| Linical Co., Ltd. (2183) |
|
||||||||
Company |
Linical Co., Ltd. |
||
Code No. |
2183 |
||
Exchange |
Mothers Market, TSE |
||
Industry |
Service |
||
CEO |
Kazuhiro Hatano |
||
HQ Address |
10 Flr., Shin-Osaka Brick Building, 6-1 Miyahara 1-chome, Yodogawa-ku, Osaka, Japan |
||
Year-end |
March |
||
URL |
|||
* Stock price as of closing on June 4, 2012. Number of shares at the end of the most recent quarter excluding treasury shares.
|
||||||||||||||||||||||||
|
|
* Estimates are those of the Company.
|
|
| Key Points |
 |
| Company Overview |
|
(CRO Business Services Description)
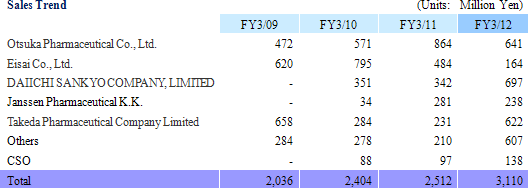
Linical' s business can be divided between the CRO and CSO segments and each accounted for 95.6% and 4.4% of sales respectively during fiscal year March 2012.
Monitoring Service
Monitoring is conducted to ensure that the planning and procedures are performed properly and comply with all laws. Monitoring is conducted by clinical research associates (CRA) by visiting the medical institution that is implementing the clinical trial and by explaining the study drug, protocol, and procedures, and monitoring the trial to collect data following the start of clinical trials.
Quality Control Service
CRAs use specific checklists to review data collected from the medical institutions.
Consulting Activities Service
Linical provides consulting services for client pharmaceutical companies that range from preparing schedules for new drugs under development to filing new-drug applications for approval. In addition, Linical' s technical support is designed to ensure that the development process progresses efficiently.
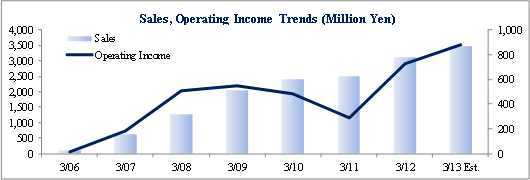 The negative impact of cancellations, postponement of consigned projects in FY3/10, FY3/11 upon sales was compounded by the burden of increases in CRA, QC staff.
However favorable orders in FY3/12 and smooth progress in consigned projects led to a "V-shaped recovery" . (Strengths, Market Environment)
One of Linical' s strengths is its specialization in monitoring services for highly difficult therapeutic areas of central nervous system and oncology, where there are few competitors. For example, in the realm of central nervous system diseases, evaluating the efficacy of drugs prescribed for patients afflicted with Alzheimer' s disease is difficult. At the same time in the realm of oncology, evaluating symptoms to determine whether they are the result of side effects of drugs or the cancer itself is difficult. Therefore high levels of responses and expertise in monitoring are necessary in these difficult realms. Similar to CNS and oncology realms, new drug development for patients of acute diseases and intractable diseases (Difficult diseases), which are also highly difficult disease realms, is very active (There are only a few number of CROs that can respond to these situations). At the same time, evaluation of the efficacy of drugs for adult lifestyle related diseases are generally straightforward because conditions of patients undergoing clinical trials tend to be relatively stable.(1) Strengths in CNS, Oncology Monitoring, Highly Difficult Realms with Few Competitors New drug development trends are shifting from adult lifestyle related diseases towards the realms of CNS and oncology where treatment satisfaction is low. However as stated above, the efficacy evaluation of CNS drugs and the safety evaluation of oncology drugs is difficult. Therefore pharmaceutical companies had performed these functions internally, but in recent years have begun to outsource these more difficult tasks to a greater extent. The CNS realm has been a main field for Linical since its establishment, and focus in the oncology realm was started two years ago along with the joining of staff from AstraZeneca K.K. The CRO industry saw a contraction in its market scale, and it underwent consolidation and restructuring in the aftermath of the Lehman Shock with a reduction in the number of companies operating within the industry. Currently the number of projects is increasing due to improvements in the business environment, but there are only a few surviving CRO companies that can take advantage of the recovery in demand for services in more difficult realms of CNS and oncology. And while competition is intensifying to gain access to human resources needed to capture this growing demand, the business environment overall remains favorable. 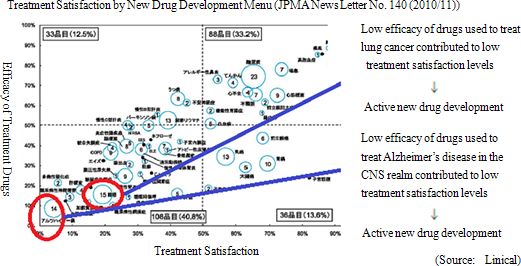 (2) High Profitability
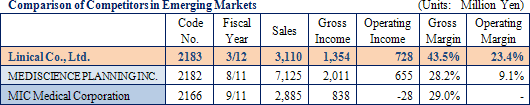
In addition to high levels of various task execution capabilities in the clinical work field for efficacy and safety confirmation, the high levels of knowledge and technology in overall CRO operations are strengths of Linical. The protocol deviation rates on projects undertaken by Linical have been held to extremely low levels, and the implementation period, including the patient enrollment and data collection, for about 20% of all projects has been shortened. Because of its high levels of service quality and quick delivery times in highly difficult therapeutic areas, Linical is able to book orders without having to offer reduced prices and is able to overcome the scale merit advantage of its larger competitors.
 (Issues and Endeavors in Achieving Growth Over the Medium to Long Term)
Among the main issues that confront Linical in its endeavors to achieve growth over the medium to long term are the need to (1) firmly establish its brand in the CRO business, (2) secure and train highly skilled staff in the CRO business, and (3) respond accurately to the needs of its customers (In the realms of oncology, CNS, CSO business, and in overseas markets).
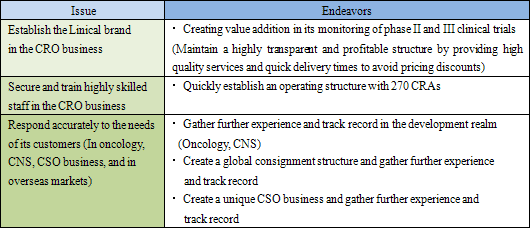 (1) Establish the Linical Brand in the CRO Business
As explained earlier, Linical has been successful in achieving extremely low protocol deviation rates on projects, and has been able to reduce the implementation period on close to 20% of its projects, evidence of its high quality levels and quick delivery capabilities. By endeavoring to create value addition (High quality levels and quick delivery times) in the monitoring of phase II and III clinical trials, Linical is on course to firmly establishing the "Linical brand" and achieving appropriate pricing to maintain its high level of profitability.
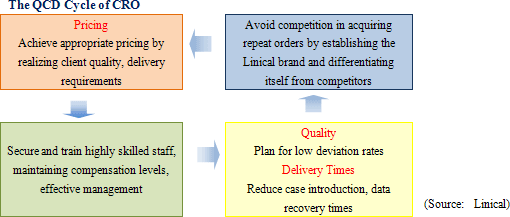 (2) Secure, Training Highly Skilled Staff in the CRO Business
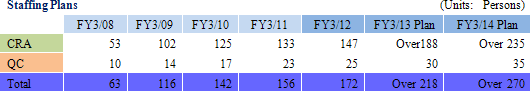
Currently, pricing on orders, which had fallen in the aftermath of the Lehman Shock, have begun to recover, but only Linical and a handful of other CROs are benefitting from this improvement because most of the new drug development activities have been focused upon applications in highly difficult therapeutic areas. At the same time, Linical and most of these other companies share the same problem of limitations in their capacity to provide services. Therefore Linical plans to increase its staff numbers by 20% through the hiring of new graduates (Hiring over 20% presents a risk of reducing quality levels) in addition to hiring similar levels of midcareer staff with experience, for which competition to hire is high. As of May 2012, Linical has 180 CRAs, and seeks to attain a structure with a total of 300 staff including CRAs, managers and quality control (QC) staff as soon as possible. (After other division staff are included, the total number of employees would reach 400).
 (3) Responding to Customers' Needs (CNS, Oncology, CSO Business, Overseas Markets)
① Responses in CNS and Oncology Realms
Linical' s efforts to fortify its responses to new drug development in the realms of CNS disease and oncology have led to successful results. Specifically, while standardization of efficacy evaluation for CNS realm diseases is difficult, Linical has focused upon this realm since its establishment and it boasts of managers and CRAs with strong experience in responding to the needs of customers. Furthermore, in the oncology realm many cases are critical in general nature and they require careful and speedy response in the safety information reporting process. Consequently Linical is promoting responses that meet the growing needs of clients and leverage the skills of the Company' s experienced managers, while at the same time allowing them to gather further knowhow and experiences.
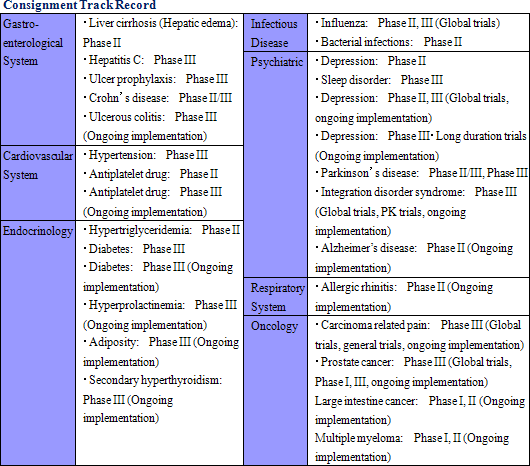 ② Overseas Market Responses (Global Development)
In order to respond to the anticipated growing demand for joint clinical trials on a global basis, Linical will promote a business strategy that focuses upon Asia, the United States and Europe in addition to Japan. On July 15, 2008, LINICAL USA, INC. (California) was established to provide consulting services to medium sized Japanese pharmaceutical manufacturers seeking to enter overseas markets. With a view to providing consigned monitoring for clinical trials in the United States, confidentiality agreements have been exchanged with the United States 9 CRO companies, which are medium sized companies providing services on a worldwide basis. Furthermore, Linical is gathering information on the CRO market in Europe. PAREXEL International, Quintiles and other companies that have functions in the three regions boast of the ability to take on consigned clinical trial related work on a global basis. In order to compete with these companies, Linical seeks to quickly establish strategic alliances with CRO companies in the United States and Europe. At the same time in Asia, Linical is hiring staff from various parts of Asia to prepare for consigned monitoring work in the Asian markets where client demand is strong.
③ CSO Business Responses
Contract sales organization in general is the business where medical representatives (MR) are dispatched to client companies. With regards to Linical, it can leverage the knowhow of its CRO business and its specialized patient realms to increase the orders for consigned product marketing services, and post market launch data planning and gathering (Not only simple human resources dispatching services, but a service contract where the responsibility for various tasks are assumed). During fiscal year March 2012, Linical was successful in capturing new orders in the CNS realm for product marketing.
 |
| Fiscal Year March 2012 Earnings Results |
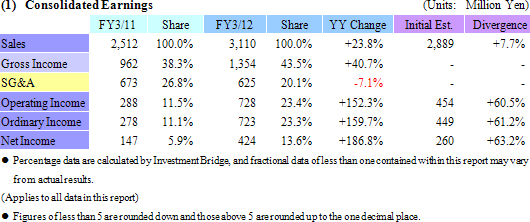 Sales and Ordinary Income Rose by 23.8% and 159.7%
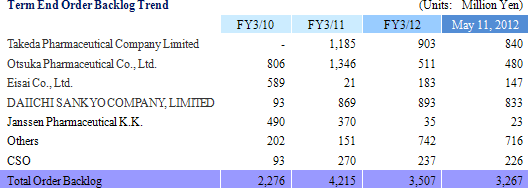
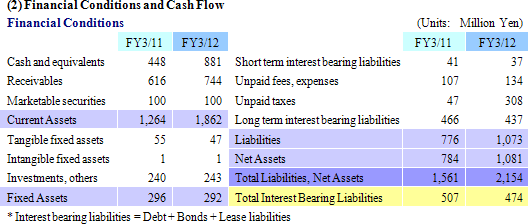
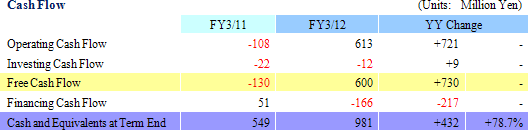
Sales rose by 23.8% year-over-year to ¥3.110 billion. Processing of bountiful order backlog and new project orders allowed the CRO business segment to grow by 23.1% year-over-year. The CSO business segment, being cultivated to become the second pillar of earnings, successfully captured physician led clinical research projects and rose by 41.5% year-over-year. With regards to profits, cost of goods sold rose along with an increase in the number of in clinical research associates (CRA), but a lack of cancellation and postponement of consigned projects and increases in the utilization rates of CRAs allowed gross margin to improve by 5.2% points. Consequently, ordinary income rose by 159.7% year-over-year to ¥723 million. Moreover, despite the upward revision to full year earnings estimates announced on February 13, both sales and profits were able to exceed these revised estimates. The revisions had called for sales, and operating, ordinary, and net incomes of ¥3,072, ¥706, ¥701, and ¥404 million respectively.
    |
| Fiscal Year March 2013 Earnings Estimates |
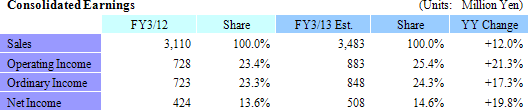 Sales and Ordinary Income Expected to Rise by 12.0% and 17.3%
Linical calls for sales to rise by 12.0% year-over-year to ¥3.483 billion in fiscal year March 2013. The processing of significant order backlogs, increases in the number of staff on existing projects, and booking of new project orders are cited as the main factors for the higher sales (Some cancellations and postponement of projects have been factored into these estimates). With regards to profits, cost of goods sold are expected to rise due to increases in CRA staff, while selling, general and administrative expenses are expected to remain in line with the previous fiscal year. Therefore the higher sales are expected to allow operating income to rise by 21.3% year-over-year to ¥883 million. Linical also expects to pay a yearend dividend of ¥11 per share.
|
| Conclusions |
|
Disclaimer
This report is intended solely for information purposes, and is not intended as a solicitation to invest in the shares of this company. The information and opinions contained within this report are based on data made publicly available by the Company, and comes from sources that we judge to be reliable. However we cannot guarantee the accuracy or completeness of the data. This report is not a guarantee of the accuracy, completeness or validity of said information and or opinions, nor do we bear any responsibility for the same. All rights pertaining to this report belong to Investment Bridge Co., Ltd., which may change the contents thereof at any time without prior notice. All investment decisions are the responsibility of the individual and should be made only after proper consideration.Copyright(C) 2012 Investment Bridge Co., Ltd. All Rights Reserved. To view back numbers of Bridge Reports on Linical Co., Ltd. (2183) and other companies and to see IR related seminars of Bridge Salon, please go to our website at the following url: www.bridge-salon.jp/ |




