical products using "cell sheet engineering" are applied not only to esophagus and knee cartilage, but clinical trials are also implemented in treatments involving cornea, teeth, ear, cartilage, lung, heart, liver and pancreas, and clinical data have already been gathered. 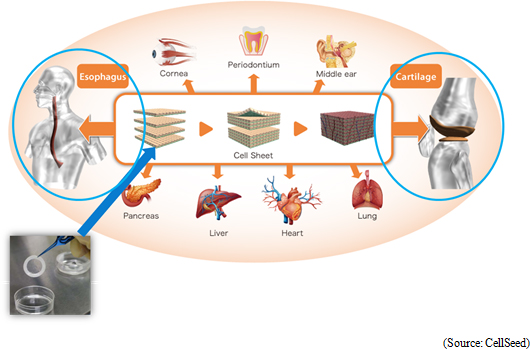 As explained earlier, CellSeed is focused upon developing treatments of culturing cells in sheet format using "cell sheet engineering" and then transplanting them to patients (regenerative medical products of esophagus and knee cartilage). The "cell sheet" is also used in "iPS cell" reprogramming (cell creation). |
| Fiscal Year 2017 Earnings Results |
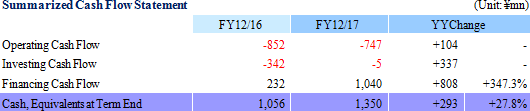
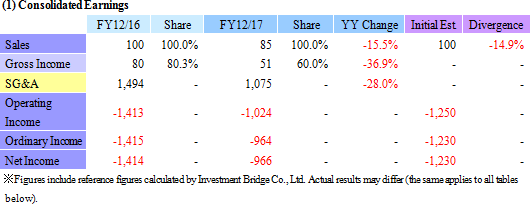 Research and Development of "Epithelial Cell Sheet for Esophageal Regeneration" and "Regenerated Cartilage Sheet" Show Smooth Progress
Sales declined by 15.5% year-on-year to ¥85 million. Sales declined from ¥50 million of the previous term to ¥15 million of the current term in the cell sheet regenerative medicine business, whereas sales of the regenerative medicine supporting business increased from ¥50 to ¥69 million over the same period due to the early start of new product sales and revisions in the sales strategy aiming at overseas markets. During the previous term, CellSeed recorded sales of ¥50 in the cell sheet regenerative medicine business as fees for data provision at the time of the contract signing with MetaTech (Taiwan) and sales of ¥16 million as consulting fees from MetaTech for concluding an exclusive alliance agreement, which grants MetaTech the rights to commercialize "epithelial cell sheet for esophageal regeneration" and "regenerative cartilage sheet."With regard to profitability, a 40.6% year-on-year reduction in research and development expenses from ¥1.065 billion to ¥632 million led the operating loss to shrink. Also, non-operating income also improved due to an increase in subsidies the company received from ¥9 to ¥67 million and a decline in foreign exchange loss from ¥13 to ¥0.6 million over the same period. 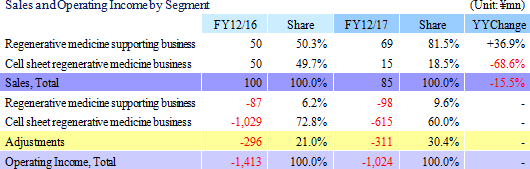 (2) Business Segment Trends
Cell Sheet Regenerative Medicine
Epithelial Cell Sheet for Esophageal Regeneration (CLS2702C/D)
This cell sheet was developed at the Institute of Advanced BioMedical Engineering and Science, Tokyo Women's Medical University for the promotion of wound healing and the prevention of inflammation and esophageal stricture after resection of cancer cells from the esophageal mucosa.Cells taken from the patient's mucosal membrane are cultivated in temperature-responsive cell cultureware for two weeks and turned into cell sheets. Cell sheets are cultured for the use of transplanting into parts of the esophagus that are infected by cancer and removed by surgery.According to CellSeed, 22,000 patients within Japan are diagnosed with esophageal cancer every year (90% of the esophageal cancer cases diagnosed within Japan are squamous cell carcinoma), with 11,500 patients dying every year. In addition, the rate of occurrence and death related to esophageal cancer in male patients is five times that of female patients, with five year survival rates for males and females said to be 36% and 44%, respectively. The endoscopic resection surgery (ESD) was posted in the drug price list from 2008 and is on the rise (about 20% of patients diagnosed with esophageal cancer undergo surgery), but its side effect of esophageal stricture after surgery has been recognized. 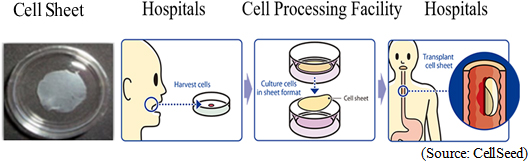 Designated under SAKIGAKE Designation System CellSeed's regenerative medicine product was designated under "SAKIGAKE Designation System" by the Ministry of Health, Labour and Welfare. Treatments selected by this system receive preferential treatments such as prioritized consultations and reviews for pharmaceutical approval, advice on manufacturing, and post-approval support for smooth introduction into the medical market. (In addition to the merits of prioritized consultations, pre-application consultation, prioritized reviews and the review partner system, it makes it possible to shorten the premarket review period to six months, the half the usual time). Obtaining CE Marking for Cell Sheet Transplant Devices in Accordance with the EU Medical Device Directive (Approval as Medical Device Required Overseas) With regard to cell sheet transplant devices, in October 2017, CellSeed obtained CE Marking in accordance with MDD, including approval of "ISO 13485," an internationally recognized standard related to a quality management system specific to medical devices. Acquisition of this certification is expected to further encourage the commercialization of the epithelial cell sheet for esophageal regeneration business in Taiwan by MetaTech. At the same time, it is also expected to accelerate CellSeed's business expansion of its epithelial cell sheet for esophageal regeneration in overseas markets. The manufacture of cell sheet transplant devices has been consigned to Yasui Co., Ltd. (Miyazaki Prefecture)  Regenerated Cartilage Sheet(CLS2703C)
Regenerated cartilage sheet is the result of research conducted jointly by CellSeed and Professor Masato Sato of Tokai University School of Medicine Department of Orthopedics, and is a treatment designed to be used on patients with damaged and deformed cartilage caused by injuries from sports and osteoarthritis due to aging. Although there are no effective treatments currently available for them, research is being conducted jointly with Professor Sato aiming for the fundamental regeneration of cartilage surface. Knee cartilage is known as "glass cartilage" and differs from cartilage of the ear and nose in its superior cushioning and wear functions, which makes its regeneration difficult. However, regeneration of "glass cartilage" has been confirmed in joint clinical research using "regenerated cartilage sheet."According to CellSeed, knee osteoarthritis is a refractory disease where articular cartilage degeneration progresses slowly and the number of patients over the age of 40 within Japan is estimated to be 25.30 million, of which some 8.00 million are estimated to have active symptoms (survey conducted by Tokyo University School of Medicine). In addition, the number of patients is expected to increase due to Japan's aging society, and therefore it is considered a disease that needs to be handled urgently from the aspects of health and longevity of the Japanese, and nursing and medical care expenses. According to the Comprehensive Survey of Living Conditions conducted by Ministry of Health, Labor and Welfare in 2013, 25% of total patients in need of support or nursing care suffer from symptoms of immobility. 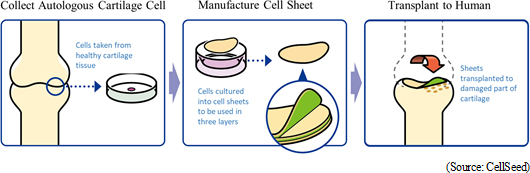 Joint Research with Tokai University
In August 2011, clinical research on transplant of autologous cell sheets was approved by the 65th Scientific Technology Committee of the Minister of Health, Labour and Welfare Medical Care Deliberation Committee. In October 2011, with the written opinion issued by the Minister of Health, Labour and Welfare (MHLW Legal Document 1003 Number 3), the first case clinical research was conducted. Furthermore, two-year evaluations of eight cases were completed in November 2015. CellSeed has signed a mutual agreement for development (agreement on owning development rights domestically and internationally) and an agreement on joint patent application with Tokai University. With regard to patents, the national patent has already been granted and international patents are currently under review. In the future, CellSeed expects to expand its patent portfolio. Starting strategic consultations with the Pharmaceuticals and Medical Devices Agency (PMDA), CellSeed will work on the accumulation of safety data and establishment of clinical protocols in association with further consultations and advice.Application for Advanced Medicine Tokai University is preparing for obtaining approval of advanced medicine. CellSeed agrees with the idea of "implementation of the advanced medicine will contribute to future evaluations as to whether our product qualifies to be covered by insurance. CellSeed will begin clinical trials considering the evaluations received after the implementation of the advanced medicine. Currently, the company cultures regenerated cartilage cell sheets as consigned by Tokai University at its Cell Processing Facility (consigned processing), but it will be able to derive profit from this consigned processing once this process is recognized as an advanced medicine. Moreover, advanced medicine targets treatments using high medical technologies or medical technologies themselves which meet the certain criteria in terms of efficacy and safety even before the sales approval. While costs of treatments before the sales approval need to be covered entirely by the patient, in certain instances health insurance may be used together, unlike medical care not covered by health insurance. (In principle, insurance cannot be used for medical care not covered by health insurance.) Allogeneic Cell Sheet Transplant (Immunosuppressant Drugs Unnecessary for Treatment) Professor Sato of Tokai University conducted the world's first allogeneic cell sheet transplant surgery (clinical research) on February 15, 2017. In this clinical research, transplant surgeries using cartilage tissue that is acquired from polydactyly patients and cultured into cell sheets for two to three weeks (cartilage tissue taken by consent from congenital infant patients with six fingers) are expected to be conducted on ten patients over three years. The technologies related to cultivation of allogenic cell sheets is being transferred from Tokai University to CellSeed. Moreover, our regenerated cartilage sheet using allogeneic cells has been selected as a research and development topic by the Japan Agency for Medical Research and Development (AMED) for its "Project Focused on Developing Key Evaluation Technology: Manufacturing Technology for Industrialization in the Field of Regenerative Medicine" (project duration: until March 2019) and received a subsidy from the government. 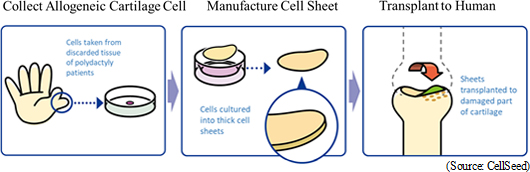 Regenerative Medicine Supporting Business
Differing from conventional dish type, the flask type "HydroCell TM" has been introduced to fortify the company's lineup of products during fiscal year 2017. During fiscal year 2018, a consigned regenerative medicine service will begin in the Cell Processing Facility on the sixth floor of the East Tower of the Telecom Center Building, in which CellSeed's headquarters are located, in addition to strengthening sales of new products. This Cell Processing Facility obtained the accreditation of foreign cell processor (FA3160008) in March 2017.
  |
| Mid-Term Business Plan (Fiscal Year 2018 to 2020) |
|
<Main Points of the Mid-term Business Plan>
 Obtain approval and start sales for epithelial cell sheet for esophageal regeneration Obtain approval and start sales for epithelial cell sheet for esophageal regeneration Accelerate development of regenerated cartilage sheet Accelerate development of regenerated cartilage sheet Start development of the next generation product Start development of the next generation product Establish a supply chain system Establish a supply chain system Develop new regenerative medicine supporting products and cultivate earnings opportunities Develop new regenerative medicine supporting products and cultivate earnings opportunities Promote business alliance for global expansion Promote business alliance for global expansionObtain Approval and Start Sales for Epithelial Cell Sheet for Esophageal Regeneration, Establish Supply Chain System
CellSeed intends to submit an application for sales approval in the fourth quarter of fiscal year 2018 at the earliest and in the first quarter of fiscal year 2019 at the latest. It will obtain the approval during fiscal year 2019 and smoothly start sales after NHI price listing. By fiscal year 2020, the sales will stabilize. From fiscal year 2018 to fiscal year 2019, it will proceed with establishment of a supply chain starting from hospitals (acquisition of mucosal membrane) to Cell Processing Facility (cell cultivation into sheet format) to hospitals (cell sheet transplant). As the most prioritized matter being domestic commercialization of the cell sheet regenerative medicine business, CellSeed will consider the priority of the development of epithelial cell sheet for esophageal regeneration in Europe as a candidate product of the "next generation product" for development (described later).
Accelerate Development of Regenerated Cartilage Sheet
As for the regenerative autologous cartilage sheets, Tokai University, the partnering research institution, plans to submit an application for approval of advanced medicine during the first half of fiscal year 2018. Corporate led clinical trials will also be conducted, taking into account the results of advanced medicine. Consigned processing of cell sheets used in advanced medicine is to be done as a fee-based service. Current clinical research for regenerated cartilage sheet using allogeneic cells at Tokai University is expected to continue in fiscal year 2019. CellSeed is expected to begin clinical trial preparations, the regulatory science strategy and regulatory science comprehensive consultations with a view to starting corporate led clinical trials during fiscal year 2020.
Consider Next Generation Product for Development
Currently, CellSeed is promoting the two main product development pipelines, which are "epithelial cell sheet for esophageal regeneration" (Japan) and "regenerated cartilage sheet" (Japan). The company is currently considering the next target product to be developed as the third pipeline for the purpose of commercialization during fiscal year 2018. At this moment, cell sheet for periodontal tissue regeneration and epithelial cell sheet for esophageal regeneration (Europe) are being considered as candidates. The development is expected to begin once preparations including contracts with research institutions for the product and region is determined.
Develop New Regenerative Medicine Supporting Products and Cultivate Earnings Opportunities
Expansion of the regenerated medicine supporting business entails the start of a consigned regenerative medicine service that supports research and development and commercialization of regenerative medicine (provision of comprehensive support for regenerative medicine). A few examples of the service being considered are 1) development of the manufacturing process and consigned manufacturing of cell sheet products, 2) operation and application support, and 3) cell culturing technician training.
Promote Business Alliance for Global Expansion
CellSeed concluded an exclusive alliance agreement with MetaTech(AP) Inc. in April 2017, granting the exclusive rights for development, manufacturing and sales of its cell sheet regenerative medicine business (epithelial cell sheet for esophageal regeneration and regenerated cartilage sheet) in Taiwan to the company. Fees for granting the exclusive rights are expected to amount to a maximum of \1.250 billion. Consequently, the development and commercialization of the cell sheet regenerative medicine business in Taiwan will be promoted by MetaTech along with the support of CellSeed. MetaTech is proactively promoting development and CellSeed expects to provide the bulk of its development and manufacturing related data to MetaTech during fiscal year 2018, according to its progress in 2017. In addition to the business alliance with MetaTech, CellSeed will pursue other business alliances and licensing opportunities to diffuse its cell sheet regenerative medicine business in Asia, North America and Europe.Moreover, MetaTech(AP) Inc., the business partner in Taiwan, was established on September 17, 1998 and is a Taiwan OTC listed company. According to this contract, CellSeed is to grant MetaTech the exclusive rights for development, manufacturing and sales of its cell sheet regenerative medicine business (epithelial cell sheet for esophageal regeneration and regenerated cartilage sheet), and provide development and manufacturing related data in line with the development progress of MetaTech. At the same time, MetaTech is expected to pay CellSeed development milestones corresponding to its development progress, fees for the data provision and a royalty corresponding to sales after the launch of the business. 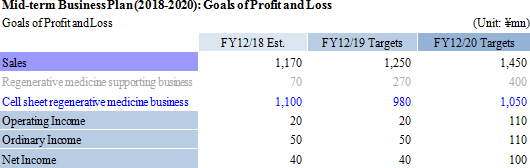 In the cell sheet regenerative medicine business, CellSeed will continuously put efforts into the development of epithelial cell sheet for esophageal regeneration and regenerated cartilage sheet. MetaTech is proactively promoting the development of cell sheet regenerative medicine business (epithelial sheet for esophageal regeneration and regenerated cartilage sheet) based upon the exclusive rights for development, manufacturing and sales which CellSeed granted, and provision of the bulk of development and manufacturing related data is expected to be completed within fiscal year 2018. In exchange for the provision of development and manufacturing related data, CellSeed is expected to receive fees of about \1.0 billion. In addition, CellSeed will pursue further business alliance and licensing opportunities in Asia, Europe and North America, aiming to diffuse the cell sheet regenerative medicine business, based on the experience of business alliance with MetaTech. As a result of these efforts, the sale of this segment is expected to amount to \1.1 billion during fiscal year 2018 |
| Future Highlights |
|
|
| <Reference: CellSeed's corporate governance> |
 ◎Corporate Governance Report
Latest Update: April 5, 2017
Basic Policy
With the missions to introduce technological innovations, to exert creativity and to contribute to people's health and welfare by providing high-quality products and services, we are enhancing corporate governance to raise quality in all of our corporate activities.In the future, we will increase our accountability further to improve the transparency of disclosed information and strengthen our checking system even more. <Reasons for Non-compliance with the Principles of the Corporate Governance Code (Excerpts)> CellSeed has stated, "Our company implements all the basic principles stipulated in the Corporate Governance Code as a JASDAQ listed company." Disclaimer
This report is intended solely for information purposes and is not intended as a solicitation to invest in the shares of this company. The information and opinions contained within this report are based on data made publicly available by the company and obtained from sources that we judge to be reliable. However, we cannot guarantee the accuracy or completeness of the data. This report is not a guarantee of the accuracy, completeness or validity of said information or opinions, nor do we bear any responsibility for the same. All rights pertaining to this report belong to Investment Bridge Co., Ltd., which may change the contents thereof at any time without prior notice. All investment decisions are the responsibility of the individual and should be made only after proper consideration.Copyright(C) 2018 Investment Bridge Co., Ltd. All Rights Reserved. |




