Bridge Report:(7776)CellSeed the second quarter of fiscal year ending December 2020
 Setsuko Hashimoto, President &CEO | CellSeed Inc.(7776) |
 |
Company Information
Market | JASDAQ |
Industry | Precision Instrument (Manufacturing) |
President | Setsuko Hashimoto, Ph.D. |
HQ Address | Telecom Center Building, Aomi 2-5-10, Koto-ku, Tokyo |
Year-end | December |
Homepage |
Stock Information
Share Price | Number of shares issued (excluding treasury shares) | Total market cap | ROE Act. | Trading Unit | |
¥339 | 14,259,265shares | ¥4,833million | - | 100 shares | |
DPS Est. | Dividend yield Est. | EPS Est. | PER Est. | BPS Act. | PBR Act. |
- | - | - | - | ¥103.21 | 3.3x |
*Stock price as of closing on September 2, 2020. Number of shares at the end of the most recent quarter excluding treasury shares. ROE is the actual values of the previous term. BPS is the actual values of the second quarter of this term.
Earnings Trend
Fiscal Year | Sales | Operating Profit | Current Profit | Net Profit | EPS | DPS |
December 2016 | 100 | -1,413 | -1,415 | -1,414 | - | - |
December 2017 | 85 | -956 | -964 | -966 | - | - |
December 2018 | 1,026 | 140 | 140 | 129 | 11.35 | - |
December 2019 | 275 | -780 | -786 | -782 | - | - |
December 2020 Est. | - | - | - | - | - | - |
* (Unit: million yen, yen). 2020Est is not available at this moment
This Bridge Report presents the second quarter of fiscal year ending December 2020 earnings results and the future outlook of CellSeed Inc.
Table of Contents
Key Points
1. Company Overview
2. Second Quarter of Fiscal Year ending December 2020 Earnings Results
3. Fiscal Year ending December 2020 Earnings Forecasts
4. Conclusions
<Reference: Regarding Corporate Governance>
Key Points
- For the first half of the term ending December 2020, sales stood at 58 million yen, down 64.1% year on year, and operating loss was 340 million yen (an operating loss of 321 million yen in the same period of the previous year). Sales in the regenerative medicine supporting business were almost the same as those posted in the same period a year ago. While sales from the cell cultureware, etc. hit a record high both in Japan and overseas in the second quarter alone and for the first half of the term, with regard to the regenerative medicine consignment services, sales generated by providing development data to MetaTech in Taiwan for this term were only 1 million yen, which were down from 100 million yen recorded in the same period last year. However, decreases in research and development expenditure and other expenses, including ones not spent, resulted in only a slight increase of operating loss.
- Business forecasts for the full term have not been provided yet. The global spread of the novel coronavirus infections has led to a delay in general developmental support for the joint venture established in Taiwan in January 2020 and has been restricting activities aimed at forging new business alliances and licensing in Asian countries, such as China and Taiwan. These impacts have caused the company to withdraw the business forecasts released on February 14, 2020 and make it difficult for it to provide new ones now because “it is impossible to perform reasonable calculations at the moment” regarding the full-year consolidated earnings for the term ending December 2020.
- The coronavirus pandemic not only has delayed clinical tests of the epithelial cell sheet for esophageal regeneration by a Taiwanese company MetaTech, but also has been placing restrictions on activities with the Taiwanese joint venture and ones aiming at building new business alliances and licensing. There, however, is a silver lining in this field; the light is seen at the end of the tunnel regarding submission of a notification of additional clinical trial plans in Japan for the epithelial cell sheet for esophageal regeneration. Besides, although a surgery for the first patient under the category of Advanced Medical Care B Program has been postponed due to the Covid-19 pandemic, the company has completed manufacturing of cartilage cell sheets on consignment for the project and sales are expected to be recorded in the second half of the current term. Consignment manufacturing of cell sheets is expected to contribute to increasing revenue. In addition, issuance of share options has allowed the company to raise funds of slightly less than 1.3 billion yen. The mid-term business prospect is becoming rosier little by little.
1. Company Overview
Regenerative medicine is a new kind of medicine for regenerating and curing lost, damaged or deteriorated tissues. CellSeed uses the fundamental technologies of “cell sheet engineering” developed in Japan by Professor Okano of the Tokyo Women’s Medical University in its “cell sheet regenerative medicine” that employs “cell sheets” for the cell regenerative medicine business, and the regenerative medicine support business, where temperature responsive cell cultureware used to fabricate cell sheets are developed and sold and the regenerative medicine consignment services, which support for research and development and commercialization of regenerative medicine, is provided.
“Cell sheet engineering” – Basic Technologies for Regenerative Medicine -
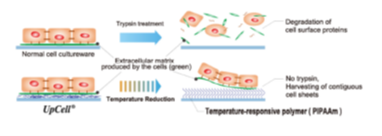 (From the company material) | “Cell sheet engineering” is the world’s first platform technology developed in Japan by Professor Emeritus Mitsuo Okano of Tokyo Women’s Medical University. Cells are cultured in cell cultureware whose surface is processed with the temperature responsive polymer that changes the molecular structure according to temperatures. The surface of the cell cultureware becomes moderately hydrophobic (waterproof) at 37 degrees Celsius to allow cells to adhere to it and hydrophilic (containing water) at a temperature of 20 degrees Celsius to prohibit cells from attaching to it. Thus, an organically combined cell sheet can be obtained from the cultureware just by changing temperatures while the extracellular matrix (adhesive proteins) is maintained. In general, cells grow by secreting the extracellular matrix and fixing themselves. In other words, they cannot develop unless they fix themselves somewhere while releasing adhesive proteins, and cells cultured are conventionally harvested by decomposing adhesive proteins using such proteolytic enzymes as trypsin (breaking down adhesive proteins was the only way to obtain cells cultured). |
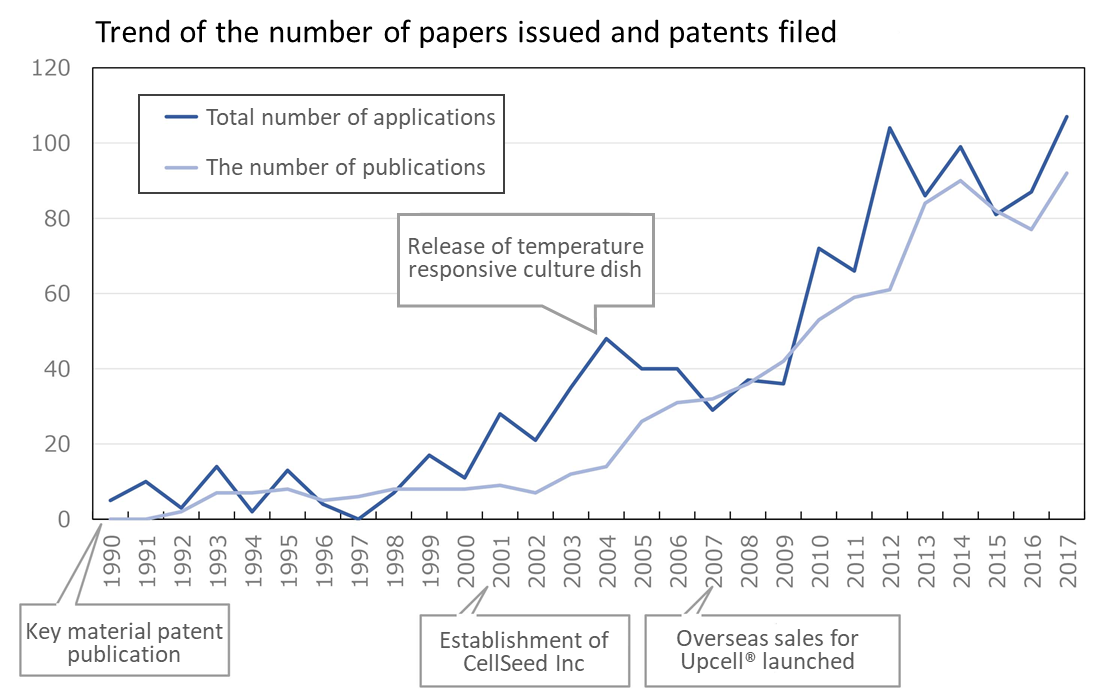
Japan Leads the World in Cell Sheet Engineering.
One of the first papers on “cell sheet engineering” was published about 30 years ago, and since then, a number of papers and patent applications have been submitted. The technology is gaining popularity as seen in that the number of papers issued and patents filed exceeds 100 every year. The greater part of those papers and patents are presented by Japanese researchers and companies, making it one of the few technologies in which Japan leads the world.
(From the company material)
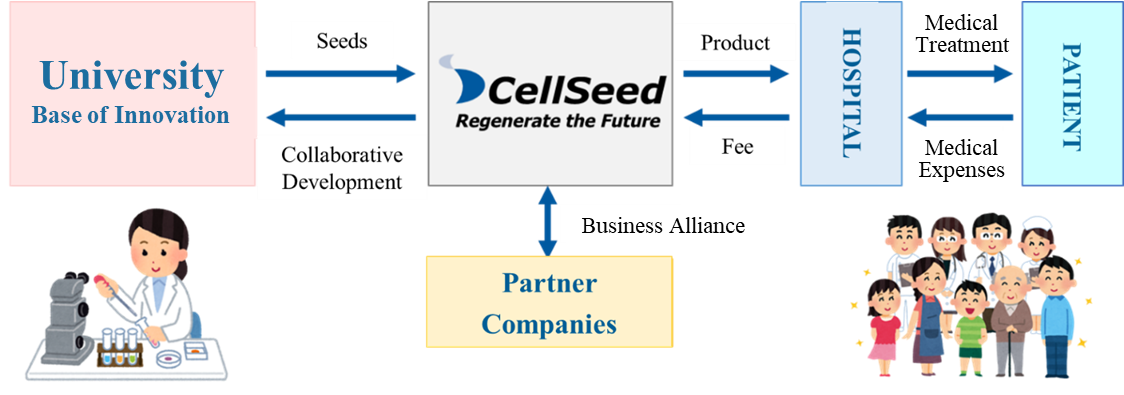
1-1 Business model of CellSeed
Mission: We take the initiative of contributing to global health care in the valuable and innovative field of regenerative medicine.
Using the outcomes of research into cell sheets conducted at universities as seeds, the company performs clinical trials, transforms them into regenerative medicine products, and provides products to patients.
(From the company material)
1-2 Business Description
Cell sheet regenerative medicine business
The current development pipeline consists of the two main realms of “epithelial cell sheet for esophageal regeneration” based upon the fundamental technology of “regenerative cell sheet engineering”, and “regenerated cartilage sheet” of knee osteoarthritis.
The company plans to submit an additional clinical trial notification for “epithelial cell sheet for esophageal regeneration” in Japan in the fourth quarter in this term. On the other hand, as for business in overseas nations, CellSeed entered into a business alliance with MetaTech (AP) Inc. (hereinafter referred to as MetaTech) in Taiwan in April of fiscal 2017 and the company submitted a clinical trial notification at the end of December 2018.
Meanwhile, the “regenerated cartilage sheet,” for which Tokai University Hospital had submitted an application for approval, was approved as advanced medicine in January 2019, allowing the company to prepare for starting to offer the medical treatment at the university hospital. Furthermore, CellSeed has licensed out the product to MetaTech and efforts are put forth to commercialize autologous cartilage sheets in accordance with the Taiwanese law (which is equivalent to those governing Japan’s Advanced Medical Care B Program).
Clinical trials of regenerative medical products utilizing cell sheet engineering have been carried out in a variety of fields, and the company decided in August 2019 to initiate discussion with Tokyo Medical and Dental University in order to consider details about clinical development of the periodontal ligament-derived mesenchymal stem cell sheets (periodontal membrane cell sheets) as the third pipeline to develop.
“Epithelial Cell Sheet for Esophageal Regeneration”
22,000 patients within Japan are diagnosed with esophageal cancer every year with 11,500 patients dying every year. The rate of occurrence and death related to esophageal cancer in male patients is five times that of female patients. In addition, 90% of the esophageal cancer cases diagnosed within Japan are squamous cell carcinoma and five years survival rates for males and females, which is said to be 36% and 44%, respectively, are low. he endoscopic resection surgery (ESD) was posted in the drug price list from 2008 and is on the rise, but its side effect of esophageal stricture after surgery has been recognized as a problem.
However, introduction of epithelial cell sheets for esophageal regeneration will allow esophageal strictures to develop less frequently, which is expected to improve the quality of life of patients. The treatment with “the epithelial cell sheet for esophageal regeneration” was developed by Tokyo Women's Medical University, in order to solve the problem with the regenerative medical treatment against esophageal cancer (treatment of esophageal tear and prevention of stenosis). Cells taken from the oral mucosa of a patient are cultured for about 2 weeks using the temperature-responsive cell cultureware to produce cell sheets. In conjunction with the process of culturing cell sheets, an endoscopic surgery for esophageal cancer excision is performed and the cell sheets are transplanted to the part of an esophageal tumor in the patient.
Clinical studies were conducted at universities between 2008 and 2014: 10 cases at Tokyo Women's Medical University, 10 cases at Tokyo Women's Medical University and Nagasaki University (long-distance transport validation: cells collected at Nagasaki University were cultured at Tokyo Women's Medical University and transplanted at Nagasaki University), 10 cases at Karolinska University Hospital (Sweden), for a total of 30 The company signed a basic development agreement with Tokyo Women's Medical University and took over the university's research results for commercialization.
Clinical trial notification was submitted in the second quarter of FY12/16 and the trial was completed in the first quarter of FY12/19. However, the trial did not prove a statistical advantage and an additional trial was required. Having completed discussion with the PMDA about additional clinical trials, CellSeed plans to submit a notification of additional clinical trial plans in the fourth quarter of the term ending December 2020.
(Source: the company)
“Regenerated Cartilage Sheet”
The company researched the “regenerated cartilage sheet” with Professor Masato Sato of Department of Orthopedics, Tokai University. Its indications are cartilage defects and osteoarthritis caused by sport injury and aging.
Knee osteoarthritis is refractory articular cartilage degeneration that progresses slowly. The number of potential patients in Japan is estimated to be about 30 million, of which about 10 million patients are thought to have subjective symptoms. Furthermore, population aging in Japan is expected to raise the number of patients diagnosed with the illness, making it a disease that needs to be dealt with immediately from the perspective of citizens’ healthy life expectancy and costs of long-term care and medical services. As of now, there are no methods to cure the injury completely, but the collaborative research with Professor Sato is aimed at regenerating the cartilage surface radically. The cartilage of the knee is called hyaline cartilage, which is hard and excellent in cushioning and abrasion resistance properties, differing from the cartilages of the ear, nose, etc., and it is difficult to regenerate. However, it was confirmed in clinical research that the “regenerated cartilage sheet,” which is being researched collaboratively with the professor, can regenerate the cartilage of the knee as hyaline cartilage.
Professor Masato Sato started clinical research into autologous cartilage sheets in 2010 and has completed the study of 8 cases. In January 2019, “the cartilage regeneration treatment with autologous cell sheets” proposed by Tokai University Hospital was approved as Advanced Medicine B at “the 71st advanced medical care meeting” hosted by the Ministry of Health, Labor and Welfare.
CellSeed will manufacture regenerative cartilage sheets on consignment for treating patients by regenerating cartilage using autologous cell sheets as an advanced medical care program, and a surgery of the first patient under the category of Advanced Medical Care B Program was completed in August of the term ending December 2020. It has licensed out the product to MetaTech, which is preparing for commercializing autologous cartilage sheets in accordance with the Taiwanese law (that is equivalent to those governing Japan’s Advanced Medical Care B Program). In addition, in November of 2019, CellSeed and Tokai University jointly applied for a patent of “Cell Culture Sheet and Manufacturing Method and Use Thereof,” one of the outcomes of the joint research with Professor Masato Sato of the university, in the U.S. and the patent application was approved. This ensures that the intellectual property right of the product is now protected in Japan, the United States of America, and Europe.
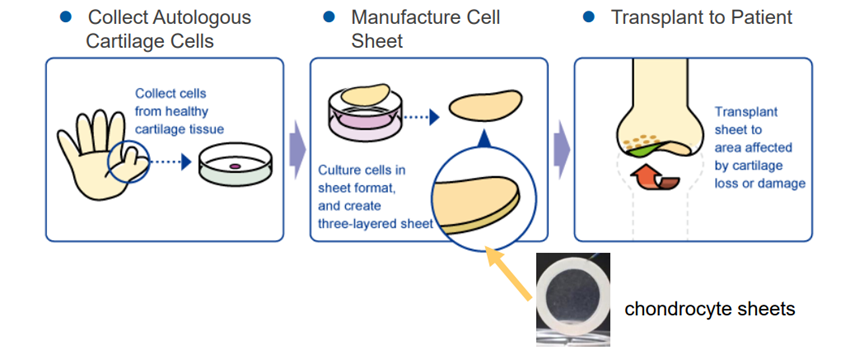
(Source: the company)
Research and development have been propelled forward regarding treatment using allogeneic cartilage sheets (cell sheets based on cells obtained from people other than the patient), and Professor Masato Sato embarked on clinical research (transplantation of allogeneic cartilage cell sheets) in February 2017 and treated the first patient. Ten patients are to be treated for three years until March 2020 and sponsor-initiated clinical trials are slated for 2021. In parallel with clinical research, a cell bank will be established and manufacturing of cell sheets will be automated. The treatment with allogeneic cartilage sheets has been adopted in the project for developing evaluation methods, etc. for the industrialization of regenerative medicine (support for acceleration of development of regenerative medicine seeds) of AMED (project period: Oct. 2018 to Mar. 2021(plan)).
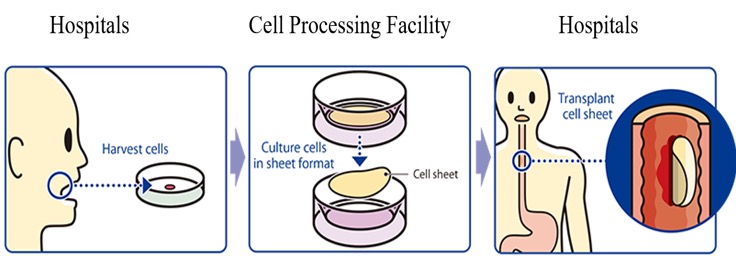
Regenerative Medicine Consignment Services
The company provides regenerative medicine services on consignment in relation to temperature responsive cell cultureware, etc., including development, manufacture, and sales and cell sheet products, including development of manufacturing methods and contract manufacturing, facility management and application support, and training and education in cell culturing technology.
In the commissioned manufacturing of cell sheet products, the company develops and manufactures mainly cell sheets on consignment for pharmaceutical companies and research institutions. The company has a number of staff members with extensive knowledge and experience with cell culturing practices, such as clinical cultivatists certified by the Japanese Society for Regenerative Medicine, and they do those services at the facility with permission for manufacturing and processing specified cell products (Facility Number.: FA3160008). The cartilage regeneration sheet was approved in January 2019 for the advanced medical treatment B that Tokai University had applied for. The cell sheet to be used in this advanced medical treatment will be cultured by the company at its cell culture center (contract manufacturing).
1-3 Cell Culture Center
With a floor space of about 763 square meters, the Cell Culture Center is equipped with an automated monitoring system that controls the cleanliness, room pressure, temperature and humidity, and operational status of equipment (such as incubators and reagent stockers), and a surveillance camera system throughout the entire facility. Besides, the facility is only twenty-minute drive from Haneda International Airport, making it possible and easy to transport products by air. In March 2017, a license to manufacture and process specified cell products as per the provisions set forth in Paragraph 1, Article 35 of the Act on Safety of Regenerative Medicine was granted by the Ministry of Health, Labour and Welfare. Consequently, CellSeed is able to process specified cell products on consignment.

(Source: the company)
2. Second Quarter of Fiscal Year ending December 2020 Earnings Results
2-1 Consolidated Earnings in the first half
| FY 12/ 19 1H | Share | FY 12/ 20 1H | Share | YY Change |
Sales | 162 | 100.0% | 58 | 100.0% | -64.1% |
Gross Income | 133 | 82.3% | 40 | 69.2% | -69.8% |
SG&A | 454 | 280.1% | 381 | 653.4% | -16.2% |
Operating Income | -321 | - | -340 | - | - |
Ordinary Income | -320 | - | -341 | - | - |
Net Income | -319 | - | -340 | - | - |
* unit: million yen
Sales were 58 million yen, down 64.1% year on year, and operating loss was 340 million yen (321 million yen in the same period a year ago).
Sales dropped 64.1% (-104 million yen) year on year to 58 million yen. Sales in the regenerative medicine supporting business were almost the same as those posted in the same period a year ago. While sales from the temperature responsive cell cultureware, etc. hit a record high both in Japan and overseas in the second quarter alone and for the first half of this term, with regard to the regenerative medicine consignment services, sales recorded in the current term by providing development data to MetaTech in Taiwan were only 1 million yen, down from 100 million yen in the same period last year.
As for profit and loss, while the decreased sales reduced gross profit by 69.8% (-93 million yen) year on year; decreases in research and development expenditure (down from 208 million yen to 181 million yen) and other expenses (down from 246 million yen to 199 million yen), including ones not spent, resulted in a 16.2% drop (-73 million yen) of selling, general and administrative expenses from the same period last year, causing operating loss to increase only slightly by 19 million yen to 340 million yen.
2-2 Trends by Segment
| FY 12/ 19 1H | Share | FY 12/ 20 1H | Share | YY Change |
Regenerative medicine supporting business | 57 | 35.3% | 56 | 96.9% | -1.4% |
Cell sheet regenerative medicine business | 105 | 64.7% | 1 | 3.1% | -98.3% |
Sales, Total | 162 | 100.0% | 58 | 100.0% | -64.1% |
Regenerative medicine supporting business | -20 | - | -7 | - | - |
Cell sheet regenerative medicine business | -127 | - | -185 | - | - |
Adjustments | -173 | - | -148 | - | - |
Operating Income, Total | -321 | - | -340 | - | - |
* Unit: million yen
Regenerative Medicine Supporting Business
Sales stood at 56 million yen, down 1.4% from year on year, and operating loss was 7 million yen (an operating loss of 20 million yen was recorded in the same period a year ago). Enhanced collaboration with the existing distributors and more proactive sales promotional activities resulted in sales that marked a record high both in Japan and overseas. In addition to rising sales of UpCell® in overseas markets as in the previous term, sales of instruments to the company’s business partner, MetaTech, also showed growth.
CellSeed’s cell culture instrument, HydroCell™, has been adopted by MiCAN Technologies, Inc. (Headquarters based in Kyoto City, Kyoto Prefecture; Kazuo Miyazaki, Representative and Chief Executive Officer), a company that develops and sells cells for research on infectious diseases caused by various viruses, including the novel coronavirus. MiCAN Technologies is one of the companies that receive investment from OUVC Fund 1 Limited Partnership for Investment (OUVC Fund 1) operated by OSAKA University Venture Capital Co., Ltd. A grant has been awarded to a project of MiCAN Technologies, “Cell development for evaluation of ADE in vaccine development against the novel coronavirus,” under the category of “Technology development project for measures against infectious diseases such as viruses,” one of the programs undertaken by the AMED.
 | UpCell® For collecting cell sheets (temperature responsive) ・Cells can be collected in a form of a sheet. ・This product can be used in 3D culture in which homo-/hetero-cell sheets are piled up. |
 | RepCell ™ For collecting cell sheets (temperature responsive) ・Cells susceptible to trypsin (including various stem cells, dendritic cells, nerve cells, the liver, and macrophages) can be cultured. ・This product can be used in research into surface antigens. ・This product can be used for treatment before flow cytometry analysis. |
 | HydroCell ™ Ultra-low adhesion cell cultureware ・Spheroids can be cultured. ・This product can be used as an alternative to Soft Agar Assay. ・Embryoid bodies of ES cells can be formed. ・The signal transduction of macrophages can be analyzed. |
| |
(Each picture is taken from the company materials)
Although CellSeed manufactured periodontal ligament cell sheets on consignment for investigator-initiated clinical trials (completed in November 2019) in the regenerative medicine consignment business that makes the most of its own Cell Culture Center, the novel coronavirus pandemic has not only put off production of autologous cartilage cell sheets that was consigned by Tokai University, the company’s joint research partner, for the first patient treated in an advanced medical care program, but also cancelled manufacturing of epithelial cell sheets for esophageal regeneration that was planned to be consigned by MetaTech (for clinical trials in Taiwan). The autologous cartilage cell sheets that the CellSeed manufactured on consignment by Tokai University to use in an advanced medical care program was transplanted to the first patient in August 2020.
Cell Sheet Regenerative Medicine Business
Sales were 1 million yen, down 98.3% year on year, and operating loss stood at 185 million yen (127 million yen in the same period of the previous year). The company completed discussion with the PMDA over clinical trials of epithelial cell sheets for esophageal regeneration and will submit a notification of additional clinical trial plans in the fourth quarter of the term ending December 2020; however, CellSeed will need some time to apply for approval for manufacturing and sale of the product, which is expected to be done in 2025, because the company has limited target patients and the PMDA requires a greater number of cases than the initial clinical trial. Therefore, the company will consider adding medical treatment facilities in order to shorten periods needed for clinical trials. Meanwhile, in Europe, medical treatment using endoscopes has not gained popularity as initially expected and thus CellSeed decided on July 16, 2020 that it terminates development of epithelial cell sheets for esophageal regeneration in Europe in order to focus on acquiring approval for manufacturing and sale of the product in Japan. As for cartilage regeneration using autologous cell sheets, a surgery of the first patient under Advanced Medical Care B Program was completed in August 2020 (CellSeed manufactured the product on consignment).
In the overseas business, CellSeed established a joint venture, Up Cell Biomedical Inc. mainly with MetaTech for development, manufacturing, and sale of other pipelines than the products for the esophagus and cartilage.
2-3 Financial condition and Cash flow
Financial Conditions
| December 19 | June 20 |
| December 19 | June 20 |
Cash | 1,065 | 1,282 | Accounts payable | 33 | 32 |
Current assets | 1,245 | 1,376 | Advances received | 30 | 30 |
Fixed assets | 210 | 211 | Liabilities | 110 | 101 |
Total assets | 1,456 | 1,588 | Net Assets | 1,345 | 1,487 |
* Unit: million yen
At the end of the second quarter, total assets were 1,588 million yen, up 132 million yen from the end of the previous term. The 18th share options (with a condition of strike price adjustment) issued in September 2019 were exercised (which was completed on May 25, 2020), increasing cash and shareholder’s equity. Capital adequacy ratio was 92.6% (91.1% at the end of the previous term).
Cash Flow(CF)
| FY12/19 1H | FY12/20 1H | YY Change | |
Operating Cash Flow | -58 | -260 | -202 | - |
Investing Cash Flow | -7 | -5 | +2 | - |
Financing Cash Flow | - | 484 | - | - |
Cash and Equivalents at the end of term | 987 | 1,282 | +295 | +29.9% |
* Unit: million yen
3. Fiscal Year ending December 2020 Earnings Forecasts
3-1 Full year consolidated earnings forecasts
| FY 12/ 19 Act. | Share | FY 12/ 20 Est. | Share | YY Change | Initial Est. | Compared to Initial Est. |
Sales | 275 | 100.0% | - | - | - | 310 | - |
Operating Income | -780 | - | - | - | - | -1,020 | - |
Ordinary Income | -786 | - | - | - | - | -1,020 | - |
Net Income | -782 | - | - | - | - | -1,020 | - |
* unit: million yen
Business forecasts for the full term have not been provided yet.
The global spread of the novel coronavirus infections has led to a delay in general developmental support for the joint venture established in Taiwan in January 2020 and has been restricting activities aimed at forging new business alliances and licensing in Asian countries, such as China and Taiwan. These impacts have caused the company to withdraw the business forecasts released on February 14, 2020 and make it difficult for it to provide new ones now because “it is impossible to perform reasonable calculations at the moment” regarding the full-year consolidated earnings for the term ending December 2020.
The company published a guidance that recommends that all of its employees work from home as much as possible and started a period during which the employees are encouraged to work from home on February 27, 2020. Recommendation to work from home will stay effective until September 30, 2020. CellSeed plans to promote diverse work styles that take into account the circumstances of each employee by flexibly allowing its workers to find a proper balance between working from home and working in office.
3-2 Issuance of 19th share options (with a condition of strike price adjustment)
The company issued the 19th share options (with a condition of strike price adjustment) which were allotted to Barclays Bank PLC on August 6, 2020, and all of the issuance cost (3,780,000 yen) was payed. The number of share options issued was 35,000 (108 yen per share option). The fund that will be raised when all of the share options are exercised is estimated at 1,295,780 thousand yen (estimated net proceeds). The period during which the share options can be exercised is one year (from August 7, 2020 to August 6, 2021). The company plans to use the fund for research and development, and business operation.
3-3 Share Options for Officers and Employees
Believing that corporate value can be enhanced by strengthening each worker’s awareness and motivation of their respective contribution to improving business performance, CellSeed issues the 20th share options for a fee to its directors and the 21st share options for free to its employees.
20th Share Options (for a fee)
Targets, No. of allotees, and No. of share options issued | 4 directors (1,200 share options) |
Total No. of share options issued | 1,200 share options |
Type and No. of share options issued | 120,000 common shares |
21st Share Options (for free)
Targets, No. of allotees, and No. of share options issued | 34 employees (1,159 share options) |
Total No. of share options issued | 1,159 share options |
Type and No. of share options issued | 115,900 common shares |
3-4 2nd Cell Sheet Engineering Innovation Forum has been postponed.
The second Cell Sheet Engineering Innovation Forum was scheduled for October 15, 2020 but has been decided to be postponed in order to prevent the novel coronavirus infections from spreading. A new schedule will be announced on the website of CellSeed as soon as it is confirmed.
4. Conclusions
The full-year business forecasts for the term ending December 2020 were withdrawn and new ones have not been provided yet. MetaTech submitted a notification of clinical trial plans for epithelial cell sheets for esophageal regeneration in Taiwan in 2018 and is preparing for the project. CellSeed was supposed to receive cells of patients from Taiwan and manufacture epithelial cell sheets for esophageal regeneration on consignment in Japan; however, the Covid-19 pandemic has made it impossible to transport biological materials between Japan and Taiwan, and thus, the company decided to manufacture the product in Taiwan. The company is therefore required to submit a new notification of clinical trial plans and is preparing for the submission. Furthermore, Up Cell Biomedical Inc., a joint venture set up in Taiwan in January 2020 through investment mainly by CellSeed and MetaTech, planned to develop, manufacture, and sell pipelines other than ones for the esophagus and cartilage, but developmental support in relation to establishing the business has been delayed generally. In addition, activities aiming at creating new business alliances and licensing in Asian countries, such as China and Taiwan, are also affected adversely by the novel coronavirus spread.
There, however, is a silver lining in this difficult situation. Sales of instrument products marked record highs in the second quarter and the first half of this term. The light is seen at the end of the tunnel regarding submission of a notification of additional clinical trial plans for epithelial cell sheets for esophageal regeneration in Japan. Besides, as a surgery for the first patient under the category of Advanced Medical Care B Program has been postponed due to the Covid-19 pandemic, sales from consignment manufacturing of cartilage cell sheets for the project are expected to be recorded in the second half of the current term. Consequently, the company will see growth in revenue later. The business prospect is becoming rosier little by little. Not only has the issuance of the 19th share options allowed the company to raise funds of slightly less than 1.3 billion yen, but it also has about 1.3 billion yen at hand. There is no concern related to the company’s current financial base over the next few years.
<Reference: Regarding Corporate Governance>
◎Organization type, and the composition of executive directors and auditors
Organization type | Company with company auditor |
Directors | 4 directors, including 2 external ones |
Auditors | 3 corporate auditors, including 2 external ones |
◎Corporate Governance Report(Latest Update:March 31, 2020)
Basic Policy
With the missions to introduce technological innovations, to exert creativity and to contribute to people’s health and welfare by providing high-quality products and services, we are enhancing corporate governance to raise quality in all of our corporate activities.
In the future, we will increase our accountability further to improve the transparency of disclosed information and strengthen our checking system even more.
<Reasons for Non-compliance with the Principles of the Corporate Governance Code (Excerpts)>
CellSeed has stated, “Our company implements all the basic principles stipulated in the Corporate Governance Code as a JASDAQ listed company.”
This report is intended solely for information purposes and is not intended as a solicitation to invest in the shares of this company. The information and opinions contained within this report are based on data made publicly available by the company and obtained from sources that we judge to be reliable. However, we cannot guarantee the accuracy or completeness of the data. This report is not a guarantee of the accuracy, completeness or validity of said information or opinions, nor do we bear any responsibility for the same. All rights pertaining to this report belong to Investment Bridge Co., Ltd., which may change the contents thereof at any time without prior notice. All investment decisions are the responsibility of the individual and should be made only after proper consideration. Copyright(C) 2020 Investment Bridge Co., Ltd. All Rights Reserved. |
To view back numbers of Bridge Reports on CellSeed (7776) and other companies, or IR related seminars of Bridge Salon, please go to our website at the following url: www.bridge-salon.jp/


