Bridge Report:(7776)CellSeed the Fiscal Year ended December 2025
 Setsuko Hashimoto, President | CellSeed Inc. (7776) |
 |
Company Information
Market | TSE Growth Market |
Industry | Precision Instrument (Manufacturing) |
President | Setsuko Hashimoto |
HQ Address | Telecom Center Building, Aomi 2-5-10, Koto-ku, Tokyo |
Year-end | December |
Homepage |
Stock Information
Share Price | Number of shares issued (End of the Term) | Total market cap | ROE Act. | Trading Unit | |
¥328 | 35,557,719 shares | ¥11,662 million | -64.7% | 100 shares | |
DPS Est. | Dividend yield Est. | EPS Est. | PER Est, | BPS Act. | PBR Act. |
¥0.00 | - | ¥-30.51 | - | ¥35.37 | 9.3 x |
*Stock price as of closing on March 9, 2026. Each number is taken from the financial results of Fiscal Year ended December 2025.
Consolidated Earnings Trend (non-consolidated)
Fiscal Year | Sales | Operating Profit | Current Profit | Net Profit | EPS | DPS |
December 2022 | 126 | -743 | -754 | -759 | -36.31 | 0.00 |
December 2023 | 190 | -697 | -710 | -846 | -29.43 | 0.00 |
December 2024 | 193 | -846 | -847 | -859 | -25.72 | 0.00 |
December 2025 | 83 | -1,046 | -1,051 | -1,104 | -31.82 | 0.00 |
December 2026 Est. | 71 | -1,225 | -1,250 | -1,320 | -30.51 | 0.00 |
* The estimates were provided by the company. Units: million yen.
This Bridge Report presents CellSeed Inc.’s earnings results for the Fiscal Year ended December 2025, the interview with President Hashimoto, and other financial details.
Table of Contents
Key Points
1. Company Overview
2. Fiscal Year ended December 2025 Earnings Results
3. Fiscal Year ending December 2026 Earnings Forecasts
4. Interview with President Hashimoto
5. Conclusions
<Reference: Regarding Corporate Governance>
Key Points
- The sales in the fiscal year ended December 2025 were 83 million yen, down 109 million yen year on year. The sales of the cell cultureware business in the regenerative medicine supporting business were sluggish in overseas markets, due to the rapid changes in the research environment, such as the significant reduction of budgets at research institutes in the U.S., and the lingering geopolitical turmoil in Europe, the Middle East, etc. They posted an operating loss of 1,046 million yen, up 199 million yen year on year. SG&A expenses augmented 132 million yen year on year, mainly due to the increase in R&D costs caused by the start of third-phase clinical trial for allogeneic cartilage cell sheets and the registration of cases. They posted a net loss of 1,104 million yen, up 244 million yen year on year. Regarding fixed assets, there was a sign of impairment loss, so they posted an impairment loss of 52 million yen as an extraordinary loss.
- For the fiscal year ending December 2026, sales and profit are projected to decline. Sales are forecast to decrease 12 million yen year on year to 71 million yen, and operating loss is projected to augment 178 million yen year on year to 1,225 million yen. Regarding the regenerative medicine supporting business, the business environment is uncertain, but they aim to keep expanding the sales of mainly cell cultureware, especially outside Japan. Through activities as a Contract Development and Manufacturing Organization (CDMO) for regenerative medicine and comprehensive support regarding regenerative medicine, they promote contract manufacturing for supporting R&D and commercialization of regenerative medicine and so on. The sales in the regenerative medicine supporting business are expected to be 71 million yen. In the cell sheet regenerative medicine business, they will promote the development of allogeneic cartilage cell sheets. R&D costs are forecast to keep augmenting. They will actively negotiate with candidate business partners for licensing out pipelines.
- We interviewed President Setsuko Hashimoto about the competitive advantages of CellSeed, her message toward shareholders and investors, etc. She said, “While it is taking time for our “cell sheet regenerative medicine business” to reach the goal, and we are having our shareholders watch over us, the third-phase clinical trial for allogeneic cartilage cell sheets was launched according to the plan. We cannot quite disclose the progress of the clinical trial at any time due to various rules, but it is progressing as scheduled. On the occasion of the 25th anniversary of the establishment of the company, we set a new mission & vision. We shall make company-wide efforts in order to demonstrate a significant development by 2030, 5 years from now. Therefore, I would be grateful if you could kindly watch over us for a while.”
- Regarding the “cell cultureware business” in the regenerative medicine supporting business, the life science-related budget was reduced significantly, in response to the revision to the national budget under the Trump administration in the U.S., affecting the company significantly. Some items are uncontrollable in house, so they plan to concentrate on popularizing cell sheet engineering, which will defy the common sense regarding cell collection, inside and outside Japan. In January 2026, they concluded a basic contract for transactions regarding cell cultureware with Cuorips Inc. (Growth Market of TSE; 4894), implying that the demand for temperature-responsive cell cultureware product is growing steadily.
- Regarding the “commissioned regenerative medicine business” in regenerative medicine supporting business, they have undertaken the manufacturing of autologous cartilage cell sheets, autologous epithelial cell sheets for children who have undergone surgery for congenital esophageal atresia, and periodontal ligament cell sheets. In 2025, they were entrusted with the production of epithelial cell sheets for esophageal regeneration for uninsured medical treatment conducted by Tokyo Metropolitan Tama-Hokubu Medical Center. They have strengthened their business foundation, by concluding a contract regarding the disclosure of technological information for undertaking the manufacturing of products for clinical trials for personalized dendritic cell vaccines against esophageal cancer, which are being developed as a regenerative medicine product by NPT Co., Ltd., with NPT. We would like to keep an eye on the progress of their initiatives for strengthening their advantages further without relying on only the “cell sheet regenerative medicine business.”
1. Company Overview
[1-1 Management philosophy]
On the occasion of the 25th anniversary of establishment of the company, they set a new mission & vision. The management and employees discussed their ideal state and what are necessary, and determined the contents of their mission & vision. They aim to contribute to the further advance of regenerative medicine, which is spreading around the world.

MISSION | We will bring smiles and hope to the world by harnessing the power of cells. |
VISION | We will create a future of new healthcare based on cell sheet engineering. |
[1-2 What is regenerative medicine?]
Regenerative medicine means a new type of healthcare in which lost, damaged or impaired organs are regenerated and cured with cells, the extracellular matrix, growth factors, etc. It is expected that the regenerative medicine market will emerge through the changes in the environment, such as the aging of the population, technological innovation, and the easing of regulations, so that diseases for which unmet medical needs are high because there have been no effective treatment methods.
“Autologous” and “allogeneic”
There are two kinds of cells used for regenerative medicine: “autologous” and “allogeneic” ones.
“Autologous” cells mean those that are collected from a patient, processed, cultured, and then administered to the patient. Since the cells are from each patient, so the body does not reject the cells, but it takes time to prepare them for administration, because cells are collected and cultured after a treatment method is determined.
“Allogeneic” cells mean those that are collected from a doner, processed, cultured, and then administered to patients. Unlike “autologous” cells, it is unnecessary to collect cells from each patient, and it is possible to store cells and produce cells of the same quality, but there is a risk that the body will reject the cells.
Concrete methods for administering regenerative medicine products include the method of injecting them into blood vessels with a syringe or drip infusion for systemic administration and the method of administering (implanting) products to the body part you want to regenerate through surgery or an endoscope. There are a variety of methods for administering regenerative medicine products.
[1-3 Regenerative medicine of CellSeed]
CellSeed focus on the administration (implantation) of “allogeneic” cells to the body part you want to regenerate through surgery or an endoscope. They operate the “cell sheet regenerative medicine business,” in which they develop products for regenerative medicine, etc. by using “cell sheets (cultured sheet-shaped cells)” produced based on the core technology of the world’s first “cell sheet engineering,” which was developed by the Professor Emeritus Teruo Okano of Tokyo Women's Medical University and transferred to CellSeed.
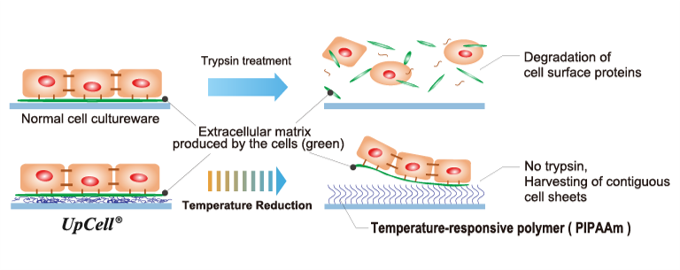
“Cell sheet engineering” – Basic Technologies for Regenerative Medicine
(From the company material)
In general, cells excrete adhesive proteins as tissue serving as a “foothold” or “framework” for filling the gap between cells and facilitating cellular activities, which is called the extracellular matrix, for multiplication. The extracellular matrix conveys information to cells, taking indispensable roles for forming, growing, and repairing tissue. When cells are cultured with ordinary devices, protein hydrolases are used for collecting cells adhering to the devices after multiplication, but cultured cells disintegrate due to the enzyme treatment.
To solve that issue, the Professor Emeritus Teruo Okano of Tokyo Women's Medical University developed “temperature-responsive cell cultureware” under a totally different concept from conventional technologies, and a cell culturing method based on them, cultivating a new field called “cell sheet engineering.” This is a platform technology originating in Japan and the first of its kind in the world.
If cells are cultured on the cell culture dish “UpCell®,” whose surface has been processed with a temperature-responsive polymer, which changes its molecular structure according to temperature, enzyme treatment like conventional technologies is not required for collecting cells, and just by changing culture temperature, it is possible to collect “cell sheets,” which organically integrate without disintegrating, from the culture dish, while retaining the extracellular matrix (adhesive proteins).
The collected cell sheets can be smoothly implanted in living tissue, because adhesive proteins are retained without disintegrating. As another remarkable feature, the intrinsic functions of cells are retained because they do not undergo enzyme treatment.
In addition, cell sheets can be produced from a variety of tissue and organs. It is possible to design applications in various ways, including the use of cell sheets as tissue for implantation, the combined use with those from other cells, and the layering of multiple cell sheets.
Such technology utilizing cell sheets is called “cell sheet engineering.” A total of 26 products for regenerative medicine (including conditions and approval for deadlines) have been approved in Japan as of March 2026. Four out of them are based on cell sheet engineering.
Huge Regenerative Medicine Market
The market size of regenerative medicine is expected to reach 1.1 trillion yen in Japan and 12 trillion yen worldwide in 2040, and a significant economic effect is expected (Source: Japan Agency for Medical Research and Development [AMED], “Final report on the market survey on regenerative medicine and gene therapy in FY 2019”)
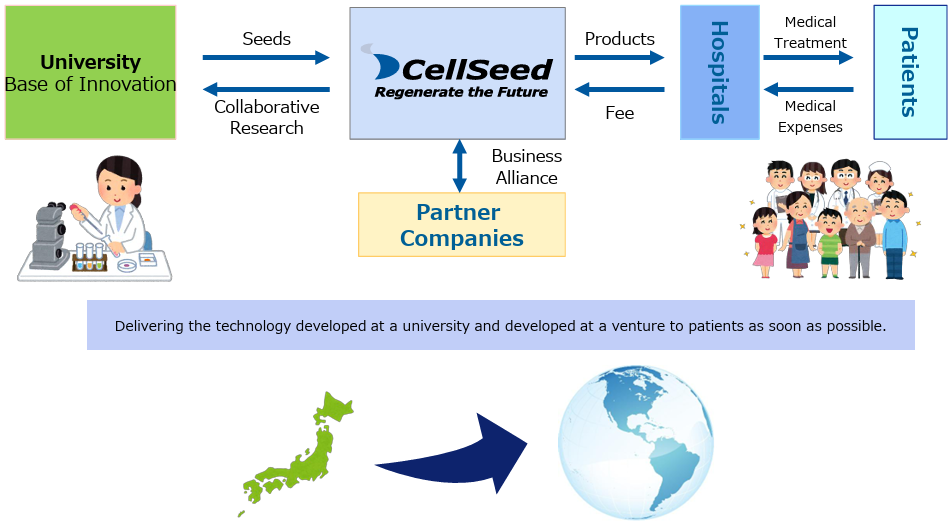
[1-4 Business model of CellSeed]
Based on the outcomes of college research using cell sheets, CellSeed conducts clinical trials and merchandize them as products for regenerative medicine, and deliver them to patients.
(From the company material)
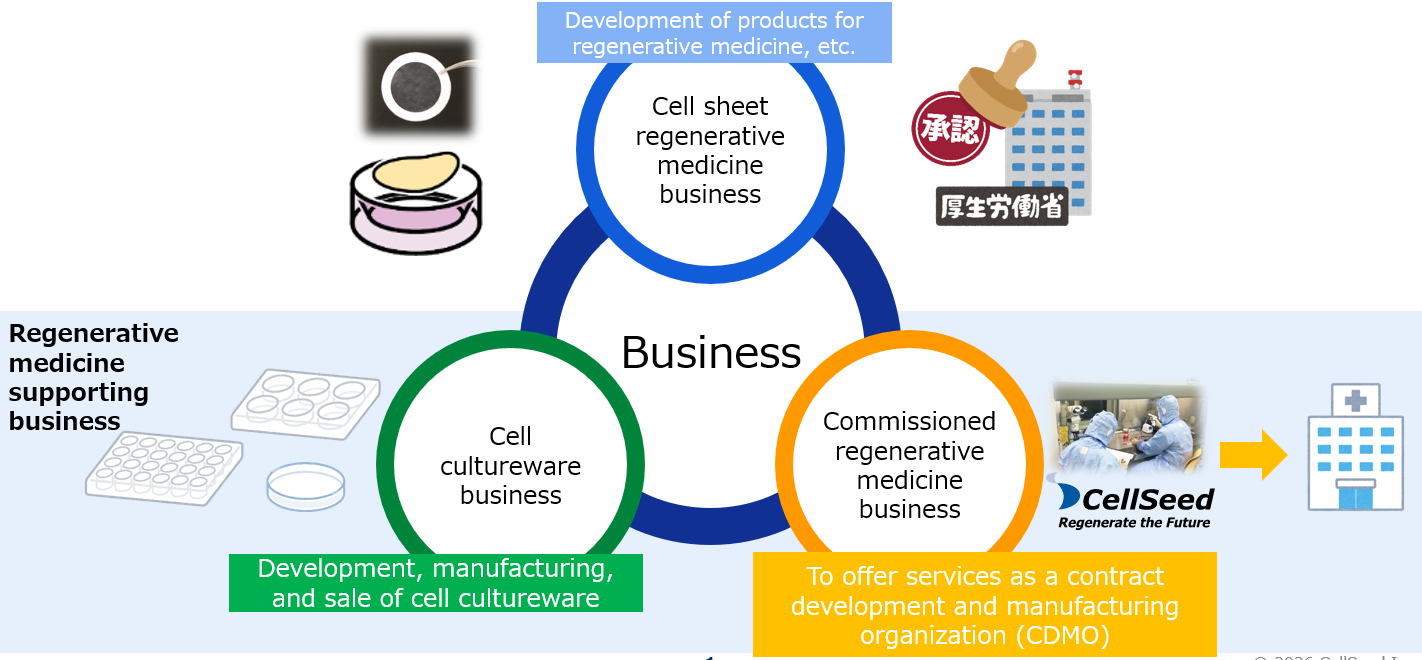
[1-5 Business Description]
The company reports on two segments – “cell sheet regenerative medicine business” and “regenerative medicine supporting business.” The regenerative medicine supporting business is constituted by the “cell cultureware business” and the “commissioned regenerative medicine business.”
(From the company material)
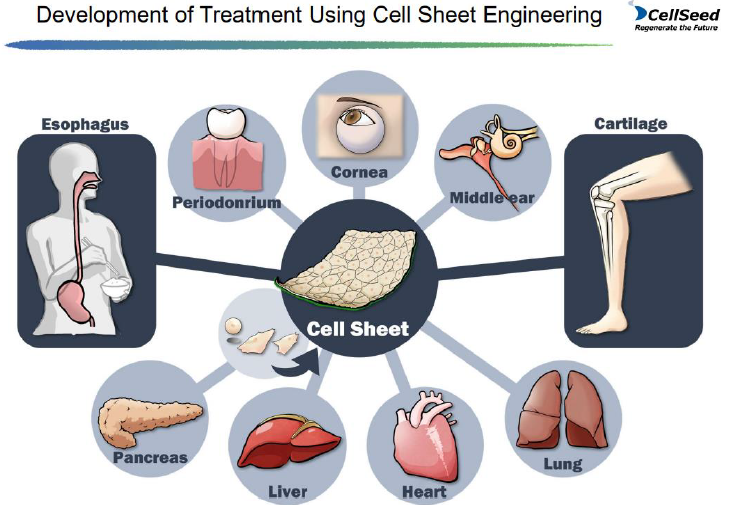
(1) Cell sheet regenerative medicine business
Although treatments based on the fundamental technology of "cell sheet engineering" is being developed in various areas, the company is focusing on "allogeneic cartilage cell sheet" for knee cartilage, and also conducting joint research with Hokkaido University on new treatments for central nerve injury-related diseases.
(From the company material)
◎ Allogeneic cartilage cell sheet
① Overview
“Allogeneic cartilage cell sheets,” which are cell sheets based on the cells of a person other than a patient, was developed by Professor Masato Sato of Tokai University School of Medicine Department of Orthopedics. CellSeed is developing them as products for regenerative medicine, etc. after technological transfer, and currently, it is indicated for knee osteoarthritis.
Knee osteoarthritis is slowly progressing, intractable degeneration of articular cartilage with no fundamental treatment. The number of potential patients in Japan is estimated to be about 30 million, of which about 10 million patients are thought to have subjective symptoms. Furthermore, population aging in Japan is expected to raise the number of patients diagnosed with the illness, making it a disease that needs to be dealt with immediately from the perspective of citizens’ healthy life expectancy and costs of long-term care and medical services. As of now, there are no methods to cure the injury completely, but the “allogeneic cartilage cell sheet” is aimed at regenerating the cartilage surface radically. The cartilage of the knee is called hyaline cartilage, which is hard and excellent in cushioning and abrasion resistance properties, differing from the cartilages of the ear, nose, etc., and it is difficult to regenerate. However, it was confirmed in clinical research in the Tokai University that the “allogeneic cartilage regeneration sheet” can regenerate the cartilage of the knee as hyaline cartilage.
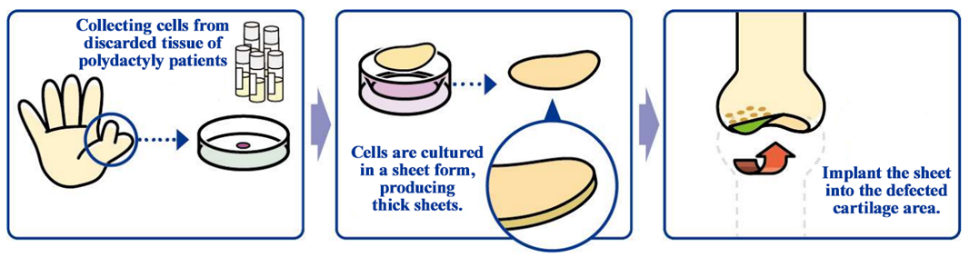
(From the company material)
② Progress of development
Professor Masato Sato performed the world's first implant surgery in 2017, and 10 patients underwent implant surgery in the three years from 2017 to 2019. The treatment with allogeneic cartilage sheets has been adopted in the project for developing evaluation methods, etc. for the industrialization of regenerative medicine (support for acceleration of development of regenerative medicine seeds) of AMED (project period: Oct. 2018 to Mar. 2021).
For developing cartilage cell sheets with allogeneic cells, the discarded tissue of patients with polydactyly who have six fingers, so it is necessary to solve ethical issues, but in December 2020, the company obtained approval for the provision of cartilage tissue collected from patients with polydactyly from Ethical Committee of National Center for Child Health and Development. Then, they established a system for stably supplying cells in Japan.
In July 2021, a research and development project proposed by CellSeed was selected by the Japan Agency for Medical Research and Development (AMED) for the "Development of Basic Technologies for the Industrialization of Regenerative Medicine and Gene Therapy (Project to Promote the Industrialization of Regenerative/Cell Medicine and Gene Therapy)," which is a subsidy project (project period: August 2021-March 2023).
In August 2022, the company reached an agreement with the center on the basic terms of a contract to establish a system for continuous provision of resected tissue during polydactyly surgery and commercialization for industrial use. Thus, it will be possible to continuously receive human tissue supply for clinical trials to manufacture and sell allogeneic cartilage cell sheets.
In April 2022, a patent was granted and registered in Japan for a "tissue regeneration cultured cell sheet, its manufacturing method, and how to use it."
In January 2023, a research group led by Professor Masato Sato of Tokai University confirmed the safety and efficacy of allogeneic cartilage cell sheets implanted into cartilage defects in the knee joints one year after surgery in all 10 patients in a clinical study of knee osteoarthritis that had been conducted since 2017. Their findings were published in the online journal npj Regenerative Medicine, a sister publication of Nature.
In addition, in March 2023, the company presented the results of the above-mentioned AMED project "Development of Basic Technologies for the Industrialization of Regenerative Medicine and Gene Therapy (Project to Promote the Industrialization of Regenerative/Cell Medicine and Gene Therapy)" at the Conference of the Japanese Society for Regenerative Medicine and is accumulating evidence of its effectiveness.
Meanwhile, before the start of clinical trials, the company is working to establish a system to ensure quality, by confirming safety as well as efficacy and establishing quality control and transportation methods for the cell sheets.
For cell banks made from tissues, a total of 20 tests, including cell count, viable cell rate, sterility test, endotoxin test, mycoplasma negative test, virus negative test, and other tests have been conducted to confirm its safety, and regarding cell sheets, 10 tests have been conducted including cell morphology, sheet properties, protein expression, virus negative test, and confirmation of the presence of chromosomal abnormalities.
Based on these results, the company submitted a notification of a clinical trial to the Pharmaceuticals and Medical Devices Agency (PMDA) on September 2023. Then, PMDA finished the 30-day examination for the clinical trial. Negotiations with Tokai University concerning the amounts of future milestone payments based on the progress of clinical trials took time, but an agreement was reached in March 2025. They concluded contracts with facilities where clinical trials will be conducted, developing a system for performing surgery, and the first case was registered in October 2025, and clinical trials are underway as planned in each facility.
(Outline of the third-phase clinical trial)
* Test design: collaboration with multiple facilities, single-blinded, randomized, parallel-group trial
* Subject patients: those who have knee osteoarthritis and are subject to proximal tibial osteotomy
* Target number of cases: 96 (48 cases of proximal tibial osteotomy and 48 cases of proximal tibial osteotomy plus implantation of cell sheets)
* Major evaluation items: reports from patients (Patients evaluate their symptoms and QOL by themselves without help from medical doctors or others.)
* Facilities for conducting the test: a total of 6 facilities; Tokai University Hospital, Yokohama Sekishinkai Hospital, Ebina General Hospital, Juntendo University Hospital, Yokohama City University Medical Center, and Yokohama Minami Kyosai Hospital
Registered patients underwent implantation after examination, etc. and were discharged from a hospital in about 1 month. One year after surgery, fixators are removed. The one year from implantation to the removal of fixators is a follow-up period.
The company is also promoting the patent strategy. The U.S. patent application on “tissue regeneration culture cell sheets, manufacturing methods, and utilization methods thereof” related allogeneic cartilage cell sheets was accepted, and the patent will be granted. This is the outcome of joint research on allogeneic cartilage cell sheets carried out with Tokai University.
As the commercial distribution for business, two types are assumed – cases where the company itself takes care of manufacturing and sale, and cases where partners sell products manufactured by the company.
(2) Regenerative medicine supporting business
The regenerative medicine support business consists of the commissioned regenerative medicine business, which conducts the development of manufacturing methods, contract manufacturing, facility management, application support, and consulting for cell sheet products, and the cell cultureware business, which develops, manufactures, and sells cell cultureware such as UpCell®, RepCell®, and HydroCell®.
① Commissioned regenerative medicine business
Regarding regenerative medicine, in accordance with the law for promotion of regenerative medicine, the government enforced “the act for securing the safety of regenerative medicine, etc.,” which facilitates the R&D in academia with clinical research, advanced medicine, and medical treatments at patients’ own expense, and “the act for pharmaceutical products and medical devices, etc.,” which specifies the procedures for acquiring the approval for manufacturing and sale after non-clinical tests and clinical trials. In reality, there exists a significant gap between the R&D in academia and the approval for manufacturing and sale.
Through the service of undertaking projects for regenerative medicine, they aim to solve issues and close gaps in academia as a Contract Development and Manufacturing Organization (CDMO) for regenerative medicine, and serve as a bridge for offering regenerative medicine to patients.
The company is mainly engaged in contract development and manufacturing of cell sheets for pharmaceutical companies and research institutions. The company employs several clinical culturists certified by the Japanese Society for Regenerative Medicine, and its staff with extensive culture experience develop manufacturing methods for regenerative medicine and other products and manufacture them at its cell culture center, which is licensed to manufacture specified cell processed products and regenerative medicine products.
The company obtained the permit for manufacturing specific processed cells (facility numbe FA3160008) in March 2017 and the permit for manufacturing products for regenerative medicine, etc. in October 2018.
In addition, the company provides support for preparing applications for regulatory approval, obtaining manufacturing and marketing licenses, and training engineers for all stages, from product development to manufacturing and marketing.
Major commissioned projects so far
* Autologous cartilage cell sheets
Autologous cartilage cell sheets were approved in January 2019 as Advanced Medical Treatment B under the Law for Ensuring Regenerative Medicine Safety. Tokai University started Advanced Medical Treatment B in 2020. CellSeed started undertaking the production of autologous cartilage cell sheets, and had continuously undertaken it until 2024.
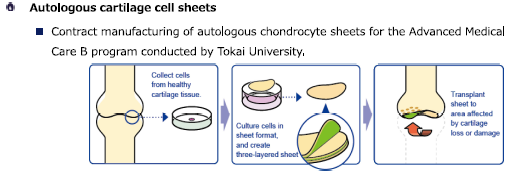
(From the company material)
In May 2023, the company contracted with Ikegami General Hospital to manufacture cell sheets for implantation of autologous cartilage cell sheets for knee joint cartilage damage. As part of its regenerative medicine and other new medical treatment initiatives, Ikegami General Hospital will provide regenerative medicine to patients with damaged knee osteoarthritis due to trauma or osteoarthritis by culturing their cartilage cells in sheet form and applying them to damaged cartilages in the knee joints to improve pain and joint function. Regenerative medicine treatment is provided at the expense of patients who are not eligible for the advanced medical treatment (cartilage regeneration therapy using autologous cell sheets) provided by Tokai University Hospital, as well as patients from overseas.
Ikegami General Hospital has submitted the provision plan necessary for the implementation of regenerative medicine to the Ministry of Health, Labour and Welfare, which has been accepted, and it is now preparing the patient acceptance system. CellSeed also expects to expand its contract manufacturing business by undertaking the manufacturing of cell sheets to be used in self-funded medical treatments.
* Pediatric autologous epithelial cell sheet*
They undertake the manufacturing of autologous epithelial cell sheets for regenerative medicine targeted at children who have undergone surgery for congenital esophageal atresia.
* Periodontal ligament cell sheets
This is the first case of contract manufacturing of cell sheets for use in investigator-initiated clinical trials.
* Epithelial cell sheets for esophageal regeneration
Regarding cell sheets used to cure esophageal stenosis following the treatment of esophageal cancer at Tokyo Metropolitan Tama-Hokubu Medical Center of Tokyo Metropolitan Hospital Organization, CellSeed has supported procedures at relevant authorities which are necessary for launching self-funded medical treatments. CellSeed is scheduled to be entrusted by the center with the manufacturing of cell sheets to be used in the self-funded medical treatments of esophageal stenosis based on epithelial cell sheets for esophageal regeneration.
Epithelial cell sheets for esophageal regeneration are used for treating patients who have just undergone endoscopic submucosal dissection (ESD) and intractable stenosis after ESD.
* Other contracted projects
In August 2025, CellSeed concluded a contract with NPT Co., Ltd concerning technology disclosure, etc. in preparation for undertaking the manufacturing of products to be used in clinical trials for personalized dendritic cell vaccine targeting esophageal cancer, which is developed by NPT as a regenerative medicine product. NPT has submitted a notification on a plan for Phase I/II clinical trials (corporate clinical trial) for dendritic cell vaccine used to treat esophageal cancer to the PMDA, which has been filed by PMDA. NPT is currently proceeding with preparations for the clinical trial, introducing new devices to be used in the manufacturing process of the vaccine. Following the disclosure of technical information from NPT based on this contract, CellSeed has examined technologies and information concerning the manufacturing and management of the vaccine to prepare for concluding a contract with NPT for manufacturing products to be used in clinical trials for the vaccine.
In the commissioned regenerative medicine business, the company also concentrate on the education of engineers in cell culture. In Aomi Cell Culture Innovation Center, they hold training for users of UpCell®, giving lectures on how to produce cell sheets and exfoliate cells.
② Cell cultureware business
The temperature-responsive cell cultureware invented by Professor Emeritus Okano of Tokyo Women's Medical University in 1989 can detach cells simply by lowering the temperature, making it possible to collect intact cell sheets for the first time in the world.
Temperature-responsive cell cultureware is sold worldwide, and research and development of treatment methods using cell sheets are being actively pursued by many researchers.
Until now, the company has developed and supplied various equipment products according to user needs such as universities, research institutes, and pharmaceutical companies, and in September 2022, the company starts to sell new products, UpCell® flasks. In 2025, they manufactured the product for selling it outside Japan, and posted overseas sales.
UpCell® is a device that can collect intact cells in a sheet without using enzymes that damage cells by fixing a temperature-responsive polymer to the surface of the device. The company also plans to sell a product with a larger culture area than the conventional UpCell® dish, enabling the collection of larger amounts of undamaged cells, which will be optimal in research related to immunology and cell therapy. It is possible to recover cell sheets that maintain a higher level of biological functions through cell culturing that is close to the biological environment, such as co-culturing using the UpCell® 6-well cell culture inserts.
The company expects to meet new demand for the mass cultivation of cells for research purposes to develop preventive and therapeutic methods for various infectious diseases and cancer diseases and anticipates medium/long-term business growth.
In December 2022, the temperature-responsive cell culture device product "UpCell® ADVANCE" was registered in the Master Files for Devices (MAF) of the U.S. Food and Drug Administration (FDA).
MAF is a system under which a supply manufacturer registers its corporate information, manufacturing know-how, and other trade secrets and various data as MAF with the FDA in advance. This enables drug and medical device manufacturers to apply for marketing approval from the FDA simply by quoting the MAF number.
Although the completion of MAF registration does not necessarily mean that the FDA has finished confirming or evaluating the quality and safety of the product, the MAF registration is expected to contribute to the promotion of "UpCell® ADVANCE" because it will no longer be necessary for drug and medical device manufacturers to request from CellSeed to submit confidential information when they file applications with the FDA for products using "UpCell® ADVANCE."
◎ Main facilities and equipment
* Cell Culture Center
The cell sheets used for advanced medicine are cultivated at the cell culture center of CellSeed on commission.
With a floor space of about 763 square meters, the Cell Culture Center is equipped with an automated monitoring system that controls the cleanliness, room pressure, temperature and humidity, and operational status of equipment (such as incubators and reagent stockers), and a surveillance camera system throughout the entire facility. Besides, the facility is only twenty-minute drive from Haneda International Airport, making it possible and easy to transport products by air.

(From the company material)
* Aomi Cell Culture Innovation Center
The full-scale operation started in September 2021. The company develops and manufactures cell culture equipment, including laboratory flask products, etc.
[1-6 Growth Strategy for the Regenerative Medicine Supporting Business]
The company's two main growth strategies are “Business expansion of cell culture equipment” and “Promotion of business cooperation for global development.”
(1) Business expansion of cell culture equipment
In 1989, Professor Emeritus Okano of Tokyo Women's Medical University invented temperature-responsive cell culture equipment that can exfoliate cells simply by lowering temperature as described above, making it possible to recover intact cell sheets for the first time, leading to the advancement of research and development of treatment methods using the cell sheet by many researchers.
While the sales of products for the R&D phase, which are intended for the application to regenerative medicine, are growing steadily, the sales of products for the new purpose of culturing a large number of cells for research are increasing rapidly, mainly overseas. In 2020, the company’s equipment business exceeded 100 million yen in sales for the first time. Currently, overseas sales account for approximately 80% of overall sales.
For further growth, they are concentrating on the development of products for offering solutions for meeting the needs in new markets, including the construction of facilities for developing and manufacturing cell cultureware, in addition to the handling of products in the conventional regenerative medicine market.
In September 2021, the company established a new development and manufacturing facility exclusively for cell culture equipment products. In December 2022, the temperature-responsive cell cultureware product "UpCell® ADVANCE" was registered in the Master Files (MAF) for Devices of Food and Drug Administration (FDA). In addition, “UpCell Flask,” a product suited for the culture and collection of a large number of cells related in Japan in 2022, is scheduled to be released outside Japan, too, in 2026.
In recent years, many efforts have been made to manufacture biopharmaceuticals using cells cultured in large quantities, perform immunotherapy using the cells themselves, and solve food and environmental problems.
However, with proteolytic enzymes, that is, a commonly used cell recovery technique, cells are recovered in a damaged state, and it is difficult to completely maintain the original functions and components of the cells. On the other hand, by adopting this product, it is possible to recover cells without damage, and it is expected to greatly contribute to improvement of industrial efficiency and effectiveness in the new market, because all functions and components of cells are maintained.

(From the company material)
In this situation, the sales of the cell cultureware business grew 3.5 times between 2016 and 2024, hitting a record high in the fiscal year ended December 2024. In the fiscal year ended December 2025, overseas sales slowed down due to the rapid changes in the research environment, such as the significant reduction of budgets at research institutes in the U.S., and the lingering geopolitical turmoil in Europe, the Middle East, etc. In the fiscal year ending December 2026, it is unlikely that the business environment will improve, but they will continue efforts in this domain, where they can use their competitive advantages.
(2) Promotion of business cooperation for global development
Overseas sales account for about 80% of sales in this business. The company is also strengthening the sales structure to further expand the overseas sales channels. The company has extended the sales contract with Thermo Fisher Scientific, the alliance partner for expanding sales of equipment products overseas, to further strengthen collaboration, provide consistent quality and service, and further enhance customer satisfaction. In order to achieve this goal, the company has built a quality management system and obtained ISO9001:2015 certification, an international standard, in January 2020.
In addition, aiming for global expansion, the company has been promoting business alliances by participating in exhibitions held not only in Japan but also in other Asian countries and Europe, such as a presentation at “Translate! 2021 – Metrics and Milestones of Success” held in Berlin in January 2021. The company aims to find business partners by participating in exhibitions held in various regions.
2. Fiscal Year ended December 2025 Earnings Results
[2-1 Non-Consolidated Earnings]
| FY 12/24 | FY 12/25 | YoY | Forecast Ratio |
Sales | 193 | 83 | -109 | +3 |
Gross Income | 110 | 42 | -67 |
|
SG&A | 956 | 1,088 | +132 |
|
R&D | 573 | 720 | +146 |
|
Operating Profit | -846 | -1,046 | -199 | -6 |
Ordinary Profit | -847 | -1,051 | -204 | -11 |
Net Income | -859 | -1,104 | -244 | -9 |
* Unit: million yen.
Sales were 83 million yen, down 109 million yen year on year. Sales on the overseas market were sluggish in the cell cultureware business, which falls under the regenerative medicine supporting business. They posted an operating loss of 1,046 million yen, up 199 million yen year on year. SG&A expenses augmented 132 million yen year on year, mainly due to the increase in R&D costs caused by the start of third-phase clinical trial for allogeneic cartilage cell sheets and the registration of cases. They posted a net loss of 1,104 million yen, up 244 million yen year on year. Having observed signs of impairment of fixed assets, the company posted an impairment loss of 52 million yen as an extraordinary loss.
[2-2 Segment trends]
| FY 12/24 | FY 12/25 | YoY |
Regenerative medicine supporting business | 192 | 81 | -110 |
Cell sheet regenerative medicine business | 1 | 1 | +0 |
Sales | 193 | 83 | -109 |
Regenerative medicine supporting business | -20 | -104 | -84 |
Cell sheet regenerative medicine business | -595 | -722 | -127 |
Adjustments | -230 | -218 | +11 |
Operating income | -846 | -1,046 | -199 |
* Unit: million yen.
Regenerative medicine supporting business
Sales were 81 million yen, and operating loss was 104 million yen (Sales were 192 million yen, and operating loss was 20 million yen in the previous fiscal year).
◎ Cell cultureware business
The company has worked on further strengthening the collaboration with existing distributors to expand the sales of devices as an initiative in the Japanese market. With regard to promotional activities, the company set up a booth at exhibitions held at the Japanese Society for Regenerative Medicine, the Pharmaceutical Society of Japan, the Japanese Society of Toxicology, and the Japanese Society for Cultivated Food by which they collected information and engaged in proactive activities to boost the sale of devices. As initiatives for overseas markets, they manufactured UpCell® flasks for selling them outside Japan and posted sales. On the other hand, overseas sales declined, due to the rapid changes in the research environment, such as the significant reduction of budgets of research institutes in the U.S., and the lingering geopolitical turmoil in Europe, the Middle East, etc.
◎ Commissioned regenerative medicine business
With regard to cell sheets used to cure esophageal stenosis following the treatment of esophageal cancer at Tokyo Metropolitan Tama-Hokubu Medical Center, CellSeed supported the completion of necessary procedures for starting uninsured healthcare, including the preparation of related documents when a provision plan required for conducting regenerative medicine was submitted to the Ministry of Health. In August 2025, they concluded a contract regarding the disclosure of technological information for undertaking the manufacturing of products for clinical trials for personalized dendritic cell vaccines against esophageal cancer, which are being developed by NPT Co., Ltd. as products for regenerative medicine, with NPT, and posted some sales related to it.
Cell sheet regenerative medicine business
Sales were 1 million yen, and operating loss was 722 million yen (Sales were 1 million yen, and operating loss was 595 million yen in the previous fiscal year).
Regarding allogeneic cartilage cell sheets, the first case was registered in the third-phase clinical trial in October 2025, as they proceeded with preparations for starting clinical trials, including the establishment of systems at each facility for conducting clinical trials. Some facilities for conducting clinical trials were added, and the third-phase clinical trial is underway as planned. In November 2025, they hosted “the 4th Cell Sheet Engineering Innovation Forum.” The forum was attended by over 100 people from outside the company, and ended on a high note, improving the popularity of “cell sheet engineering.”
Regarding business alliances, the company engages in discussions for forming business alliances and concluding collaborative research contracts with multiple companies in Japan and overseas, for accelerating commercialization and the sale of allogeneic cartilage cell sheets.
[2-3 Financial Condition]
◎ Summary of BS
| December 2024 | December 2025 | Increase/ decrease |
| December 2024 | December 2025 | Increase/ decrease |
Current Assets | 2,312 | 1,521 | -790 | Current Liabilities | 103 | 248 | +144 |
Cash | 2,134 | 1,318 | -815 | ST Borrowings | 10 | 27 | +16 |
Receivables | 37 | 6 | -30 | Fixed Liabilities | 166 | 139 | -27 |
Inventories | 45 | 78 | +32 | LT Borrowings | 132 | 105 | -27 |
Fixed assets | 120 | 134 | +13 | Total Liabilities | 269 | 387 | +117 |
Total Assets | 2,433 | 1,655 | -777 | Net Assets | 2,163 | 1,268 | -894 |
* Unit: million yen |
|
|
| Total Liabilities and Net Assets | 2,433 | 1,655 | -777 |
Total assets decreased 777 million yen from the end of the previous fiscal year to 1,655 million yen, mainly due to the decrease of cash and deposit.
Total liabilities augmented 117 million yen from the end of the previous fiscal year to 387 million yen due to an increase in accounts payable, etc.
Net assets decreased 894 million yen from the end of the previous fiscal year to 1,268 million yen due to the posting of net loss, etc.
The capital adequacy ratio decreased 12.5 percentage points from the end of the previous fiscal year to 76.0%.
[2-4 Topics]
(1) CellSeed issued share acquisition rights through third-party allotment.
CellSeed issued the 25th share acquisition rights (with Exercise Price Adjustment Clause) with the allotment date being December 8, 2025, and the allottee being Barclays Bank PLC.
The exercise period is to be two years (from December 9, 2025 to December 8, 2027), and the amount of funds raised will be about 3.2 billion yen.
About 2 billion yen of the funds raised will be used for research and development projects, about 500 million yen will be allocated to the operation of the business of cell cultureware, and about 700 million yen will be used as operating capital.
(2) CellSeed entered into a basic transaction agreement regarding cell cultureware
In January 2026, CellSeed entered into a basic transaction agreement regarding cell cultureware with Cuorips Inc. (the Growth Market of Tokyo Stock Exchange; Securities Code: 4894).
In April 2025, Cuorips Inc. submitted to the Ministry of Health, Labour and Welfare an application for the approval for marketing of regenerative medicine products for the production of the human (allogeneic) iPS cell-engineered cardiac tissue sheet indicated for severe heart failure resulting from ischemic cardiomyopathy (and obtained a conditional and time-limited marketing approval in March 2026).
This agreement between the two companies is aimed at stable provision of a temperature-responsive cell cultureware product (UpCell® ADVANCE), which is used in producing the sheet, to Cuorips Inc. from CellSeed Inc.
(3) CellSeed decided to adopt Hitachi’s platform for integrated value chain management for regenerative medicine products.
In December 2025, toward the commercial production of regenerative medicine products, CellSeed decided to adopt Hitachi Value Chain Traceability service for Regenerative Medicine, a platform for integrated value chain management for regenerative medicine products offered by Hitachi Ltd., in the third-phase clinical trial of the allogeneic cartilage cell sheet with the aim of enhancing the efficiency of the tasks involved in the clinical trial and fostering smooth cooperation with the facilities participating in the clinical trial project.
(Overview of Hitachi Value Chain Traceability service for Regenerative Medicine)
Hitachi Value Chain Traceability service for Regenerative Medicine is a platform technology offered by Hitachi Ltd. for managing cell and traceability information in an entire value chain of regenerative medicine products. The service is Japan’s first common service infrastructure relating to regenerative medicine products that is available to all stakeholders involved in a value chain (including medical institutions, pharmaceutical companies, logistics companies, and manufacturers).
(Purpose and effects of the adoption of the service)
As the first step, among the functions available in Hitachi Value Chain Traceability service for Regenerative Medicine, CellSeed will adopt the function of case registration management for the facilities involved in the clinical trial. Then, as the second and subsequent steps, the company intends to establish a system for strictly controlling the quality and ensuring information traceability because the GCTP Ministerial Ordinance (*) requires a system for making sure that all the processes between the raw material sourcing and the shipment of final products can be traced as regenerative medicine products are produced using cells and tissues derived from donors.
*GCTP Ministerial Ordinance
Ministerial Ordinance on Standards for Manufacturing Control and Quality Control of Regenerative Medicine Products
(4) CellSeed hosted the 4th Cell Sheet Engineering Innovation Forum.
CellSeed hosted the first Cell Sheet Engineering Innovation Forum in 2019 with the aim of encouraging more researchers to learn about the world’s first cell sheet engineering originally developed in Japan. The fourth Cell Sheet Engineering Innovation Forum was held in November 2025 and facilitated communication and discussion regarding regenerative medicine and its application to new fields through such activities as presentations on innovative ideas especially by young researchers who are engaged in peripheral technologies.
Speakers
・Tatsuya Shimizu (Director and Professor of the Institute of Advanced Biomedical Engineering and Science Tokyo Women’s Medical University)
“Aiming at a new development of cell sheet engineering”
・Akihiro Umezawa (Director of the Research Institute at the National Center for Child Health and Development)
“Cell sheet engineering for childhood diseases”
・Yasushi Fuchimoto (Senior Professor of the Department of Pediatric Surgery, Faculty of Medicine, at International University of Health and Welfare)
“Regenerative treatment by transplanting autologous oral mucosal epithelial cell sheet for childhood esophageal stenosis”
・Hidetoshi Masumoto (Specific Professor of the Department of Cardiovascular Surgery at Kyoto University Hospital)
“Outlook for the next-generation cardiovascular medicine with cardiac organoids based on the cell sheet technology”
In addition to the presentations given by the professors above, CellSeed granted the best poster presentation award to a presenter and the outstanding poster presentation award to three presenters for the presentations that showed notable achievements or future potential in fields such as basic research and new applications of cell sheets.
3. Fiscal Year ending December 2026 Earnings Forecasts
[3-1 Earnings forecasts]
| FY 12/25 Act. | FY 12/26 Est. | YoY |
Sales | 83 | 71 | -12 |
Operating Income | -1,046 | -1,225 | -178 |
Ordinary Income | -1,051 | -1,250 | -198 |
Net Income | -1,104 | -1,320 | -215 |
* Unit: million yen
Both sales and profit are expected to decrease.
Sales are forecast to decrease 12 million yen year on year to 71 million yen, and operating loss is projected to augment 178 million yen year on year to 1,225 million yen.
Regarding the regenerative medicine supporting business, the business environment is uncertain, but they aim to keep expanding the sales of mainly cell cultureware, especially outside Japan. Through activities as a Contract Development and Manufacturing Organization (CDMO) for regenerative medicine and comprehensive support regarding regenerative medicine, they promote contract manufacturing for supporting R&D and commercialization of regenerative medicine and so on. The sales in the regenerative medicine supporting business are expected to be 71 million yen.
In the cell sheet regenerative medicine business, they will promote the development of allogeneic cartilage cell sheets. R&D costs are forecast to keep augmenting. They will actively negotiate with candidate business partners for licensing out pipelines.
[3-2 Significant Events Related to Going Concern]
The balance of cash on hand (cash and deposits) as of the end of December 2025 was 1,318 million yen. By exercising share acquisition rights, they procured funds amounting to 400 million yen in January 2026. There remain a significant number of share acquisition rights unexercised, so they consider that their financial standing will be free from cash-flow problems for the foreseeable future.
On the business side, however, the company has not yet been able to show the path to the early commercialization of its first cell sheet regenerative medicine product, which is a significant issue in the cell sheet regenerative medicine business. The company has determined that, as of the end of December 2025, there continue to be circumstances that raise substantial doubt on the going concern assumption.
The company will proceed with the development of allogeneic cartilage cell sheets and try to seize earning opportunities by commercializing the first product for cell sheet-based regenerative medicine as soon as possible and finding business alliance partners, to improve the above situation.
4. Interview with President Hashimoto
We interviewed the President Setsuko Hashimoto about the competitive advantage of CellSeed, her message toward shareholders and investors, etc.
After graduating from the Department of Biology, Faculty of Science of Kyushu University, President Hashimoto studied abroad at Dartmouth College, completed the master’s degree at the Department of Biology, Graduate School of Science at Kyushu University, and earned her doctorate at Heidelberg University, following which she engaged in research into genetically engineered pharmaceuticals and the construction of a plant for manufacturing pharmaceuticals as a researcher at a foreign-owned pharmaceutical company. Afterward, she worked in marketing of equipment and reagents for research. As a result, she did a one eighty from a researcher using such products to a provider of these products. However, she was consequently able to discover an interesting side of business by engaging in marketing as somebody who understands the needs of researchers, making use of the knowledge and experience she had built up until then. At a Swedish start-up company, she was involved in the establishment of the Japanese corporation as well as developing the market in Japan and Asia, responsible for marketing. In 2009, she was appointed as head investment officer in charge of life science at the Investment Office, the Embassy of Sweden. As she facilitated a mutual connection between the Karolinska Institute (Sweden), famous for awarding the Nobel Prize in Physiology or Medicine, and the Institute of Advanced Biomedical Engineering and Science Tokyo Women’s Medical University, joint research on “cell sheet engineering” by these two parties was launched. This encounter led to her being invited to CellSeed, which faced hardships in terms of business despite receiving the “cell sheet engineering” technology from Tokyo Women’s Medical University. She joined the company in March 2014 as a director and vice-president, and was appointed as representative director and president in June of the same year, demonstrating leadership in aiming for the commercialization of the cell sheet engineering technology and success of the second business startup.
Q: Please elaborate on the competitive advantage of CellSeed, which is a bio start-up company.
When cells proliferate, they generally secrete adhesive proteins, tissue that fill in the gap between separate cells and become the “scaffolding” and “framework” for cell activity, called the extracellular matrix, on their own. The extracellular matrix transmits information to cells and fulfils a role indispensable for the formation, growth and restoration of the tissue. When cultivating cells in regular cell cultureware, proteolytic enzymes are used to collect proliferated cells attached to the surface of the cultureware, but this ends up disintegrating the cells.
In contrast, the Professor Emeritus Teruo Okano at Tokyo Women’s Medical University developed a “temperature-responsive cell cultureware,” based on a concept completely different from conventional technologies, and a method for cultivating cells that uses this cultureware, opening up a new field of cell sheet engineering. This is a platform technology developed in Japan, and the first of its kind in the world.
“UpCell®” is a cell culture dish with a temperature-responsive polymer, which changes its molecular structure depending on the temperature, and requires no enzyme treatment for the collection of cultivated cells. Merely changing the temperature is sufficient for recovering an intact “cell sheet,” while maintaining organic junction and the extracellular matrix (adhesive proteins).

This “cell sheet engineering” is a highly original and groundbreaking technology for regenerative medicine, and CellSeed was founded to receive this technology in order to commercialize it. As almost no competition can be seen at this point, the company harbors an extremely robust competitive advantage.
Moreover, CellSeed not only develops regenerative medicine products using cell sheets, but also engages in “cell cultureware business,” manufacturing and selling cultureware for producing sheets, and “commissioned regenerative medicine business,” which consists of the development of manufacturing methods, contract manufacturing, facility management, application support and consulting for cell sheet products, under the “regenerative medicine supporting business.” This significantly differentiates our company from other bio start-ups which operate business dependent solely on drug discovery.
Q: What is necessary for further refinement of this competitive advantage, leading up to commercialization?
While “cell sheet engineering” is a unique and beneficial technology, when it comes to commercialization, it is necessary to concretely show in which part of the body and in what kind of way it will be useful in the actual medical scene, such as in case of the esophagus or in case of the cornea. I believe that the key lies in how many such cases we can demonstrate.
Therefore, we focus our efforts on a symposium called “Cell Sheet Engineering Innovation Forum,” which we organize biennially, introducing only our technologies.
The number of participants is growing every year, and we have received reports on a variety of achievements from researchers and clinicians. Moreover, there are more and more researchers who try to use the technology in other fields after reading such reports, and this is inducing synergy. We also focus on poster announcement and public recognition in order to support younger researchers.
It goes without saying that securing excellent human resources is also vital for enhancing our competitive advantage and achieving successful commercialization.
As there is an enormous difference between succeeding in producing cultureware at the level of a graduate school, and running a company that continues to produce cultureware of consistent quality, personnel able to manage research and development as well as production is required. Furthermore, in order to acquire approval from the Ministry of Health, Labour and Welfare, it is necessary to secure professionals in respective fields, such as cell research, data management, clinical trials, and manufacturing.
As our company’s technology is a completely new one, our policy lies in recruiting persons keen on taking on a new challenge and academic professionals interested in helping patients from a business direction.
Q: Please elaborate on the latest situation and the future development of the regenerative medicine supporting business.
As for “cell cultureware business” in the regenerative medicine supporting business, the life science-related budget was significantly curtailed as a result of revisions made to the national budget by the Trump government in the U.S., which gravely impacted our cell cultureware business as well. While there are aspects we unfortunately cannot control, we will endeavor to popularize our new method for cell collection, which would upturn common knowledge, both in Japan and overseas.
In the “commissioned regenerative medicine business” in the regenerative medicine supporting business, we have previously been entrusted with the manufacturing of autologous cartilage cell sheets, and autologous epithelial cell sheets and periodontal ligament cell sheets for children after surgery to treat congenital esophageal atresia. However, in 2025 we were not only commissioned with the manufacturing of epithelial cell sheets for esophageal regeneration used in self-funded medical treatment offered by Tokyo Metropolitan Tama-Hokubu Medical Center, but also concluded a contract pertaining to technology disclosure, etc. as a step toward commissioned manufacturing of clinical trial products for individualized dendritic cell vaccine used to treat esophageal cancer, researched as a product for regenerative medicine, etc., by NPT CO., Ltd and NPT. Now that our business foundations have been further cemented, we shall go on to further broaden the scope of our business.
Q: Lastly, we would like to ask you for a message toward shareholders and investors.
While it is taking time for our “cell sheet regenerative medicine business” to reach the goal, and we are having our shareholders watch over us, the third-phase clinical trial for allogeneic cartilage cell sheets was launched according to the plan. We cannot quite disclose the progress of the clinical trial at any time due to various rules, but it is progressing as scheduled.
On the occasion of the 25th anniversary of the establishment of the company, we set a new Mission & Vision. While companies in Japan possess many outstanding technologies, they unfortunately tend to avoid taking a risk and venturing overseas. By spreading “cell sheet engineering” abroad as well, we hope to contribute to resolving such challenges faced by Japanese companies.
While striving to expand the “regenerative medicine supporting business,” we shall make company-wide efforts in order to demonstrate a significant development by 2030, 5 years from now. Therefore, I would be grateful if you could kindly watch over us for a while.
5. Conclusions
Regarding the “cell cultureware business” in the regenerative medicine supporting business, the life science-related budget was reduced significantly, in response to the revision to the national budget under the Trump administration in the U.S., affecting the company significantly. Some items are uncontrollable in house, so they plan to concentrate on popularizing cell sheet engineering, which will defy the common sense regarding cell collection, inside and outside Japan. In January 2026, they concluded a basic contract for transactions regarding cell cultureware with Cuorips Inc. (Growth Market of TSE; 4894), implying that the demand for temperature-responsive cell cultureware product is growing steadily.
Regarding the “commissioned regenerative medicine business” in regenerative medicine supporting business, they have undertaken the manufacturing of autologous cartilage cell sheets, autologous epithelial cell sheets for children who have undergone surgery for congenital esophageal atresia, and periodontal ligament cell sheets. In 2025, they were entrusted with the production of epithelial cell sheets for esophageal regeneration for uninsured medical treatment conducted by Tokyo Metropolitan Tama-Hokubu Medical Center. They have strengthened their business foundation, by concluding a contract regarding the disclosure of technological information for undertaking the manufacturing of products for clinical trials for personalized dendritic cell vaccines against esophageal cancer, which are being developed as a regenerative medicine product by NPT Co., Ltd., with NPT. We would like to keep an eye on the progress of their initiatives for strengthening their advantages further without relying on only the “cell sheet regenerative medicine business.”
<Reference: Regarding Corporate Governance>
◎ Organization type, and the composition of executive directors and auditors
Organization type | Company with audit and supervisory committee |
Directors (excluding audit and supervisory committee members) | 3 directors, including no external ones(including no independent ones) |
Auditors and supervisory committee members | 3 committee members, including 3 external ones (including an independent one) |
◎ Corporate Governance Report (Latest Update:April 7, 2025)
Basic Policy
With the missions to introduce technological innovations, to exert creativity and to contribute to people’s health and welfare by providing high-quality products and services, we are enhancing corporate governance to raise quality in all of our corporate activities. We will implement timely and appropriate information disclosure to ensure the transparency of decision-making and to enhance accountability. We will also improve our business management and auditing systems and strengthen our management checking functions.
<Reasons for Non-compliance with the Principles of the Corporate Governance Code (Excerpts)>
CellSeed has stated, “Our company implements all the basic principles stipulated in the Corporate Governance Code as a TES Growth listed company.”
This report is not intended for soliciting or promoting investment activities or offering any advice on investment or the like, but for providing information only. The information included in this report was taken from sources considered reliable by our company. Our company will not guarantee the accuracy, integrity, or appropriateness of information or opinions in this report. Our company will not assume any responsibility for expenses, damages or the like arising out of the use of this report or information obtained from this report. All kinds of rights related to this report belong to Investment Bridge Co., Ltd. The contents, etc. of this report may be revised without notice. Please make an investment decision on your own judgment. Copyright(C) Investment Bridge Co., Ltd. All Rights Reserved. |
For back numbers of Bridge Reports on CellSeed Inc. (7776) and Bridge Salon (IR seminar), please go to our website at the following URL. www.bridge-salon.jp/


