Bridge Report:(2183)Linical the Fiscal Year ended March 2025
 Kazuhiro Hatano CEO | Linical Co., Ltd. (2183) |
 |
Company Information
Market | TSE Standard Market |
Industry | Service |
CEO | Kazuhiro Hatano |
HQ Address | Shin-Osaka Brick Building, 6-1 Miyahara 1-chome, Yodogawa-ku, Osaka, Japan |
Year-end | End of March |
HP |
Stock Information
Share Price | Number of shares issued (excluding treasury shares) | Total market cap | ROE Act. | Trading Unit | |
¥326 | 22,586,431shares | ¥7,363million | -7.0% | 100shares | |
DPS Est. | Dividend yield Est. | EPS Est. | PER Est. | BPS Act. | PBR Act. |
¥16.00 | 4.9% | ¥6.64 | 49.0x | ¥321.14 | 1.02x |
* Stock price is as of closing on June 25, 2025. Number of shares issued is the term-end figure stated in the summary of financial results for FY3/25 excluding treasury shares.
* ROE and BPS are based on FY3/25 results. EPS and DPS are based on the estimates of FY3/26.
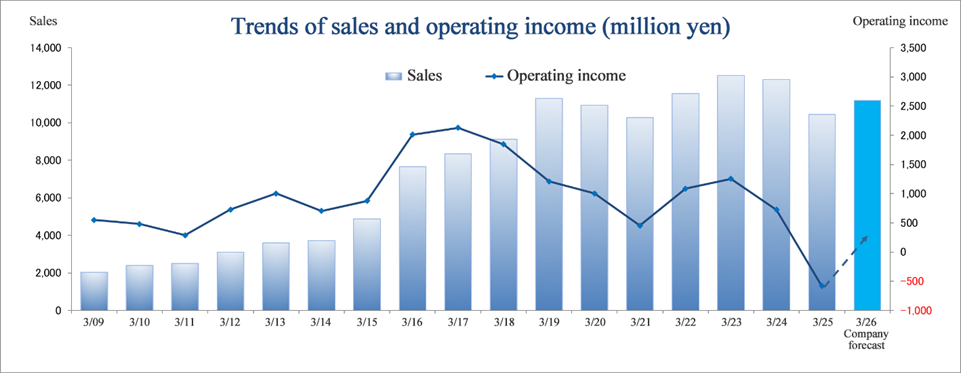
Consolidated Earnings Trend
Fiscal Year | Sales | Operating Income | Ordinary Income | Parent Net Income | EPS | DPS |
March 2022 Act. | 11,555 | 1,085 | 1,183 | 790 | 35.00 | 14.00 |
March 2023 Act. | 12,516 | 1,256 | 1,283 | 1,004 | 44.47 | 14.00 |
March 2024 Act. | 12,307 | 725 | 790 | 338 | 14.98 | 15.00 |
March 2025 Act. | 10,437 | -583 | -498 | -539 | -23.87 | 16.00 |
March 2026 Est. | 11,200 | 300 | 320 | 150 | 6.64 | 16.00 |
*Unit: Million yen.
*Estimates are those of the company.
This Bridge Report reviews on the overview of Linical Co., Ltd.’s earnings results for the fiscal year ended March 2025 and its forecast for the fiscal year ending March 2026.
Table of Contents
Key Points
1. Company Overview
2. Management Strategy
3. Fiscal Year ended March 2025 Earnings Results
4. Fiscal Year ending March 2026 Earnings Forecasts
5. Conclusions
<Reference:Regarding Corporate Governance>
Key Points
- In the fiscal year ended March 2025, sales decreased 15.2%, and they posted an operating loss of 583 million yen (an operating income of 725 million yen in the previous fiscal year). Although sales increased significantly in the U.S., they saw a decline in overall sales, due to significant decreases in Japan and other Asian countries. In terms of profit, an operating loss was posted, as the impact of the decline in sales in Japan and other Asian countries was significant.
- For the fiscal year ending March 2026, sales are expected to grow 7.3% and an operating income of 300 million yen is projected (an operating loss of 583 million yen in fiscal year ended March 2025). The company expects increased revenue and profit due to the growth in the United States and the recovery in Europe, Japan, and other Asian countries. In Japan, sales activities are expected to lead to an increase of inquiries about new projects from both domestic and overseas pharmaceutical companies. In other Asian countries, the section in South Korea continues to experience delays in existing projects due to medical strikes, but it is seeing an increase of orders for new projects, including data management and statistical analysis. In China, they are expected to receive new orders from Japanese enterprises, and in Taiwan, they are projected to receive new orders from domestic and overseas clients. In the U.S., existing large-scale projects will be completed as scheduled, but there are many inquiries about new projects, including large-scale ones, so they will keep enhancing sales activities. The section in Europe is expected to make progress in securing new orders by promoting collaboration with its U.S. division.In addition, the dividend is expected to be 16 yen/share, unchanged from the previous fiscal year.
- As growth strategies, they uphold the enhancement of corporate governance, marketing, and investment in IT. For corporate governance, they will reform and strengthen the organizational structure in order to improve profitability while expanding services. For marketing, they will strengthen the capability of giving well-designed proposals to clients to differentiate themselves from leading global CROs. For investment in IT, they will meet the needs for streamlining of clinical trials and improve the efficiency of business administration by utilizing digital technologies. It is noteworthy what kinds of outcomes will be delivered through the implementation of growth strategies.
1. Company Overview
The company is a global contract research organization (CRO) based in Japan, which provides a comprehensive range of services ranging from the initial phase of clinical trials to post-marketing studies as a professional in development of pharmaceutical products. Linical Co., Ltd. provides contract research organization (CRO) services that support the drug development processes of pharmaceutical companies on an outsourced consignment basis, and sales and marketing functions for pharmaceutical products and post market launch clinical research and surveys on a consigned basis in the Contract Medical Affairs Business (CMA). In addition, the company offers meticulous services to meet the requests from clients and optimal solutions as a global CRO. They give full support with proposals for planning of early small-scale trials, late-stage trials, and applications for approval, which can be achieved because the scale is medium, and contribute to healthcare as a partner required for developing new treatment methods, including pharmaceutical products. In the past 5 years, they have dealt with over 600 clinical trials, the retention rate of clients exceeds 85%, and they registered cases for over 80% of trials. Then, they offer services in over 30 countries.
Linical has conducted various efforts to eradicate oncology, central nervous system and other diseases globally since its founding, and it has deployed its CRO Business in therapeutic areas where there is strong demand for new drug development. Linical is a leading company in the field of clinical trials for cancer treatment, and has a vast amount of experience in the field of central nervous system (CNS) diseases. In addition, since the inauguration of business, they have focused on immune system diseases, and have plenty of experience of Phases I to IV trials and profound knowledge of rare diseases and common diseases, and made clinical trials of clients successful. As an awarded CRO, the company possesses expertise for supporting a variety of therapeutic areas.
[Management philosophy]
The management philosophy is “Linical promotes the greater wellbeing of all our stakeholders—patients, clients and employees— we strive constantly to offer professional, high-quality services to support all aspects of new drug development.”

The blue color represents “Integrity&Honesty,”
the red color “Unending Enthusiasm,” and
the yellow color “Continuing Spirit of Inquiry.”
This corporate logo depicts the company’s hope of pursuing the wellbeing of patients around the world through business, and the company has the mission to “wings to new drugs.”
[Corporate History]
Linical Co., Ltd. was established in June 2005 by nine members who worked at Fujisawa Pharmaceutical Co., Ltd. (Currently known as Astellas Pharma Inc.) on the development of immunosuppressant drugs. Established with the objective of becoming the ideal drug development outsourcing (CRO) company from Osaka, Linical focused its efforts upon the realms of central nervous system diseases (CNS) and oncology since its founding, and received one of its first orders from Otsuka Pharmaceutical Company shortly after its establishment. Thereafter, the Company fortified its staffing as part of its efforts to strengthen its order taking capabilities. In addition, Linical is benefitting from the bountiful experiences of its employees in the realm of oncology pharmaceutical product development and experiences having worked at foreign pharmaceutical companies. Consequently, Linical is successfully expanding orders in the near term.
With its advance into the site management organization (SMO, clinical trial facility support organization) business, Aurora Ltd. was turned into a subsidiary in January 2006. However, all shares held in Aurora were later sold in May 2007 in order to focus management resources upon the CRO Business. In July 2008, Linical USA, Inc. was established in California, United States to provide support to Japanese pharmaceutical companies seeking to enter the United States market. Also, in October of the same year, Linical listed its shares on the Mothers Market of the Tokyo Stock Exchange, and subsequently moved its listing to the First Section of the Tokyo Stock Exchange in March 2013. In May 2013, Linical Taiwan Co., Ltd. and Linical Korea Co., Ltd. were established in Taiwan and Korea respectively. In April 2014, Linical teamed up with its Linical Korea to acquire the Korean CRO company P-pro. Korea Co., Ltd. On October 29, 2014, all of the shares of Nuvisan CDD Holding GmbH, which conducts CRO Business in Europe, were acquired and it was converted to a 100% owned subsidiary effective on December 1, 2014. In order to strengthen the collaboration within the Group, the company name of Nuvisan CDD was changed to Linical Europe GmbH. In addition, Linical U.K. Ltd. was established in March 2016, and a local subsidiary called Linical Poland SP. Z.O.O. was also established in October of the same year. Moreover, LINICAL Czech Republic s.r.o was established in September 2017. In addition, Accelovance, Inc. was acquired in April 2018 and its company name was changed to Linical Accelovance America, Inc. In addition, Linical Hungary Kft. was established in March 2019, and Linical China Co., Ltd. was established in May 2019. Furthermore, the company further strengthened their system for undertaking global joint clinical trials, through the enhancement of their business in the European region by integrating the European subsidiary of Linical Accelovance America, Inc. (LAA) into LINICAL Europe GmbH in December 2019, and the establishment of a Shanghai branch in February 2020. In April 2020, Linical Benelux BV and Linical Accelovance Europe BV were merged to form Linical Netherlands BV, and Linical China Co., Ltd. and Linical Accelovance China Ltd. are scheduled to be integrated in the fiscal year ending March 2023. The company grew steadily through overseas mergers and acquisitions, and achieved record sales consecutively in fiscal year ended March 2022 and fiscal year ended March 2023. In the fiscal year ended March 2024, profit declined due to the decrease in sales in Japan and Europe, and in the fiscal year ending March 2025, too, sales and profit declined due to the drop in sales in Japan and other Asian countries.
Produced by Investment Bridge Co., Ltd. with reference to disclosed material.
[Strengths]
Global one-stop full services |
They have established an international development system, to offer services in Europe, the U.S., and Asia, mainly Japan. They offer comprehensive services, including the planning of development of pharmaceutical products, monitoring, pharmaceutical affairs, and data management, in a one-stop manner. |
To create, develop, and improve medicines thoroughly |
They deal with all processes, including the development of new medicines and lifecycle management after approval, as professionals in development of pharmaceutical products. |
A track record of conducting highly difficult tests |
They concentrate on the fields of cancer, the central nervous system, immunological diseases, etc. in which unmet medical needs are significant and clinical trials are very difficult, and have plentiful experiences. Currently, they are expanding their business in the fields of regenerative medicine, ophthalmology, dermatology, etc. |
[Business Description]
As a global CRO founded in Japan, the company operates primarily in Japan, and also in Asia, Europe and the U.S, providing a comprehensive range of services ranging from the drug discovery stage to clinical development to post-marketing drug development. The company has extensive experience and achievements in the trending areas of drug development, notably in oncology, neurology , and immunology.
A CRO is an organization that receives requests from pharmaceutical companies and others to act on their behalf and provide support for clinical trials conducted during the development phase of a pharmaceutical product. It is an organization with high expertise in clinical trials and is a professional in the field of drug development. The scope of work includes monitoring activities to ensure that clinical trials are conducted in compliance with regulatory requirements and the clinical trial protocol, including data management and medical writing activities.
Linical mainly conducts contract research organization (CRO) business, post market launches clinical trial and clinical research and marketing support activities in the Contract Medical Affairs Business, and new drug development support business. As a true partner, the company contributes to the maximization of the value of the medical drugs by helping the procedure from the non-clinical tests to clinical development and after-release surveys and clinical trial, and making it possible to shorten the time needed to start selling the drugs and prolong the life-cycle of the products. On top of that, the company supports not only pharmaceutical companies but also the bio-ventures in various ways including exit strategies.
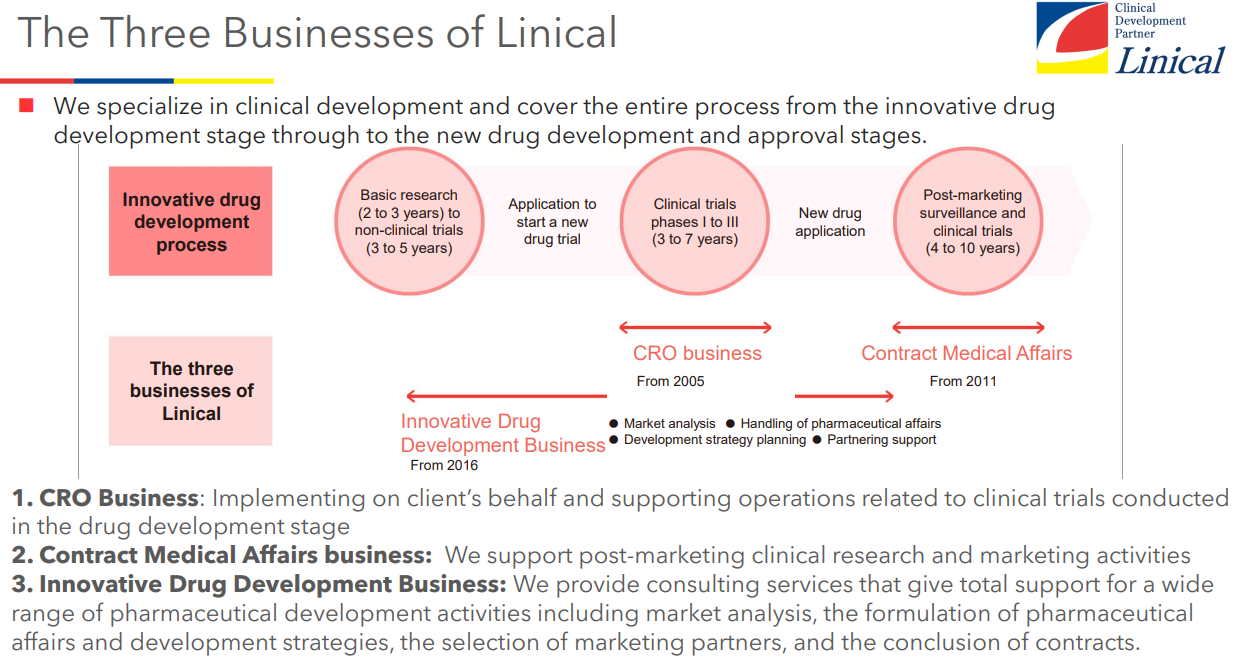
(Source: Linical)
CRO Business (Contract Research Organization)
The CRO Business undertakes part of the clinical trial operations conducted by pharmaceutical companies, including monitoring, data management, medical writing, pharmacovigilance, statistical analysis, and quality control. The company employs highly skilled and experienced staff with the aim of supporting high-quality, highly efficient clinical trials that will lead to the rapid launch of new drugs on the market. The company has opened facilities in Asia (Korea, Taiwan, Singapore, China), Europe and the United States to be able to respond to growing demand for global studies. They offer one-stop services, including the design of clinical development plans, monitoring, data management, statistical analysis, pharmacovigilance, and support for application for approvals for pharmaceutical products and medical apparatus. Among the new drug development projects spanning from 10 to 20 years, Linical is specialized in the processes of “Phase II” and “Phase III” that require 3 to 7 years targeting patients who are particularly important in clinical trials, and it provides “monitoring” services that are the core of the clinical trials in the contract-based business style in conjunction with “quality control” and “consulting.” It collects highly reliable data and supports the rapid and reliable development of new drugs.
In addition, the company offers high-quality services in the fields of schedule management, standard procedure documents for clinical trials, compliance with GCP, the reliability of data and case reports, etc. Furthermore, the company has engaged in clinical trials in the fields where unmet medical needs are significant as many patients are waiting for the advent of new medicines, which are difficult to develop, since its establishment, and concentrates on the projects regarding cancer, immunity, and the central nervous system.
* Global jointly conducted clinical trials
“Global jointly conducted clinical trials” refers to conducting clinical trials simultaneously in multiple countries or regions in order to develop new drugs on a global scale and aim for early launch.
*GCP (Good Clinical Practice)
“GCP” is the international rule the companies are supposed to obey when they conduct the clinical trial. It is enacted by Ministry of Health, Labor and Welfare as a ministerial ordinance so that they can conduct it properly in Japan.
Contract Medical Affairs Business
In the contract medical affairs business, the company provides support for the organizational structure and construction of corporate and doctors-led clinical research, as well as planning for surveys, monitoring, and auditing services for post-release clinical trials and investigations. The Clinical Trials Act is enacted, and the environment surrounding clinical research is changing drastically. Under this circumstance, to obtain information in a timely manner and be the best partner for the medical affairs department of pharmaceutical companies, Linical provides full-service support including data management and statistical analysis with a focus on monitoring and research administration works of clinical trials. It has a policy to respond to the latest regulations and contribute to the creation of evidence in the challenging areas based on the know-how cultivated in the past development works.
Innovative Drug Development Business
Following the existing CRO Business and Contract Medical Affairs Business, Linical is cultivating the third business called Innovative Drug Development Business. In the innovative drug development business, the company provides consulting services to support entry into the Japanese market. This business is mainly operated by employees who are involved with licensing, business development, clinical trials development, development pharmacy, and marketing at major domestic pharmaceutical companies and have abundant results and experiences in determining developed products, introduction and derivation negotiation, and clinical development. In the Innovative Drug Development Business, 3 types of consulting services: a) market analysis of developed products, b) support for PMDA consultations, and c) licensing support, are provided. With these experiences as a weapon, they support pharmaceutical companies and bio technology companies inside and outside Japan in the broad therapeutic field.
[Services]
Strategy for drug development
Protocol Development and Study Design | The company has a track record in protocol development and study design, resulting in numerous successful clinical developments. It formulates a plan according to the project's needs and develop a roadmap for high-quality, efficient testing while mitigating risks. |
Regulatory Consulting | The company is an expert in global drug development and provides world-class consulting services in pharmaceutical affairs. It proposes the most appropriate strategies and assists in the most cost-effective and fastest response in the pharmaceutical process. |
Regulatory Affairs | The pharmaceutical affairs team at the company has extensive expertise and experience in supporting early to late-phase clinical development. Furthermore, they understand both regulatory and clinical affairs and provide comprehensive support for drug and medical device development, including regulatory application strategies, support for meetings with regulatory authorities, as well as coordination during the initiation of clinical trials. The company has extensive experience working with clients in the U.S, Europe, and Asia. |
Quality Assurance | The company places the highest emphasis on quality. It offers services worldwide, from developing SOP to QA consulting to auditing. |
Medical Writing | Medical writing is essential for clear communication and consistency in the preparation of documents related to clinical trials, as well as to ensure the safety of subjects and deal with regulatory reviews. The company aims to provide additional value through high-quality medical writing by utilizing its high level of expertise to meet the client's requirements. |
Clinical trials
Feasibility and Study Set-up | To conduct feasibility studies and select medical facilities to initiate clinical trials more swiftly. With a practical and strategic approach based on extensive field experience, the company works closely with clients to understand their objectives, propose innovative solutions, and initiate clinical trials to ensure prompt completion of incorporation. |
Project Management | The experienced project team of the company works as a partner with clients, assisting them until the completion to ensure that the trials are on schedule, within a budget, and obtain data of the expected quality. Furthermore, the company accompanies clients on projects to ensure that their needs are satisfied, responding quickly to their requests while leveraging its previous experience. |
Pharmacovigilance | The pharmacovigilance of the company is a global team of experts. They provide rapid and accurate support to clients in responding to safety information. |
Clinical Monitoring | Monitoring is essential for protecting the human rights and safety of subjects as well as ensuring regulatory compliance, data quality, and the integrity of clinical trial results. Since its establishment, the company has specifically dedicated itself to monitoring and is renowned for its quality among its clients. |
Data Management and Biostatistics | The data management and biostatistics of the company are aimed at providing both deep insights and efficiency at every stage of the process. The professionals of the company perform everything ranging from consulting to full-service data management to statistical design. |
Patient Recruitment | Finding suitable subjects for clinical trials is not easy, and the inclusion of cases is the major factor in the success or failure of a clinical trial, which can lead to significant delays in the trials and losses. It is essential for the success of a clinical trial that the recruitment plan for subjects is carefully considered. |
Training | The CRA training offered by the company provides more practical training, with lectures given by skilled professionals and clinical trial managers (CTMs) from clinical sites, enabling trainees to become excellent clinical development monitors with exceptional monitoring skills. |
[Global expansion]
The company is a global CRO based in Japan, with a focus on Japan, it is operating its offices around the world, including Asia, Europe and the U.S. The company operates in about 20 countries/regions, and about 30 countries/regions if countries where it can provide services through partners are included. Experts in each function, who are familiar with local regulations and customs, work together globally to provide detailed services customized to each and every project.
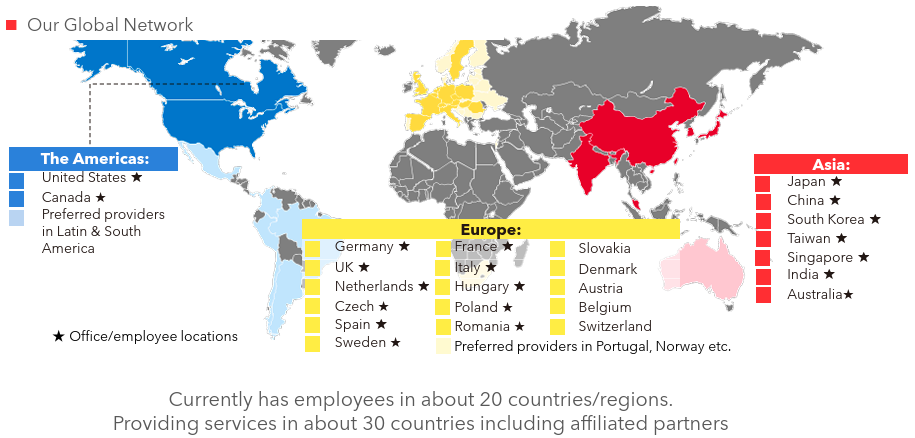
(Source: Linical)
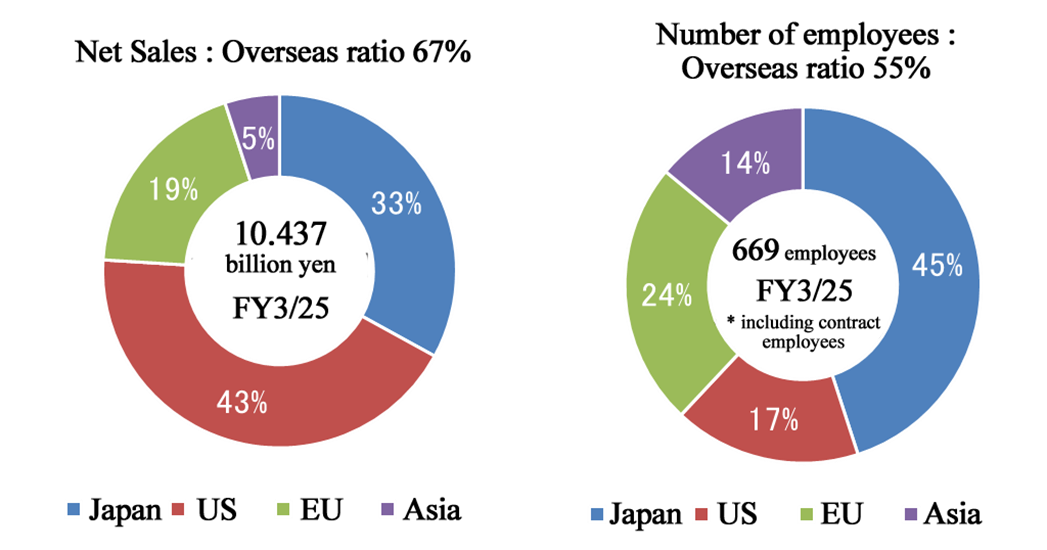
Source: Linical)
In the fiscal year ended March 2025, the overseas ratio was 67% for sales and 55% for the number of employees.
[Order backlog by region]
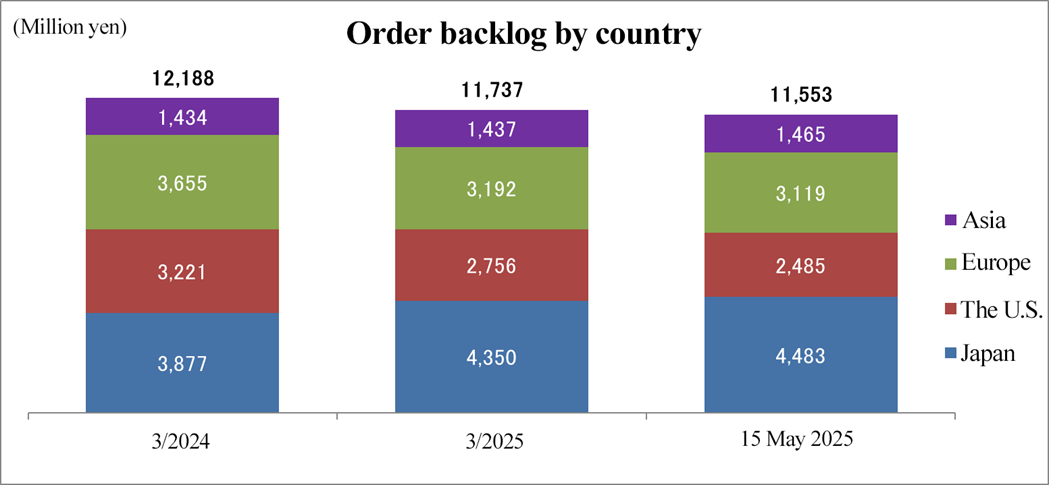
*Produced by Investment Bridge Co., Ltd. with reference to disclosed material.
Order backlog refers to the balance of the amount of orders received for undertaking of tasks for which contracts have been already concluded. It shows sales that will be earned in the next one to five years and is an indicator that serves as the basis for the corporate group's future earnings forecasts. As of May 15, 2025, order backlog stood at 11,553 million yen, down 5.2% from the end of the fiscal year ended March 2024. Order backlog dropped significantly, as the development in large-scale projects was cancelled and their periods were shortened in fiscal year ended March 2024. In Japan and other Asian countries, order backlog increased from fiscal year ended March 2024, as they secured multiple new projects and revised some contracts. On the other hand, in the U.S. and Europe, the sales from existing projects were posted, but order backlog dropped from the end of March 2024, due to the postponement of trials for which they have received informal orders.
[Selected as the Best CRO in the U.S.]

(Source: Linical)
2. Management Strategy
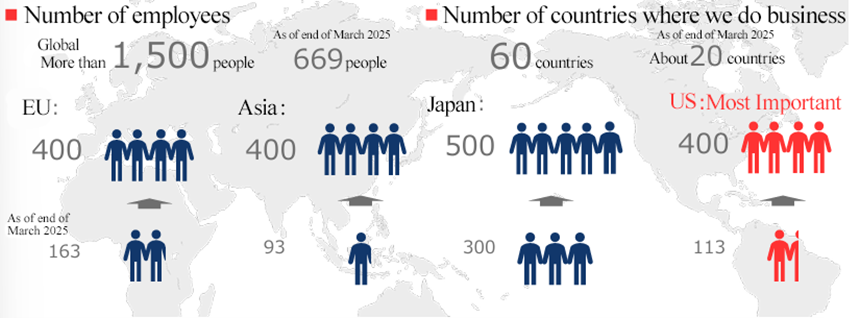
[Medium and Long term Goal]
(1) To build a system with more than 1,500 employees where there will be 500 in Japan, 400 in Asia, 400 in Europe, and 400 in the U.S.
(2) To maintain and improve profitability while making growth investments (including M&A) in all main business regions
(3) The company will expand its business to about 60 countries around the world.
(Source: Linical)
[Growth strategies]
In order to expand the overseas business further, they will enhance the provision of high-quality services and the financial base.
① Corporate governance
In order to improve profitability while expanding services, they will reform and strengthen their organizational structure.
To enhance communication among business bases |
◆To swiftly share the information on progress and issues at each base, and fortify their cooperative system |
To secure and train personnel |
◆To enhance measures for retaining personnel, while recruiting and training excellent personnel so as to offer highly technical services |
Linkage among services |
◆To strengthen the linkage among different services, including clinical monitoring, innovative drug development business, and data management, and offer services swiftly and flexibly |
② Marketing
They will improve the capability of giving meticulous proposals to clients, to differentiate themselves from leading global CROs.
Reform of the marketing organization |
◆They will accumulate know-how while standardizing procedures, so as to conduct organized marketing activities globally. |
Enrichment of marketing strategies for each client |
◆While targeting mainly emerging biopharmaceutical companies in Europe and the U.S., they will enrich marketing strategies to meet the needs that vary among clients, including major pharmaceutical companies and Japanese pharmaceutical companies. |
Training of global sales staff |
◆ To secure personnel who can engage in marketing activities based on global cooperation |
③ Investment in IT
By utilizing digital technologies, they will respond to the needs for streamlining of clinical trials and improve the efficiency of business administration.
Utilization of AI for clinical trial operations |
◆To discuss the adoption of systems and develop personnel who are versed in both technology and clinical development, in order to utilize AI for clinical trial operations |
Promotion of DX for each function |
◆To develop digital tools and a system environment, which will be shared by group companies, and improve productivity through DX |
Cementing of cooperative relationships |
◆To strengthen the network of partners for system development, who are required for distributed clinical trials and AI utilization |
3. Fiscal Year ended March 2025 Earnings Results
(1) Consolidated results
| FY3/24 | Ratio to sales | FY3/25 | Ratio to sales | YoY | ||||
Sales | 12,307 | 100.0% | 10,437 | 100.0% | -15.2% | ||||
Gross profit | 3,778 | 30.7% | 2,375 | 22.8% | -37.1% | ||||
SG&A | 3,052 | 24.8% | 2,959 | 28.4% | -3.1% | ||||
Operating income | 725 | 5.9% | -583 | -5.6% | - | ||||
Ordinary income | 790 | 6.4% | -498 | -4.8% | - | ||||
Profit attributable to owners of parent | 338 | 2.7% | -539 | -5.2% | - | ||||
*Unit: million yen
*The figures include figures calculated by Investment Bridge Co., Ltd., and may differ from actual figures. (Abbreviated hereafter)
Sales declined 15.2% year on year, and an operating loss of 583 million yen was recorded.
In the fiscal year ended March 2025, sales decreased 15.2% year on year to 10,437 million yen, and they posted an operating loss of 583 million yen (an operating income of 725 million yen in the previous fiscal year). Although sales increased in the U.S., they saw a decline in overall sales, due to significant decreases in Japan and other Asian countries.
In terms of profit, an operating loss was posted, as the impact of the decline in sales in Japan and other Asian countries was significant.
In Japan, both sales declined, resulting in an operating loss. The company was unable to recover from the impact of the cancellation and shortened periods of several large-scale existing projects in the previous fiscal year. In the U.S., sales increased, but profit declined. While sales rose due to the faster-than-expected progress of ongoing tests and contract revisions involving increased workloads, profit declined due to rising labor costs and higher outsourcing expenses. In Europe, sales dropped, but operating loss narrowed. Although the strengthened sales structure began to produce results, its contribution to the results in fiscal year ended March 2025 was limited, resulting in a drop in sales. However, cost reduction helped shrink the deficit.
Gross profit margin decreased 7.9 points year on year to 22.8%. SG&A expenses decreased 3.1% year on year, as cost optimization initiatives were implemented across all regions. In addition, ordinary loss was 498 million yen, smaller than operating loss, due to factors such as increased interest income and a decline in losses on valuation of investment securities. No significant extraordinary gains or losses were recorded.
Sales and profit by segment
In the CRO Business, sales decreased 14.1% year on year to 9,921 million yen and operating income decreased 44.8% year on year to 1,499 million yen.
In the Contract Medical Affairs Business, sales decreased 32.2% year on year to 515 million yen, and operating loss was 66 million yen (operating income of 148 million yen in the same period of the previous year).
(2) Performance trend in each region
| FY3/24 Act. | FY3/25 Act. | ||||
Sales | Operating income | Sales | Increase/ decrease rate | Operating income | Increase/ decrease rate | |
Japan | 5,101 | 363 | 3,676 | -27.9% | -585 | - |
U.S. | 4,498 | 648 | 4,789 | +6.5% | 625 | -3.6% |
Europe | 3,220 | -44 | 3,089 | -4.1% | -37 | - |
Korea | 970 | 128 | 751 | -22.6% | -79 | - |
Taiwan | 107 | -19 | 84 | -21.4% | -39 | - |
China | 280 | -61 | 188 | -32.8% | -22 | - |
Adjustment | -1,872 | -289 | -2,142 | - | -445 | - |
Total | 12,307 | 725 | 10,437 | -15.2% | -583 | - |
*Unit: million yen
*Amortization of goodwill is recorded as an adjustment. “Sales” means the value before exclusion of internal transactions.
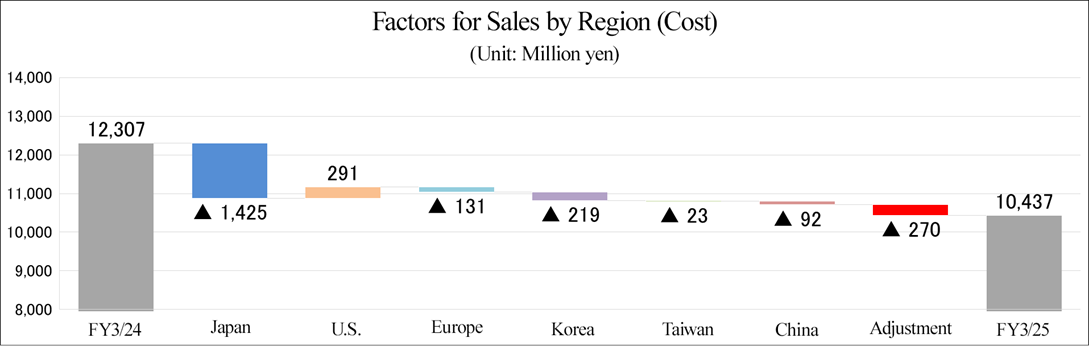
*Produced by Investment Bridge Co., Ltd. with reference to disclosed material.
[Japan]
In Japan, sales declined significantly year on year, resulting in an operating loss. Although the company received multiple large-scale contracts from domestic and overseas clients in the second half of the fiscal year, it was unable to offset the impact on sales from the cancellation and contract revisions involving shortened durations of several large-scale existing projects in the previous fiscal year. As a result, both sales and profit decreased considerably year on year, leading to an operating deficit. On the other hand, order backlog increased from the end of the previous fiscal year, owing to sales activities targeting overseas clients conducted in collaboration with businesses in Europe, the U.S., and Asia.
[Korea]
In South Korea, sales declined significantly year on year, and an operating loss was recorded, due to the impact of a nationwide medical strike, a decrease in revenue caused by contract revisions for existing projects, and delays in the progress of multiple projects. However, by collaborating with businesses in Japan and other Asian countries, the company successfully received numerous orders from overseas clients through group companies. With the progress of new projects including related services such as data management, the South Korean business moved into the black in the fourth quarter. While the business environment remains challenging, the company will continue its marketing efforts to secure orders from both domestic and overseas clients.
[China]
In China, sales declined year on year due to the completion of existing projects. However, operating loss narrowed thanks to cost reductions. Interest of medium-sized Japanese pharmaceutical companies in entering the Chinese market has been increasing, and as a result of ongoing sales efforts, the company has secured new projects that are expected to contribute to sales from the next fiscal year onward.
[Taiwan]
In Taiwan, sales declined year on year as the company struggled to secure new projects and was unable to offset the impact of cancellations and completions of existing projects that occurred in the previous fiscal year. Consequently, operating loss widened. However, there were signs of improvement in sales activities, including receiving informal orders for projects in Taiwan.
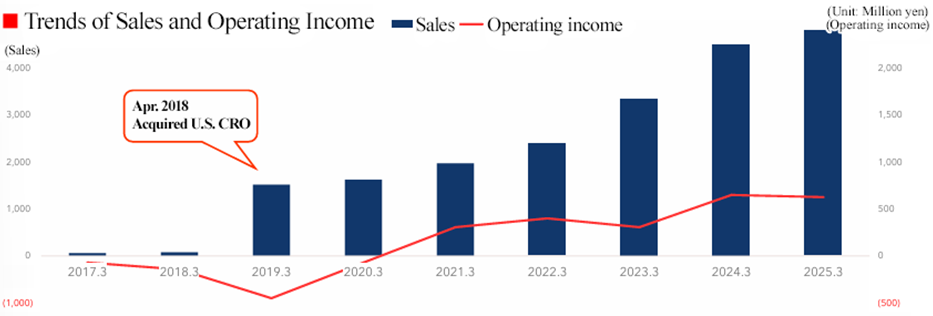
[United States]
In the U.S., sales increased, but profit declined year on year. Sales rose as contracted projects progressed as planned and additional work led to contract modifications. However, profit decreased due to the augmentation of labor and outsourcing costs. Since acquiring a local CRO in April 2018, the U.S. business has grown steadily, achieving sales growth again in the fiscal year ended March 2025. For the fiscal year ending March 2026, the company aims to capture strong development demand from biotech firms and achieve sustainable growth.
(Source: Linical)
[Europe]
In Europe, although the company began to see results from strengthened sales activities driven by collaboration with the U.S. business division, it was not sufficient to contribute to sales growth in fiscal year ended March 2025. As a result, sales declined year on year, and while operating loss narrowed, it remained in the red. The company will continue to focus on group-wide coordinated marketing efforts to receive more orders. At the same time, it will strive to enhance profitability by improving the efficiency of back-office operations.
[Goodwill balance and remaining amortization period (at the end of FY 3/25)]
| Goodwill | Related intangible assets other than goodwill *2 | ||||
Balance at end of term | Remaining Amortization Period (year) | Annual Amortization *3 | Balance at end of term | Remaining Amortization Period (year) | Annual Amortization *3 | |
Korea | Termination of depreciation in FY 3/19 | Termination of depreciation in FY 3/19 | ||||
Europe*1 | 1,195 | 8-9 | 147 | 6 57 | 2 5.7 | 3 10 |
United states*1 | 1,949 | 9 | 220 | 22 | 3 | 11 |
Total | 3,144 | - | 368 | 85 | - | 24 |
*Unit: million yen
*1 Goodwill generated by the acquisition of Linical Accelovance America, Inc., has been apportioned pro rata to its European subsidiary.
*2 Intangible assets other than goodwill recognized by purchase price allocation.
*3 Figures have been converted at the exchange rate as of the end of the fiscal year ended March 2025.
(3) Change in order backlog
| End of FY 3/24 (A) | FY 3/25 Term-end | As of May 15, 2025 (B) | Difference from the end of the previous term (B-A)/(A) |
Japan | 3,877 | 4,350 | 4,483 | +15.6% |
United States | 3,221 | 2,756 | 2,485 | -22.8% |
Europe | 3,655 | 3,192 | 3,119 | -14.6% |
Asia | 1,434 | 1,437 | 1,465 | +2.1% |
Total | 12,188 | 11,737 | 11,553 | -5.2% |
*Unit: million yen
In the CRO Business, the total amount of clinical trials commissioned to the company, which has an implementation period of one to three years, is determined by the difficulty of the clinical trials due to the number of cases and the target disease. A consignment contract is concluded with the client for this implementation period, and sales are generated according to the contract. In the Contract Medical Affairs Business, the company enters into a consignment contract with a client for a similar period of time, and sales are generated in accordance with the contract.
The order backlog is the balance of orders received for contracted services for which contracts have already been concluded. This is an indicator of sales that will occur over the next one to five years, and is the basis for the company's future earnings forecasts.
The order backlog as of May 15, 2025 stood at 11,553 million yen, down 5.2% from the end of the fiscal year ended March 2024.
Although the business environment remains challenging because some new medicines are still to be approved and cannot be used in Japan, order backlog increased from the end of the fiscal year ended March 2024, as a result of multiple new projects and contract revisions. In addition, there are several projects under negotiation with domestic pharmaceutical companies that are not yet included in the above figure, but for them, they have received informal orders. Furthermore, inquiries for new projects, including large-scale international collaborative trials, are increasing, indicating that the effects of ongoing sales activities are beginning to materialize.
Despite delays in revenue recognition from existing projects and challenges in receiving new orders in South Korea due to the impact of the medical strike, the company concluded contracts for several new projects from Taiwan and China, including data management and statistical analysis services. As a result, order backlog increased from the end of the fiscal year ended March 2024. The company will continue persistent sales efforts by leveraging collaboration between the Japan/Asia and Europe/U.S. business divisions, including proposals to overseas biotech firms for entering the markets in Japan and other Asian countries.
In the United States, while sales from existing projects were posted steadily, new projects did not progress due to the postponement of trials for previously accepted projects, etc. and order backlog declined from the end of the fiscal year ended March 2024. Although large-scale existing projects are progressing smoothly and are expected to be completed as planned, there are several new projects currently under contract negotiation that are not reflected in the reported order backlog. In addition, the company continues to receive a high volume of inquiries, especially from biotech firms, regarding multiple global project opportunities. To build up order backlog, the company is actively enhancing its sales efforts.
In Europe, the company secured additional orders through contract revisions involving extensions and increased workloads for existing projects. As a result, order backlog declined from the end of the fiscal year ended March 2024, as those existing projects progressed smoothly and were recorded as sales. Meanwhile, they are receiving orders for new projects through the cooperation with the business in the U.S. and there are multiple projects to be ordered, which are not included in the above order backlog. They will further enhance global synergy in the aspect of marketing, to receive more orders for new projects in Europe, etc. from U.S. enterprises.
(4) Variation in performance in the fourth quarter (January to March)
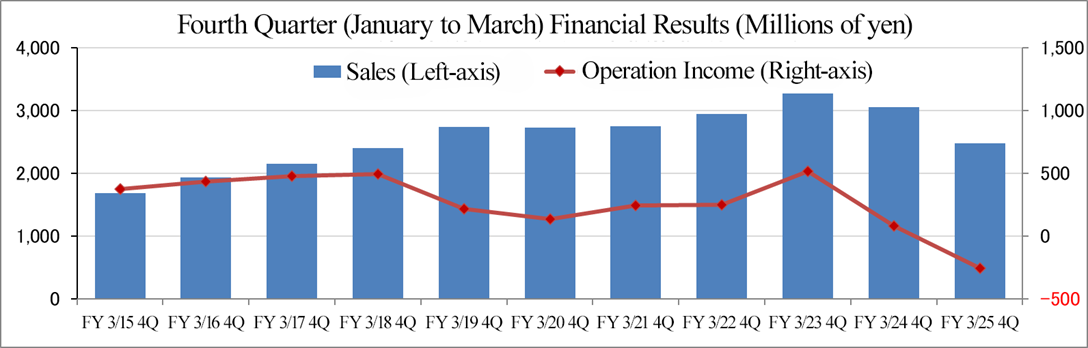
In the fourth quarter (January to March) of the fiscal year ended March 2025, the company recorded an operating loss due mainly to a sales decline in Japan.
(5) Financial Conditions and Cash Flow(CF)
Financial Conditions
| March 2024 | March 2025 |
| March 2024 | March 2025 |
Cash | 7,465 | 7,039 | ST Interest-Bearing Liabilities | 1,093 | 1,000 |
Receivables and contract assets | 3,463 | 2,774 | Payables, Accrued Expenses | 1,196 | 922 |
Advance payment | 1,265 | 841 | Advances received | 2,521 | 2,420 |
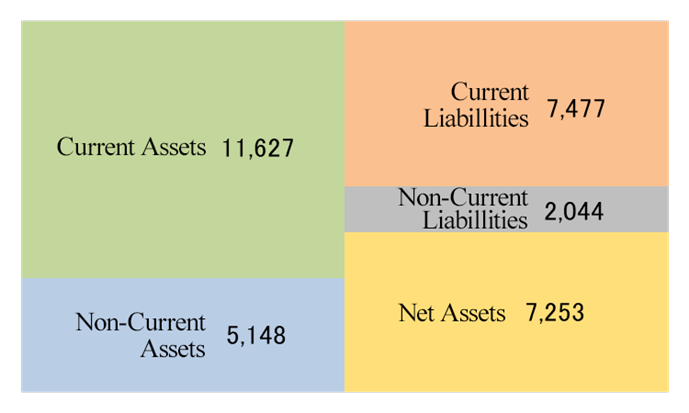
Current Assets | 12,748 | 11,627 | LT Interest-Bearing Liabilities | 1,804 | 1,327 |
Tangible Assets | 518 | 395 | Liabilities | 10,304 | 9,521 |
Intangible Assets | 3,665 | 3,239 | Net Assets | 8,235 | 7,253 |
Investments and Others | 1,607 | 1,512 | Total Liabilities and Net Assets | 18,539 | 16,775 |
Noncurrent Assets | 5,791 | 5,148 | Total Interest-Bearing Liabilities | 2,898 | 2,327 |
* Unit: million yen
* Interest-bearing liabilities=Borrowings + Lease Obligations
*Produced by Investment Bridge Co., Ltd. with reference to disclosed material.
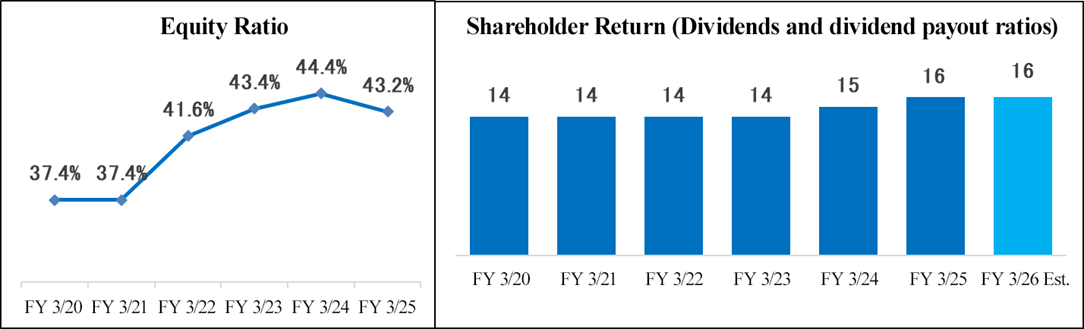
Total assets as of the end of March 2025 stood at 16,775 million yen, down 1,764 million yen from the end of the previous fiscal year. On the asset side of the balance sheet, principally, cash and deposits, accounts receivable, advance payment, and goodwill went down while other current assets and long-term prepaid expenses grew. On the liabilities and net assets side, chiefly, short-term and long-term debts, income taxes payable, and retained earnings went down, and deposits received grew. The equity ratio as of the end of March 2025 was 43.2%, down 1.2 percentage points from the end of the previous fiscal year.
Cash Flow |
|
|
| |
| FY3/24 | FY3/25 | YoY | |
Operating cash flow(A) | 1,065 | 595 | -469 | -44.1% |
Investing cash flow(B) | -28 | -45 | -16 | - |
Free cash flow(A+B) | 1,036 | 550 | -486 | -46.9% |
Financing cash flow | -960 | -939 | 21 | - |
Cash and Equivalentsat the end of period | 7,465 | 7,039 | -425 | -5.7% |
* Unit: million yen

Regarding cash flows, the cash inflow from operating activities shrank chiefly because the net loss before taxes and other adjustments was recorded and the decrease in accounts payable. The surplus of free cash flow decreased mainly because the cash outflow from investing activities augmented for reasons such as a rise in expenditure resulting from the acquisition of long-term prepaid expenses and a drop in income from distributions from investment partnerships. In addition, cash outflow from financing activities shrank, due to the decrease in expenditure for repaying long-term debt. As a result, the cash position as of the end of March 2025 was down 5.7% year on year.
(6) Major financial indicators
Operating income margin and ROE
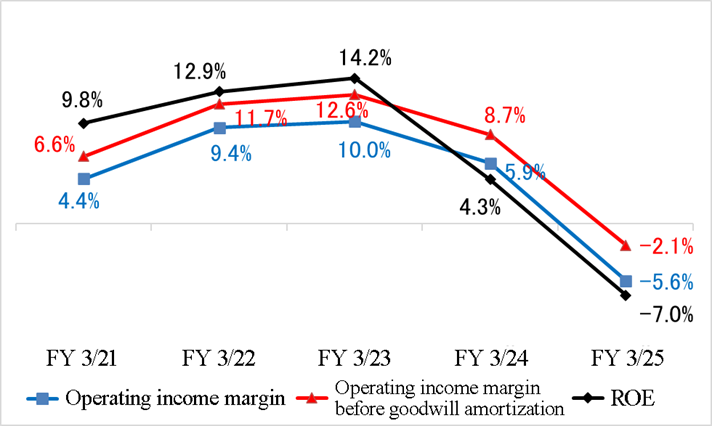
These indicators worsened due to the decreases in sales and profit caused by the cancellation, suspension, etc. of projects in fiscal year ended March 2024. In fiscal year ending March 2026, they aim to move into the black by completing the compensation for the cancelled and suspended projects.
4. Fiscal Year ending March 2026 Earnings Forecasts
(1) Consolidated results
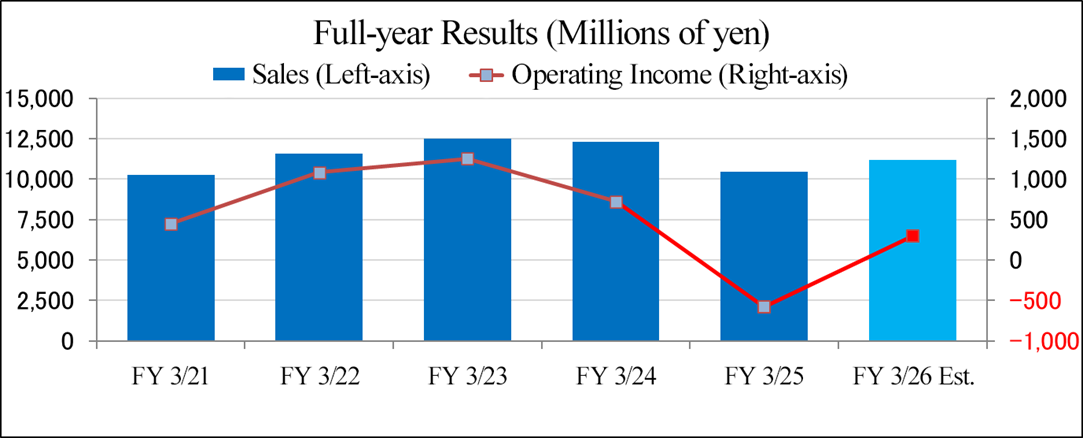
| FY 3/25 Act. | Ratio to sales | FY 3/26 Est. | Ratio to sales | YoY |
Sales | 10,437 | 100.0% | 11,200 | 100.0% | +7.3% |
Operating income | -583 | -5.6% | 300 | 2.7% | - |
Ordinary income | -498 | -4.8% | 320 | 2.9% | - |
Profit attributable to owners of parent | -539 | -5.2% | 150 | 1.3% | - |
*Unit: million yen
A 7.3% increase in revenue from the previous fiscal year, with an operating income of 300 million yen.
The company's plan for the fiscal year ending March 2026 forecasts sales of 11.2 billion yen, up 7.3% year on year, and an operating income of 300 million yen, up 883 million yen year on year.
The company expects increased revenue and profit due to the growth in the United States and the recovery in Europe, Japan, and other Asian countries. In Asia, Japan is the main region, where the business environment remains challenging, as some new medicines are still to be approved and cannot be used in Japan. However, global sales activities have led to an increase of inquiries about new projects from both domestic and overseas pharmaceutical companies. Therefore, the company expects to see a recovery in performance due to the receipt of orders for multiple international collaborative trials, including those in Japan. While the section in South Korea continues to experience delays in existing projects due to medical strikes and other factors, it is seeing an increase of orders for new projects, including data management and statistical analysis, from Japan, Taiwan, and other countries, which are contributing to revenue growth. The company aims to improve its performance by receiving orders for a large number of new projects proposed by companies in South Korea. The section in Taiwan has received orders for several new projects through sales activities targeting pharmaceutical companies in Taiwan and overseas, and expects a recovery in performance. In the United States, while existing large-scale projects are expected to be completed as scheduled, demand for new drug development in the U.S. market remains strong, with numerous inquiries for new projects, including large-scale ones. The company aims to recover its order backlog by strengthening its sales activities and securing these projects. The U.S. market is the most important for the company's business, and it will further strengthen cooperation with other bases and enhance its order-taking capabilities through investment in human resources and systems to achieve sustainable growth. The section in Europe is making progress in securing orders for new projects by promoting collaboration with its U.S. business, and by further strengthening global synergies in sales, it will expand its ability to secure orders for new projects from U.S. companies, including those in Europe.
Operating income margin is forecast to be 2.7%, up 8.3 points from the previous fiscal year.
In addition, the dividend is expected to be 16 yen/share, unchanged from the previous fiscal year.
*Produced by Investment Bridge Co., Ltd. with reference to disclosed material.
(2) Regional Outlook and Strategy
Japan | ◆The challenging business environment as some new medicines are still to be approved and cannot be used in Japan ◆On the other hand, as a result of sales activities, there has been an increase of inquiries about new projects from domestic and overseas pharmaceutical companies. ◆The company aims to receive more orders by expanding its range of services, such as early-phase clinical trial operations and FSP model operations. |
Other Asian countries | ◆Although South Korea continues to be affected by the medical strike, new orders are increasing, mainly for data management and statistical analysis. ◆The section in China is receiving orders for new projects from Japanese companies, while the section in Taiwan is receiving orders for new projects from both domestic and overseas sources. |
United States | ◆Existing large-scale projects are expected to be completed as scheduled. ◆There are many inquiries for new projects, including large-scale ones, and the company will continue to strengthen its sales efforts to secure them. ◆The impact of the presidential decree on drug price reductions remains unclear, and future developments will be closely monitored. |
Europe | ◆They are receiving orders through strengthened cooperation with the section in the United States. ◆The company will continue its global sales activities with the aim of securing global jointly conducted clinical trials. |
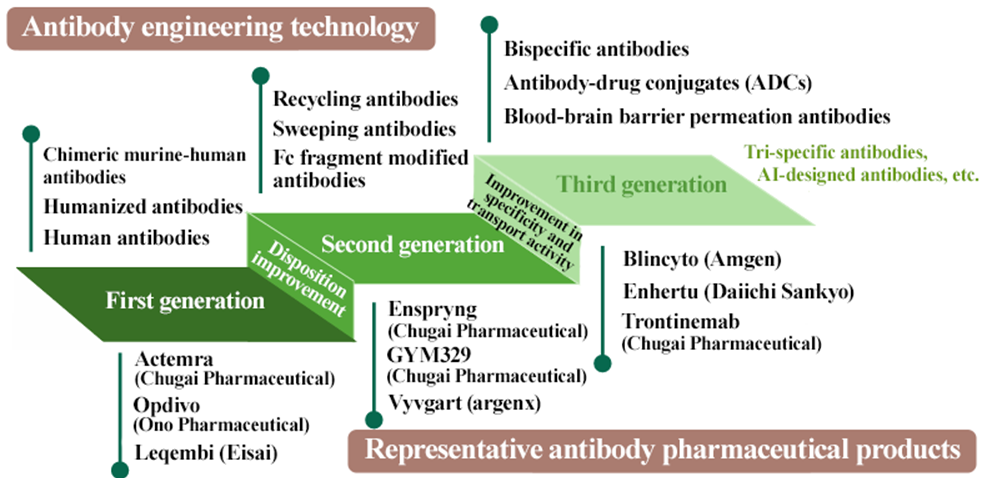
(3) Increasing the development of evolving antibody drugs and strengthening the capability of dealing with them
The company plans to actively engage in the antibody drug field, which is expected to experience rapid growth in the future.
(Source: Linical)
(4) Dividend Policy
The company is also actively working to strengthen its financial base. In order to secure the financial resources necessary for investments to expand overseas operations, the company will ensure sustainable growth in earnings per share by increasing revenue through its growth strategy, maintaining high operating rates, and thoroughly managing costs. At the same time, the company aims to increase its current ratio and equity ratio, enable flexible financing, and strive to balance shareholder returns and securing funds for growth.
5. Conclusions
The financial results of Linical in fiscal year ended March 2025 were very severe, as sales dropped 15.2% year on year and an operating loss of 583 million yen was posted. Sales declined significantly from the previous fiscal year, mainly because they were not able to compensate for the impact of the cancellation of existing large-scale projects, the shortening of their periods, and contract revisions in the previous fiscal year, although they undertook multiple large-scale trials in Japan from domestic and overseas clients in the second half of the fiscal year. Linical is cash-rich and has a healthy financial standing, so the deficit in a single fiscal year won’t affect their business, but these results are very disappointing, failing to meet the expectations of the market. Amid this situation, many signs of recovery were observed. In Japan, where the company is struggling the most, order backlog at the end of the fiscal year was larger than that at the end of the previous fiscal year, as they secured multiple new projects and revised contracts. While they have received informal orders from Japanese pharmaceutical companies for projects for which contracts are still to be signed, there are an increasing number of inquiries for new projects, including large-scale international collaborative trials. In the U.S., which is the largest market, year-end order backlog dropped from the previous fiscal year, but there are new projects for which contracts are to be signed and they have received many inquiries for global projects, mainly from biotech firms. In Europe, too, year-end order backlog declined from the previous fiscal year, but through the cooperation with the U.S. business, they are receiving orders for new projects, and there exist prospective projects that are not included in year-end order backlog. In Asia, too, year-end order backlog increased from the previous fiscal year, as they concluded contracts for multiple new projects for data management and statistical analysis with companies in Taiwan and China, although the recording of sales from existing projects and the receipt of new orders in South Korea did not progress as assumed due to the medical strike in South Korea. Order backlog is the most noteworthy management indicator, because it is a leading indicator of their business performance. The company aims to fortify a foothold for recovery in fiscal year ending March 2026, and achieve a V-shaped recovery of performance in fiscal year ending March 2027 and fiscal year ending March 2028. It is noteworthy how much they will be able to increase order backlog for business performance recovery.
As growth strategies in fiscal year ending March 2026, they uphold the enhancement of corporate governance, marketing, and investment in IT. For corporate governance, they will reform and strengthen the organizational structure in order to improve profitability while expanding services. For marketing, they will strengthen the capability of giving well-designed proposals to clients to differentiate themselves from leading global CROs. For investment in IT, they will meet the needs for streamlining of clinical trials and improve the efficiency of business administration by utilizing digital technologies. It is noteworthy whether they can see the outcomes of the growth strategies early as they mentioned.
Furthermore, they will actively enter the field of antibody drugs, which are expected to grow rapidly. We would like to pay attention to the progress of order receipt in the field of antibody drugs with expectation, although it may take some time.
<Reference: Regarding Corporate Governance>
◎ Organization type, and the composition of directors and auditors
Organization type | Company with an Audit & Supervisory Committee |
Directors who are not Audit Committee Members | 6 directors, including 5 outside ones |
Directors who are Audit Committee Members | 3 directors. All of them are outside directors |
◎ Corporate Governance Report
Last updated on July 2, 2024
<Basic Policy>
(1) Management Philosophy
Our management philosophy is “To promote the greater wellbeing of all our stakeholders — patients, business partners, shareholders, and employees — we strive constantly to offer professional, high-quality services to support all aspects of new drug development.” We aim to contribute to the development, evolution and diffusion of new therapeutic technologies including new pharmaceuticals, and ultimately to the healthy lives of human beings, by continuously developing and maintaining the knowledge and experience of our executives and employees, as well as the know-how and systems of our organization.
(2) Basic Approach on Corporate Governance
Based on the above management philosophy, our company will contribute to the birth and growth of new disease prevention and therapeutic technologies, including new pharmaceuticals, with our know-how and technologies in pharmaceutical development. As a partner of healthcare companies and medical institutions, including domestic and foreign bio-venture firms, pharmaceutical companies, and medical device manufacturers, our company will contribute to the development of healthcare and meet the expectations of patients and the entire society.
Since our business activities impact people's lives, our executives and employees are required to have high ethical standards as well as expertise. Thus, we thoroughly comply with the Corporate Code of Conduct, including strict compliance with laws. In addition, we strive to improve corporate value and business development by enhancing internal control and ensuring the soundness and transparency of management.
<Regarding the implementation of the principles of the corporate governance code>
Major principles for not implementing and the reasons
Principles | Reasons for not implementing the principles |
[Supplementary Principle 4-1 (2) Medium-term Management Plan] | The Medium-term Management Plan of the company is reviewed by the Management Board, with progress checked and analyzed at each meeting, reviewing the medium-term targets and policies as necessary and appropriate. The Board of Directors approves the Medium-term Management Plan formulated by the Management Board while receiving reports on progress and analysis results, while monitoring and supervising the plan. In December 2021, the company announced a three-year Medium-term Management Plan ending in the fiscal year ending March 2025. In the future, the company will consider revising its targets and policies as required based on progress, disclosing and explaining them together with its vision and management strategies to develop a common understanding with its shareholders and investors. |
[Supplementary Principle 4-2 (1) Compensation System] | In addition to fixed compensation, the company has adopted a performance-based compensation system, a monetary compensation linked to performance over a single fiscal year for directors responsible for executing the company's business. On the other hand, the most executive officers (CxOs) are founding members of the company and already possess a certain number of the company's shares. Therefore, the increase or decrease in shareholder value reflecting medium to long-term performance is linked to the increase or decrease in the value of the shares possessed, which virtually includes incentives similar to medium to long-term performance-based compensation, enabling mutual interest and value-sharing with the shareholders. In this context, no non-monetary compensation, such as share-based compensation linked to medium to long-term performance, has been established. Furthermore, the company will consider necessary revisions to its executive compensation system, including medium to long-term performance-based compensation, in line with changes in the composition of the board of directors, including the appointment of directors other than founding members who will be responsible for business execution in the future. |
[Principle 4-9 Criteria and Eligibility for Independence of Independent Outside Directors] | In addition to the requirements of the Companies Act, the Board of Directors appoints candidates who it believes, based on their knowledge and experience, can actively provide appropriate opinions regarding the management of the company and other matters based on the same objective perspective as ordinary shareholders, after confirming that they substantially meet the criteria of the Tokyo Stock Exchange for determining the independence of independent directors. |
<Disclosure Based on the Principles of the Corporate Governance Code (Excerpts)>
Principles | Disclosure contents |
[Supplementary Principle 2-4 (1) Ensuring diversity in the appointment of core human resources] [Supplementary Principle 3-1 (3) Initiatives for Sustainability]
| The corporate group has formulated a “Sustainability Policy” based on its management philosophy and promotes sustainability management in line with this policy. Sustainability-related initiatives and policies on human resources development, including ensuring the diversity of human resources, and policies on the development of the internal environment, are disclosed in the Annual Securities Report under “2. Policies and Initiatives on Sustainability.” The status regarding the promotion of diversity in the core workforce is as follows. (1) Women The promotion of female managers is progressing at the headquarters in Japan as well as throughout the group, and we will further improve the working environment and provide career development support to develop female leaders at the executive officer level and above, who will play core management roles in the future. [Headquarters (Japan)] Female employees/all employees (%) 61.5% in 2024, 62.9% in 2023, 61.6% in 2022. Female managerial positions/All managerial positions (excluding executive officers) (%) 42.6% in 2024, 44.2% in 2023, 42.6% in 2022. Female Executive Officers/All Executive Officers (%) 16.7% in 2024, 16.7% in 2023, 16.7% in 2022. [Corporate Group] Female employees/all employees (%) 68.1% in 2024, 68.5% in 2023, 67.5% in 2022. Female managerial positions/All managerial positions (excluding executive officers) (%) 58.6% in 2024, 59.7% in 2023, 56.9% in 2022. Female Executive Officers/All Executive Officers (%) 27.8% in 2024, 31.8% in 2023, 28.6% in 2022. (2) Approximately 50% of the group’s 662 employees (as of March 31, 2024) are locally hired employees residing overseas, and the key positions in the overseas group companies are occupied by highly qualified local human resources. As drug development is getting more and more borderless, we will endeavor to recruit talented human resources with foreign nationality. (3) Mid-career hires: The percentage of mid-career hires among all the employees working at our headquarters in Japan is 48.4% as of the end of March 2024. Mid-career hires account for 100% of the executive officers and 70.2% of the employees who hold managerial positions. For our corporate group as a whole, mid-career hires make up for 71.0% of all the employees as of the end of March 2024. The percentages of mid-career hires among the executive officers and the employees who took up managerial positions are 88.9% and 80.5%, respectively. |
[Principle 3-1 Enhancement of Disclosure of Information] | (i) Our company’s Goals (Management Philosophy, etc.), Management Strategy, and Management Plan Our management philosophy is “To promote the greater wellbeing of all our stakeholders — patients, business partners, shareholders, and employees — we strive constantly to offer professional, high-quality services to support all aspects of new drug development,” and we aim to achieve sustainable growth and to improve corporate value over the medium/long term. In order to achieve this, the company has established a three-year Medium-term Management Plan ending in the fiscal year ending March 2025, which was announced in December 2021. Details of the management strategy and management plan are disclosed in the Annual Securities Report and other documents. (ii) Basic Approach and Basic Policy on Corporate Governance Based on Each of the Principles in this Code Our basic approach on corporate governance is described in “I. Basic Approach” of this report. An overview of our company’s corporate governance, including the above, is disclosed on our company’s website. (iii) Policies and Procedures for the Board of Directors in Determining Remuneration for Executives and Directors Compensation of the company's directors shall be paid within the limits of the total compensation approved by the General Meeting of Shareholders. The Compensation Committee, which consists of 3 or more members, the majority of whom are outside directors, discusses and reports on the decisions and procedures of such policies in consultation with the Board of Directors, to ensure objectivity, transparency, and fairness. Further details are disclosed in “4. Status of corporate governance, (4) Compensation of directors and officers” in Annual Securities Report. (iv) Policies and Procedures for the Board of Directors’ Selection and Dismissal of Executives and Nomination of Candidates for Directors and Executive Officers The appointment and nomination of candidates for executive directors and executive officers are made through the resolution of the Board of Directors based on the candidates' insight and integrity of character appropriate for senior management with regard to compliance with laws, regulations, and corporate ethics, their ability to make decisions accurately and promptly, as well as individual knowledge, experience and ability, taking into consideration the overall balance between the Board of Directors, including outside directors, and the management team as a whole. In addition, the reappointment (or non-reappointment) is resolved by the Board of Directors, based on whether the expected performance and results have been achieved on a constant basis. Candidates for outside directors who are not Audit & Supervisory Committee members shall be appointed and reappointed based on a resolution by the Board of Directors according to the criteria and qualities set out in Principle 4-9. Candidates for directors who serve as Audit & Supervisory Committee members shall be appointed and reappointed based on a resolution by the Board of Directors not only according to the criteria and qualities defined in Principle 4-9, but also by including at least one person who has sufficient knowledge of finance and accounting and obtaining the consent of the Audit & Supervisory Committee while taking into consideration the balance of the Audit & Supervisory Committee so that the committee can audit the business management properly. We will enhance the objectivity, transparency, and fairness of the resolutions made by the Board of Directors on these matters through a process of discussion and reporting regarding candidates by the Nomination Committee consisting of at least three members, of which outside directors constitute the majority. Furthermore, the Board of Directors appoints and dismisses the representative director, president and CEO through a process of discussion and reporting by the Nomination Committee while comprehensively considering such matters as whether our company can respond to changes in the overall business environment, proactively formulate and promote business strategies, and continuously improve our business performance on the premise that appointment and dismissal of the representative director, president and CEO are some of the most crucial decisions to make. Candidates to succeed the representative director, president and CEO are being developed through knowledge training and planned rotations. (v) Explanation of Individual Election and Dismissal of Executives, and Nomination of Candidates for Directors and Corporate Auditors by the Board of Directors Based on the (iv) Above With regard to the nomination of candidates for non-audit committee members and audit committee members as directors, the professional backgrounds and the reasons for the nomination of each candidate shall be stated in the Notice of Convocation of the General Meeting of Shareholders.<Supplementary Principle 4-1 (1) Scope of the delegation to the management> Our company has adopted the executive officer system and established the position of Chief x Officer (CxO) in order to separate supervision of the business management and business execution and swiftly make business decisions. The scopes of matters that the management discusses with the Board of Directors, matters that the management reports to the Board of Directors, and matters that are delegated to the management have been specified in our internal regulations. The Board of Directors makes decisions on matters that are crucial for our business, and decision-making on other business execution that can be delegated in accordance with the law has been delegated to the representative director, president and CEO at our company. |
[Principle 5-1 Policy for constructive dialogue with shareholders] | Through constructive dialogue with shareholders (including institutional and individual investors as potential shareholders), our company aims for sustainable growth in corporate value, which is the common goal of our company and shareholders. Our company is committed to strengthening accountability, is continuously promoting enhanced disclosure of information, and is promoting dialogue with investors in Japan and overseas. The company has continuous, constructive, transparent, fair dialogue regarding business performance, managerial strategies, capital policies, risks, corporate governance systems, etc. with the following method. ・Dialogue with shareholders is led by the Executive Officer CFO. Considering the purpose and effect of the interview, and the attributes of shareholders, the dialogue method is examined thoroughly by the senior management such as CEO and the Executive Officer CFO. ・As for IR, mainly the financial affairs department and the public relations division gather necessary information from relevant sections of the company, prepare reference material and give explanations in an understandable manner, to enrich the dialogue with shareholders.・In addition to the Ordinary General Meeting of Shareholders, financial results briefings, and briefings for individual investors, our company provides opportunities for the dialogue through individual meetings with domestic and overseas institutional investors, by disclosing IR information on our company’s website, including English versions, through phone calls and emails from individual investors, and reflects questions, requests, information on participants at briefings, and survey results in our IR activities. ・Shareholders’ interests and concerns grasped through the dialogue with them are reported to the Executive Officer CFO and the information is utilized for analyzing business administration, discussing how to disclose information, etc.・Concerning IR activities and the dialogue with shareholders, the company manages insider information appropriately in accordance with in-company rules. The quiet period, in which the company refrains from having dialogue about financial results, is from the day after the closing date of each quarter to the date of brief reporting.
[Measures for achieving a business management conscious of cost of capital and share price (under consideration)] While defining Earnings per Share (EPS) as our business indicator and discloses it via financial results briefing material and our website, our company is currently considering what information to disclose regarding policies, targets, and initiatives. |
This report is not intended for soliciting or promoting investment activities or offering any advice on investment or the like, but for providing information only. The information included in this report was taken from sources considered reliable by our company. Our company will not guarantee the accuracy, integrity, or appropriateness of information or opinions in this report. Our company will not assume any responsibility for expenses, damages or the like arising out of the use of this report or information obtained from this report. All kinds of rights related to this report belong to Investment Bridge Co., Ltd. The contents, etc. of this report may be revised without notice. Please make an investment decision on your own judgment. Copyright(C) Investment Bridge Co., Ltd. All Rights Reserved. |


